Percutaneous Mitral Valve Repair Coronary Sinus Approach Maurice

















- Slides: 48

Percutaneous Mitral Valve Repair Coronary Sinus Approach Maurice Buchbinder, MD Foundation for Cardiovascular Medicine La Jolla, CA Maurice Buchbinder, MD Foundation for Cardiovascular Medicine

Disclosure. Stents? Why Degradable Speaker’s name: Maurice Buchbinder, MD �I have the following potential conflicts of interest to report: �Consulting �Employment in industry �Stockholder of a healthcare company �Owner of a healthcare company I do not have any potential conflict of interest Maurice Buchbinder, MD Foundation for Cardiovascular Medicine

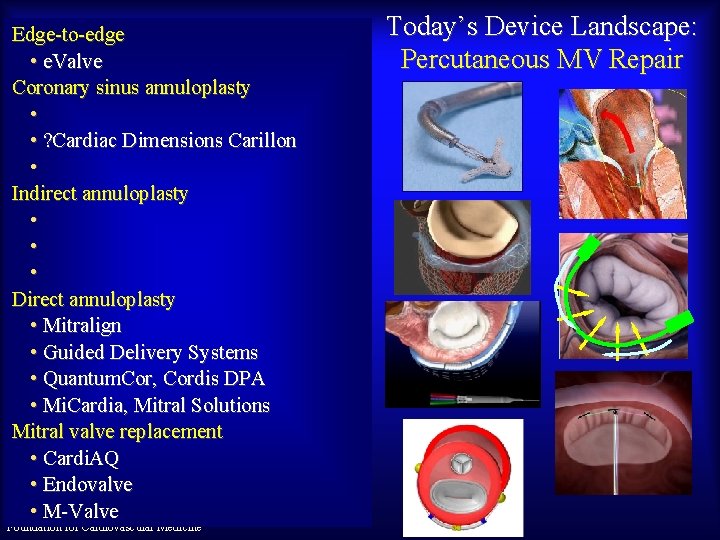
Edge-to-edge • e. Valve • Edwards Mobius Coronary sinus annuloplasty • Edwards Monarc • Cardiac Dimensions Carillon • Viacor PTMA Indirect annuloplasty • Ample PS 3 • Myocor i-Coapsys • St. Jude AAR Direct annuloplasty • Mitralign • Guided Delivery Systems • Quantum. Cor, Cordis DPA • Mi. Cardia, Mitral Solutions Mitral valve replacement • Cardi. AQ • Endovalve Maurice Buchbinder, MD • M- Valve Foundation for Cardiovascular Medicine Device Landscape: Percutaneous MV Repair

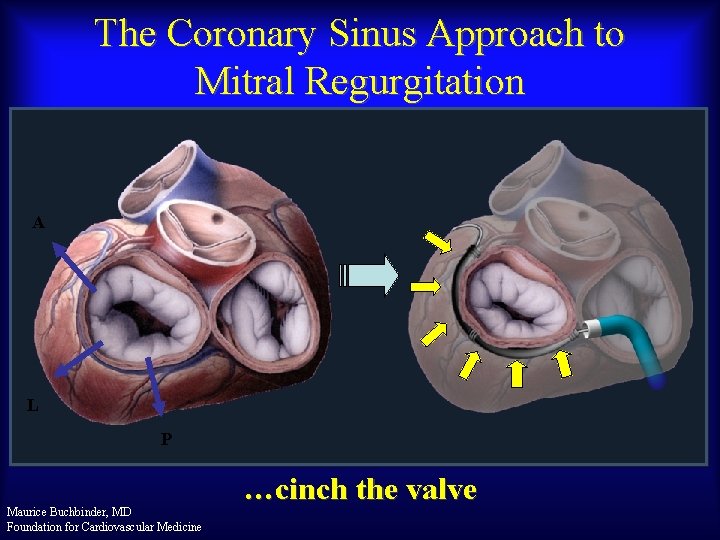
The Coronary Sinus Approach to Mitral Regurgitation A L P Maurice Buchbinder, MD Foundation for Cardiovascular Medicine …cinch the valve

