Perfecting the Carbon Nanotube Forest James Harper Robert

Perfecting the Carbon Nanotube Forest James Harper Robert Mifflin Advisors: Prof. Prab Bandaru Prof. Sung. Ho Jin Prof. Frank Talke June 7 th, 2007 Jacobs School of Engineering University of California – San Diego

Outline Introduction ◦ Selecting the Area of Nanotechnology to Enhance Why is this area important? Does it pass the Moral / Ethics Test? ◦ Background Growth and Chirality Separation Techniques Analyses of Separation Techniques Creating Pure Lines of Carbon Nanotubes Conclusion ◦ Dielectrophoresis ◦ Flow Fractionalization Analysis and Improvement ◦ Pulsed dielectrophoresis ◦ Selection and Release ◦ The Perfect Carbon Nanotube Forest

Area of Nanotechnology to Enhance Generating Pure Sets of CNTs on Demand ◦ Why is this area important? [1]

Area of Nanotechnology to Enhance Carbon nanotubes can be used to enhance materials and create new sensors that impact everyday life Electrical arena Wires, Batteries and Capacitors, [2] [3] Flex displays [4]

Area of Nanotechnology to Enhance Carbon nanotubes can be used to enhance materials and create new sensors that impact everyday life Electrical arena ◦ Conductive plastics, adhesives Structural Arena ◦ Adhesives, [5] Flexible circuits, [6] Composites [7]

Area of Nanotechnology to Enhance Carbon nanotubes can be used to enhance materials and create new sensors that impact everyday life Electrical arena ◦ Conductive plastics, adhesives Structural Arena ◦ Adhesives, textiles, composites Bio-molecule sensing [8] [9] [10]

Area of Nanotechnology to Enhance ◦ Does it pass the Moral / Ethics Test? [11]

Background Growth and Chirality of Carbon Nanotubes ◦ Formed from several processes, resulting in a sheet of graphene in the form of a hollow continuous tube. ◦ Differences between SWCNT, M-SWCNT, S-SWCNT – Single Wall CNT MWCNT – Multi-Wall CNT M-SWCNT – Metallic SWCNT S-SWCNT – Semiconductor SWCNT [12]
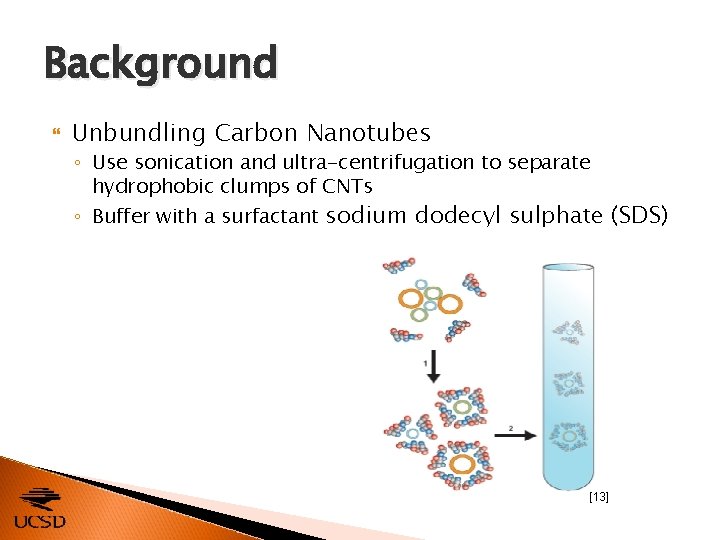
Background Unbundling Carbon Nanotubes ◦ Use sonication and ultra-centrifugation to separate hydrophobic clumps of CNTs ◦ Buffer with a surfactant sodium dodecyl sulphate (SDS) [13]