Three Fundamentally Different CS Repair Methods • Edwards-MONARC: Variable length device; chronic cinching/plication of CS between self-expanding stentanchors; IJ access • Cardiac Dimensions-CARILLON: Fixed length device; acute cinching tension/plication of CS between deployable/reconstrainable anchors; IJ access • Viacor-PTMA: Distributed anatomical bending with variable diameter Nitinol rods; subclavian access via pacing lead configuration device Maurice Buchbinder, MD Foundation for Cardiovascular Medicine

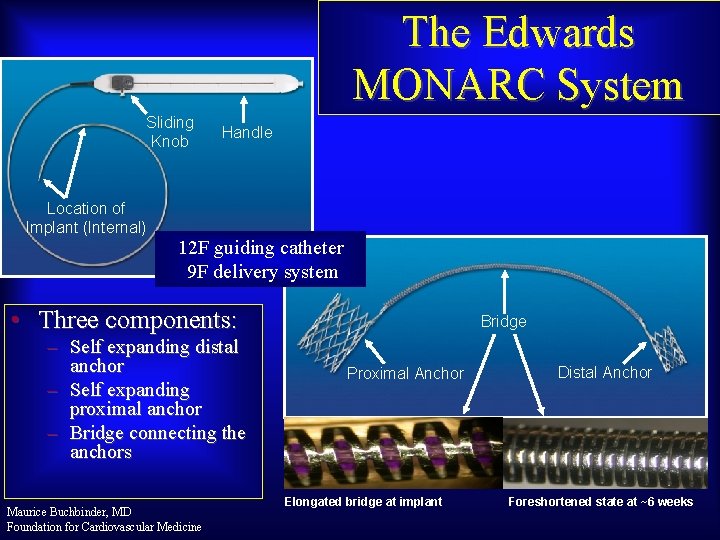
The Edwards MONARC System Sliding Knob Location of Implant (Internal) Handle 12 F guiding catheter 9 F delivery system • Three components: – Self expanding distal anchor – Self expanding proximal anchor – Bridge connecting the anchors Maurice Buchbinder, MD Foundation for Cardiovascular Medicine Bridge Proximal Anchor Elongated bridge at implant Distal Anchor Foreshortened state at ~6 weeks

EVOLUTION Study Overview • Prospective, multi-center feasibility study • Primary objective of the study was to evaluate the acute safety (30 d, 90 d) of the MONARC system in treating functional mitral regurgitation in heart failure patients • Secondary objective of the study is reduction in MR by at least one grade at 90 days Maurice Buchbinder, MD Foundation for Cardiovascular Medicine

Inclusion / Exclusion Criteria INCLUSION • Functional mitral valve regurgitation : dilated or ischemic cardiomyopathy • MR grade 2+ to 4+ • Coronary Sinus Dimensions – Target Area is ≥ 14 cm and ≤ 18 cm in length, – Distal section of Target Area, the AIV is ≥ 3 mm in diameter Maurice Buchbinder, MD Foundation for Cardiovascular Medicine EXCLUSION • Degenerative mitral regurgitation • Ischemia requiring cardiac revascularization within 3 months prior to or planned after the implant procedure • Implanted cardiac defibrillator (ICD) or pacing leads within the coronary sinus • Ejection Fraction < 25% • Moderate to severe mitral annulus calcification

EVOLUTION Study Procedural Success Patients Enrolled n = 72 Device implanted n=59 (82%) • Procedural Time: 84 58 min Maurice Buchbinder, MD Foundation for Cardiovascular Medicine Device not implanted n=13 (18%) • Tortuous Anatomy • Size Outside of Offered Range

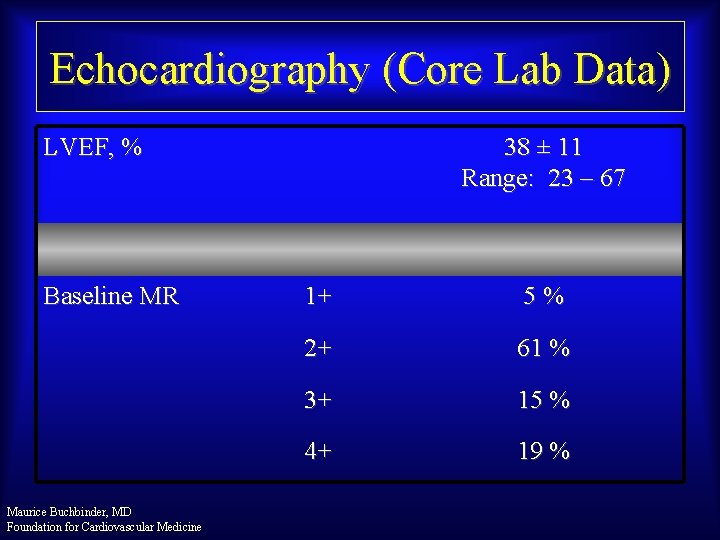
Echocardiography (Core Lab Data) LVEF, % Baseline MR Maurice Buchbinder, MD Foundation for Cardiovascular Medicine 38 ± 11 Range: 23 – 67 1+ 5 % 2+ 61 % 3+ 15 % 4+ 19 %

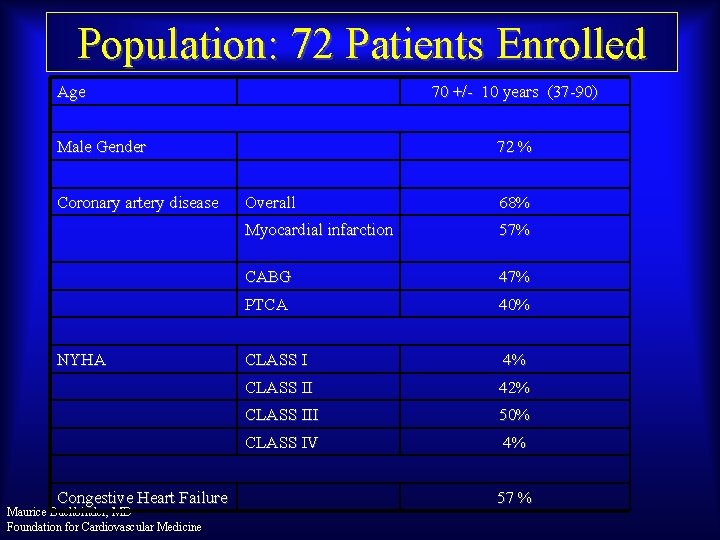
Population: 72 Patients Enrolled Age 70 +/- 10 years (37 -90) Male Gender Coronary artery disease NYHA Congestive Heart Failure Maurice Buchbinder, MD Foundation for Cardiovascular Medicine 72 % Overall 68% Myocardial infarction 57% CABG 47% PTCA 40% CLASS I 4% CLASS II 42% CLASS III 50% CLASS IV 4% 57 %

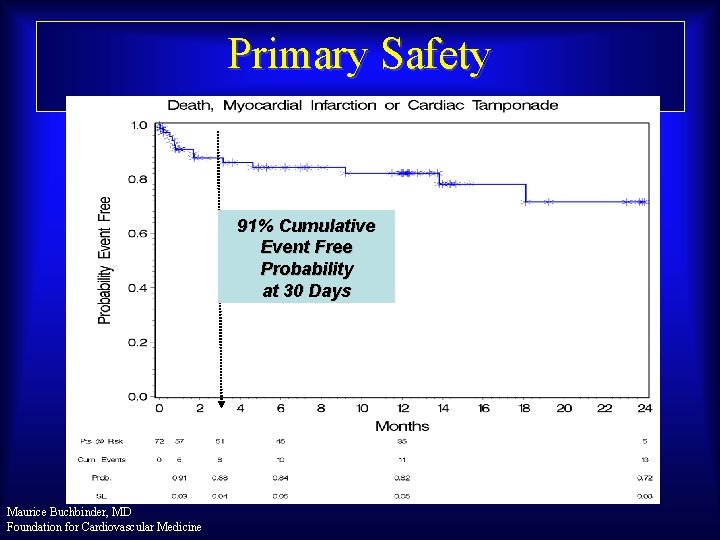
Primary Safety 91% Cumulative Event Free Probability at 30 Days Maurice Buchbinder, MD Foundation for Cardiovascular Medicine