Background Unbundling Carbon Nanotubes ◦ Use sonication and ultra-centrifugation to separate hydrophobic clumps of CNTs ◦ Buffer with a surfactant sodium dodecyl sulphate (SDS) Purification / Sorting Techniques ◦ ◦ Ultra-centrifugation Optical sorting Fluid flow fractionalization Dielectrophoresis
![Background Purification / Sorting Techniques ◦ Ultra-centrifugation [14] [15] Background Purification / Sorting Techniques ◦ Ultra-centrifugation [14] [15]](http://slidetodoc.com/presentation_image_h/2c0f356718546856259b2b127522c83f/image-11.jpg)
Background Purification / Sorting Techniques ◦ Ultra-centrifugation [14] [15]
![Background Purification / Sorting Techniques ◦ Optical sorting [16] Background Purification / Sorting Techniques ◦ Optical sorting [16]](http://slidetodoc.com/presentation_image_h/2c0f356718546856259b2b127522c83f/image-12.jpg)
Background Purification / Sorting Techniques ◦ Optical sorting [16]
![Background Purification / Sorting Techniques ◦ Optical sorting [28] Background Purification / Sorting Techniques ◦ Optical sorting [28]](http://slidetodoc.com/presentation_image_h/2c0f356718546856259b2b127522c83f/image-13.jpg)
Background Purification / Sorting Techniques ◦ Optical sorting [28]
![Background Purification / Sorting Techniques ◦ Fluid flow fractionalization [29] Background Purification / Sorting Techniques ◦ Fluid flow fractionalization [29]](http://slidetodoc.com/presentation_image_h/2c0f356718546856259b2b127522c83f/image-14.jpg)
Background Purification / Sorting Techniques ◦ Fluid flow fractionalization [29]
![Background Purification / Sorting Techniques ◦ Dielectrophoresis [30] Background Purification / Sorting Techniques ◦ Dielectrophoresis [30]](http://slidetodoc.com/presentation_image_h/2c0f356718546856259b2b127522c83f/image-15.jpg)
Background Purification / Sorting Techniques ◦ Dielectrophoresis [30]

Background Unbundling Carbon Nanotubes ◦ Use sonication to separate clumps and ultra-centrifugation Purification / Sorting Techniques ◦ ◦ Ultra-centrifugation Optical sorting Fluid flow fractionalization Dielectrophoresis Problem? ◦ Each technique allows for partial separation of the desired carbon nanotubes from the bulk solution – However ……

Background There is an overlap of sorting parameters! ◦ Use of one technique independently will not discriminate nanotubes with overlapping parameters [17]
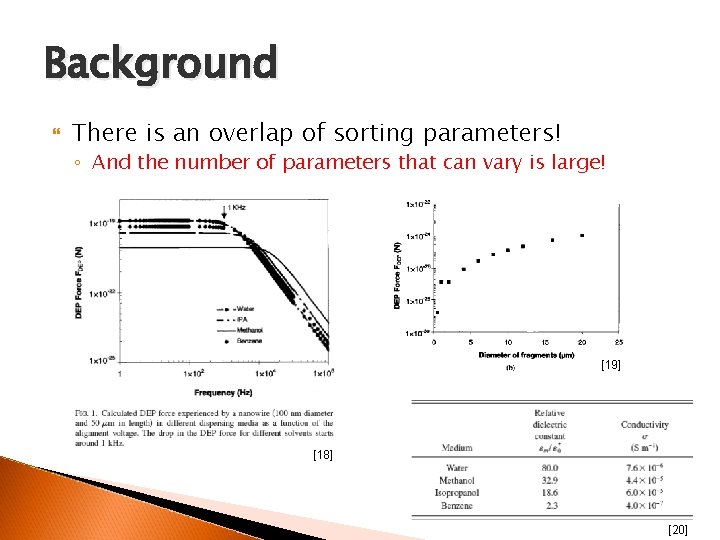
Background There is an overlap of sorting parameters! ◦ And the number of parameters that can vary is large! [19] [18] [20]

Background Partial Solution? ◦ Multiple techniques must be used for to obtain a rough sort of the material. Ultra-centrifugation Optical sorting Fluid flow fractionalization Dielectrophoresis And the resulting subset will still have a mixture of different nanotubes - albeit a set with many overlapping attributes.

Solution Realize that absolute purity of nanotubes through top down or bottom up fabrication may not be achievable. Recast the problem – what other system/industry has high variability – yet desires near exact to exact duplicates be used?

Solution Look to the Bio Labs – ◦ Generating a clone murine line for laboratory study. Bio Process Select an species Isolate the individual Sequence the DNA ------- Maps to CNT – rough sort desired CNTs CNT – individual capture Check the Chirality – using Raman scattering Release the individual --- Release the individual CNT Clone the individual --- Clone the CNT (into a controlled environment) and conduction properties [21]

Solution ◦ Can all of these steps be done? ◦ If so, perfect sorting may not be required.

Outline Introduction ◦ Selecting the Area of Nanotechnology to Enhance Why is this area important? Does it pass the Moral / Ethics Test? ◦ Background Growth and Chirality Separation Techniques Analyses of Separation Techniques Creating Pure Lines of Carbon Nanotubes Conclusion ◦ Dielectrophoresis ◦ Flow Fractionalization Analysis and Improvement ◦ Pulsed dielectrophoresis ◦ Selection and Release ◦ The Perfect Carbon Nanotube Forest

Dielectrophoresis Uncharged particle + non-uniform electric field = force ◦ Caused by uneven charge distribution ◦ Depends strongly on… Medium’s and particles' electrical properties Particles' morphology Frequency of the electric field More polarizable particles move toward stronger electric field For CNTs, where , and
Dielectrophoresis +++++++ -- ----- + + --- -
![Dielectrophoresis [22] [23] Dielectrophoresis [22] [23]](http://slidetodoc.com/presentation_image_h/2c0f356718546856259b2b127522c83f/image-26.jpg)
Dielectrophoresis [22] [23]

Flow Fractionalization Analysis and Improvement CNTs with dissimilar conductivities and morphologies develop different terminal velocities within a fluid flow, as described by Separation is most efficient when v. T of different sizes of CNTs is most dissimilar. ◦ Adjust friction factor f by changing orientation [24]
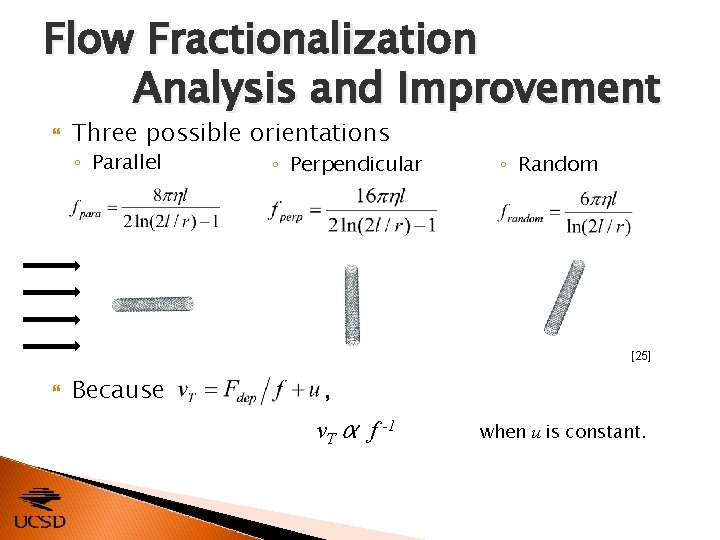
Flow Fractionalization Analysis and Improvement Three possible orientations ◦ Parallel ◦ Perpendicular ◦ Random [25] Because , v. T α f -1 when u is constant.
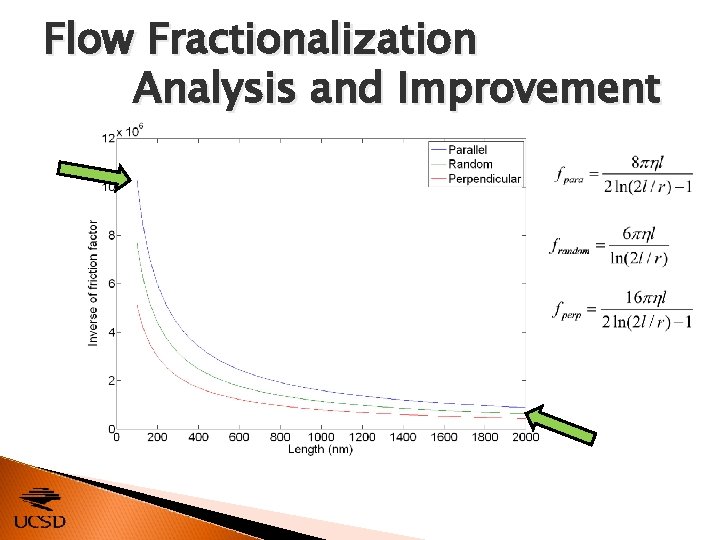
Flow Fractionalization Analysis and Improvement
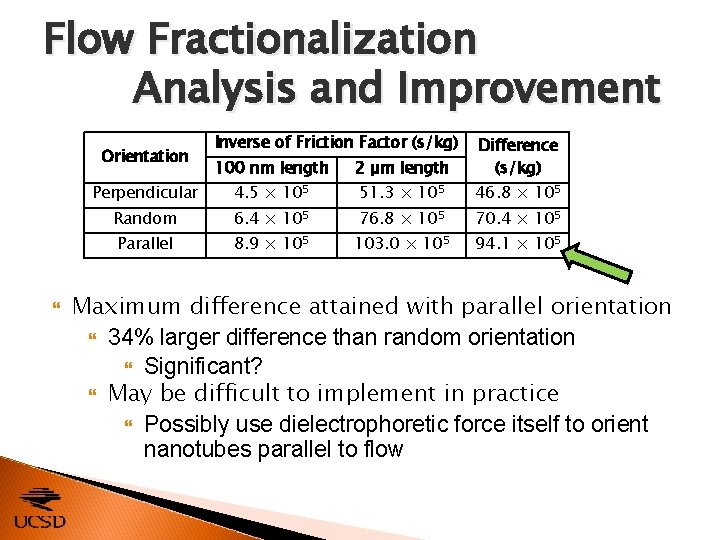
Flow Fractionalization Analysis and Improvement 100 nm length 2 μm length Difference (s/kg) Perpendicular 4. 5 × 105 51. 3 × 105 46. 8 × 105 Random 6. 4 × 105 76. 8 × 105 70. 4 × 105 Parallel 8. 9 × 105 103. 0 × 105 94. 1 × 105 Orientation Inverse of Friction Factor (s/kg) Maximum difference attained with parallel orientation 34% larger difference than random orientation Significant? May be difficult to implement in practice Possibly use dielectrophoretic force itself to orient nanotubes parallel to flow