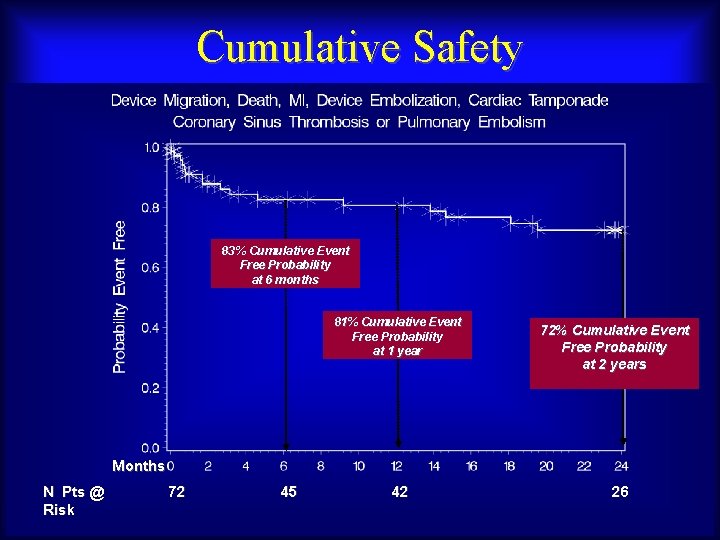
Cumulative Safety 83% Cumulative Event Free Probability at 6 months 81% Cumulative Event Free Probability at 1 year 72% Cumulative Event Free Probability at 2 years Months N Pts @ Risk Maurice Buchbinder, MD 72 Foundation for Cardiovascular Medicine 45 42 26

The procedure does not preclude subsequent interventions • Seven patients had subsequent mitral valve surgery for severe residual MR – (day 70, 110, 164, 222, 232, 426, 430) • Five patients had a CRT device implanted through MONARC device – (day 109, 137, 149, 170, 222) • All procedures successful Maurice Buchbinder, MD Foundation for Cardiovascular Medicine

Device malfunctions seen on routine angiography at 90 -days • No coronary sinus thrombosis • 5 device malfunctions with no clinical complication – 1 Migration • Improper Sizing – 4 Proximal Anchor Device Separation • Device modification implemented Maurice Buchbinder, MD Foundation for Cardiovascular Medicine

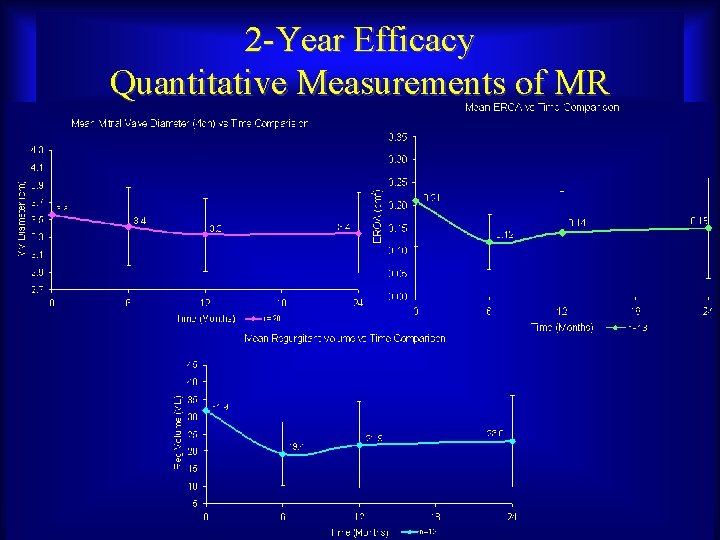
2 -Year Efficacy Quantitative Measurements of MR Maurice Buchbinder, MD Foundation for Cardiovascular Medicine

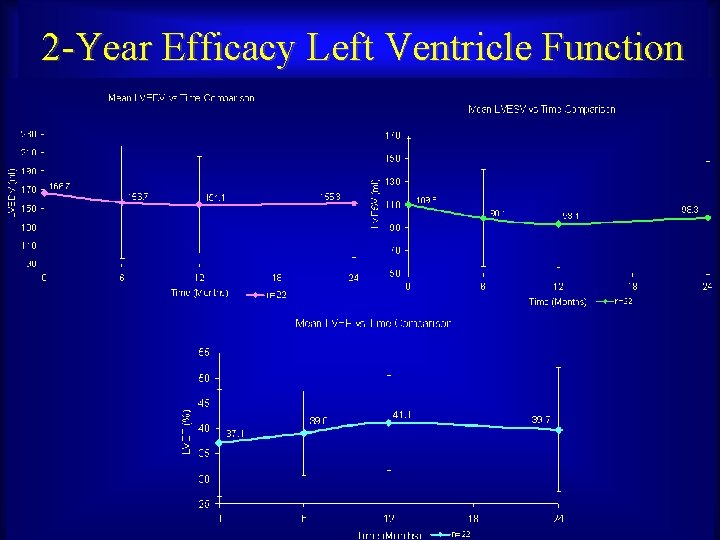
2 -Year Efficacy Left Ventricle Function Maurice Buchbinder, MD Foundation for Cardiovascular Medicine

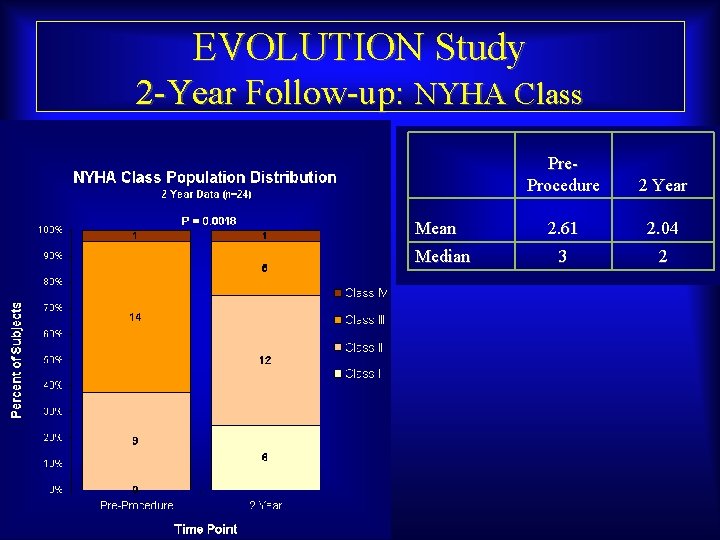
EVOLUTION Study 2 -Year Follow-up: NYHA Class Mean Median Maurice Buchbinder, MD Foundation for Cardiovascular Medicine Pre. Procedure 2 Year 2. 61 2. 04 3 2

Conclusions • EVOLUTION interim data with the MONARC system suggests: • Implantation in the coronary sinus is feasible • At 24 -months, 72% of patients are event-free as defined by the protocol • Encouraging 24 -months results in terms of MR reduction and physiological parameters. Maurice Buchbinder, MD Foundation for Cardiovascular Medicine

Limitations of EVOLUTION • EVOLUTION solely designed as a safety study – No control group – Limited insight in quantifying clinical benefit • EVOLUTION II was designed: – – – With two prospective comparison groups MR and Hemodynamic responses Clinical Outcomes: NYHA, 6 MWT, Qo. L Maurice Buchbinder, MD Foundation for Cardiovascular Medicine