Pulsed Dielectrophoresis Difference between Dielectrophoresis (DEP) and Pulsed Dielectrophoresis (PDEP) ◦ DEP is typically set up for an asymmetrical field with constant frequency. We would like to look at varying the duty cycle to try to separate CNT that have very closely overlapping properties. ◦ Example - Lets look at some cells Distributed populations of spherical shell models of mammalian cells. (Top Left) 10% variation across all three DEP parameters, radius, permittivity, and conductivity. (Top Right) Constant conductivity with varying permittivity and radius. (Lower Left) Constant radius. (Lower Right) Constant permittivity.

Pulsed Dielectrophoresis Difference between Dielectrophoresis (DEP) and Pulsed Dielectrophoresis (PDEP) Distributed populations of spherical shell models of mammalian cells. (Top Left) 10% variation across all three DEP parameters, radius, permittivity, and conductivity. (Top Right) Constant conductivity with varying permittivity and radius. (Lower Left) Constant radius. (Lower Right) Constant permittivity.

Pulsed Dielectrophoresis The equations Complex Permittivity of CNT Metallic = 2000 Media = 18. 6 Modified Clausius Mossotti E field between electrodes volts per meter And the friction factor
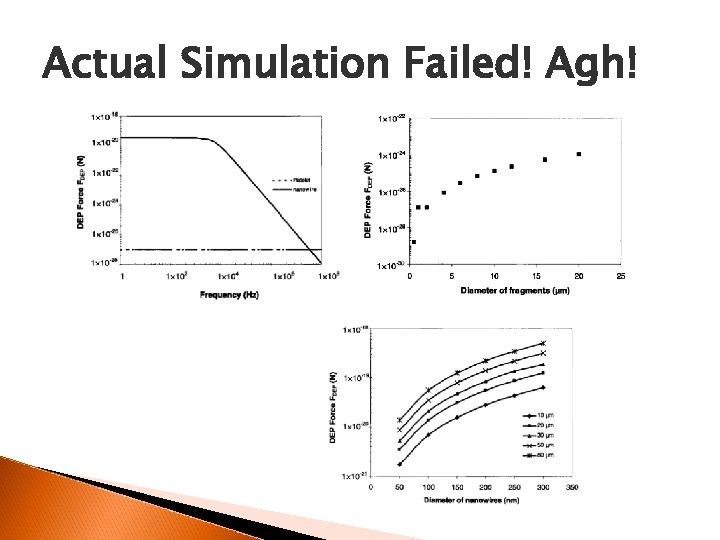
Actual Simulation Failed! Agh!