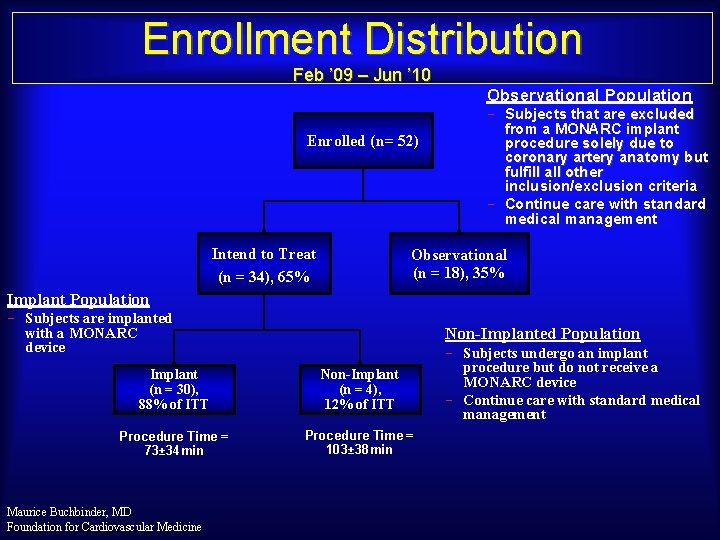
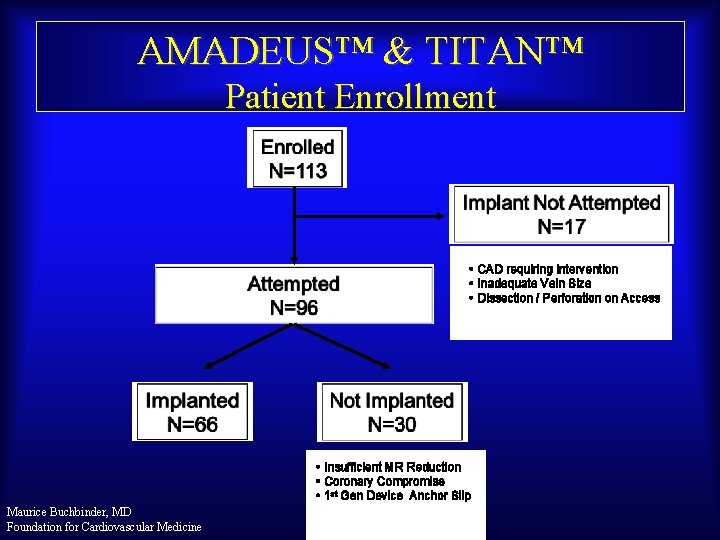
Enrollment Distribution Feb ’ 09 – Jun ’ 10 Enrolled (n= 52) Intend to Treat (n = 34), 65% Observational Population Subjects that are excluded from a MONARC implant procedure solely due to coronary artery anatomy but fulfill all other inclusion/exclusion criteria Continue care with standard medical management Observational (n = 18), 35% Implant Population Subjects are implanted with a MONARC device Non-Implanted Population Implant (n = 30), 88% of ITT Non-Implant (n = 4), 12% of ITT Procedure Time = 73± 34 min Procedure Time = 103± 38 min Maurice Buchbinder, MD Foundation for Cardiovascular Medicine Subjects undergo an implant procedure but do not receive a MONARC device Continue care with standard medical management

Primary Safety Endpoint @ 30 days Group Total N at 30 Total Events days Implant Non-Implant Observational 30 4 18 2 (6. 7%) 1 (25%) 0 Death MI Cardiac Tamponade 1 (3. 3%) 0 0 0 1 (25%) 0 • Implant – Death: Due to Sepsis, day 25 – MI non-Q wave: Misinterpreted CT, day 12 • OM impingement • Non-Implant – Cardiac Tamponade, day 0 Maurice Buchbinder, MD Foundation for Cardiovascular Medicine

Adverse Events up to 6 Mo Total N at 6 mo Implant 24 Non-Implant 3 Observational 13 Group Total Events 2 (8. 3%) 2 (67%) 0 Cardiac CS Pulmonary Tamponade Thrombosis Embolism 1 (4. 2%) 0 0 0 1 (33%) 0 0 0 0 Death MI Additional Events • Non-Implant – Death: Due to MV repair surgery, day 51 Maurice Buchbinder, MD Foundation for Cardiovascular Medicine