Capture of subset of CNTs Capture occurs due to Dielectrophoresis attracting the CNT dipoles. ◦ CNT lands on the probes and causes the field to be modified ◦ Thus self assembly/placement Modify the probe surface with LBL deposited material for sticktion and later lift off [26]

Analysis of subset of CNTs Use Raman scattering and conduction parameters to analyze the CNTs Electronic and mechanically stringency wash cartridge.

Release the desired CNTs Decorate CNTs with bio-particles to ease later handling. CNTs are then released as needed from the storage cartridge. Moved to cloning cell off chip [27]

Release the desired CNTs Sonicated into seeds Embedded into an LBL deposited layer Used to grow Final CNTs

Conclusions Sorting of CNTs difficult, yet improvable ◦ Flow fractionalization ◦ Pulsed dielectrophoresis Best solution: avoid problem of perfect sorting with capture and release of CNTs ◦ The perfect carbon nanotube forest
![References Pictures ◦ ◦ ◦ [7] “Carbon nanotubes enter Tour de France. ” CNet. References Pictures ◦ ◦ ◦ [7] “Carbon nanotubes enter Tour de France. ” CNet.](http://slidetodoc.com/presentation_image_h/2c0f356718546856259b2b127522c83f/image-40.jpg)
References Pictures ◦ ◦ ◦ [7] “Carbon nanotubes enter Tour de France. ” CNet. com. ◦ ◦ [10] “Drug Delivery and Biomolecular Transport. ” Nanotubes Monthly. ◦ [24] Dielectrophoresis of carbon nanotubes using microelectrodes: a numerical study. ” Maria Dimaki and Peter Bøggild. MIC–Department of Micro and Nanotechnology, Building 345 East, Technical University of Denmark, DK-2800, Kgs. Lyngby, Denmark. ◦ [1, 2] “The Application of Vertically Aligned Carbon Nanotube Arrays in Electronics and Biosensors” by Dr. Jun Li, NASA Ames Research Center, MS 229 -1, Moffett Field, CA 94035 [8 -9] “ Carbon Nanotube Based Biosensors. ” Massood Z. Atashbar 1, Bruce Bejcek 2, Srikanth Singamaneni 1, and Sandro Santucci. Electrical and Computer Engineering Department, Western Michigan University, Kalamazoo, MI-49008, USA [17 -20] “Simple model for dielectrophoretic alignment of gallium nitride nanowires. ” Abhishek Motayeda et al. Material Science and Engineering Laboratory, National Institute of Standards and Technology, Gaithersburg, Maryland 20899 and Department of Electrical and Computer Engineering, University of Maryland, College Park, Maryland 20742 [29] “High-Speed Integrated Particle Sorters based on Dielectrophoresis. ” J. H. Nieuwenhuis 1, A. Jachimowicz 1, P. Svasek 2, M. J. Vellekoop 1, Industrial Sensor Systems, ISAS, Vienna University of Technology, Gusshausstrasse 27 -29, A 1040, Vienna, Austria, nieuwenhuis@tuwien. ac. at, Ludwig Boltzmann Institute of Biomedical Microtechnology, Vienna, Austria Articles ◦ ◦ ◦ Dielectrophoresis of carbon nanotubes using microelectrodes: a numerical study. ” Maria Dimaki and Peter Bøggild. MIC– Department of Micro and Nanotechnology, Building 345 East, Technical University of Denmark, DK-2800, Kgs. Lyngby, Morgan H and Green N G 2003 AC Electrokinetics: Colloids and Nanoparticles Research Studies Press Ltd p. 76 -77. Pohl, H. A. (1978) Dielectrophoresis, Cambridge University Press, Cambridge Arnold, W. M. and Zimmerman, U. (1982) Z. Naturforsch. 37 c, 908 -915 Mischel, M. , Voss, A. and Pohl, H. A. (1982) J. Biol. Phys. 10, 223 -226. More references available for this document upon request.

Are there any questions?
- Slides: 41