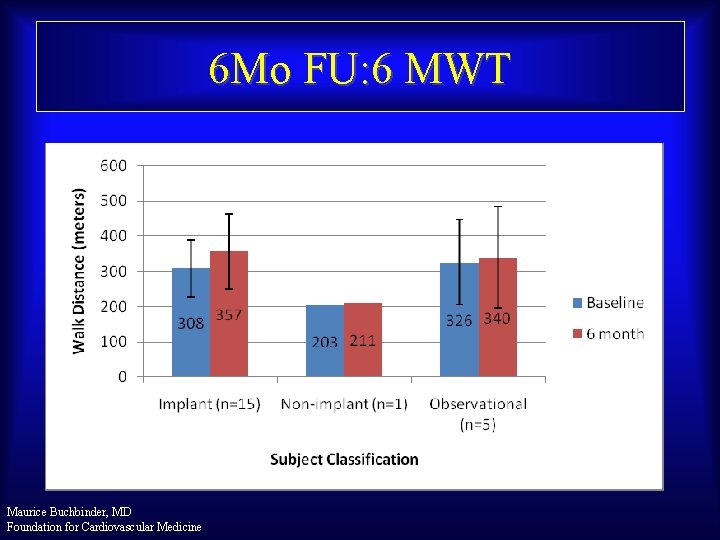
6 Mo FU: 6 MWT Maurice Buchbinder, MD Foundation for Cardiovascular Medicine

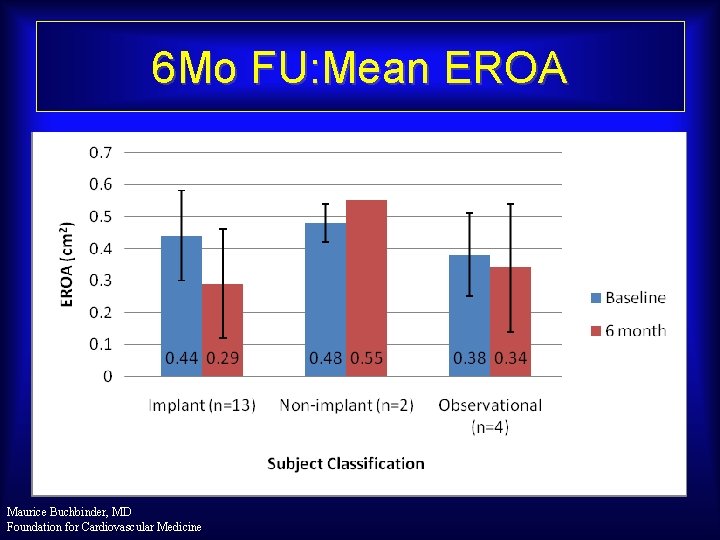
6 Mo FU: Mean EROA Maurice Buchbinder, MD Foundation for Cardiovascular Medicine

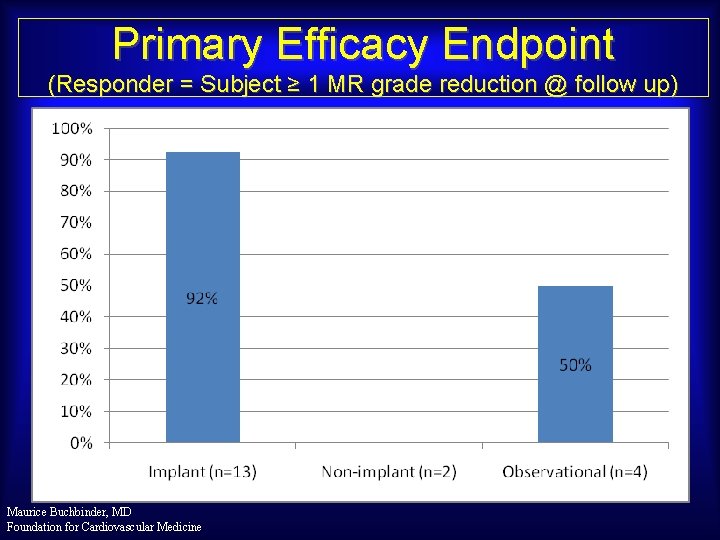
Primary Efficacy Endpoint (Responder = Subject ≥ 1 MR grade reduction @ follow up) Maurice Buchbinder, MD Foundation for Cardiovascular Medicine

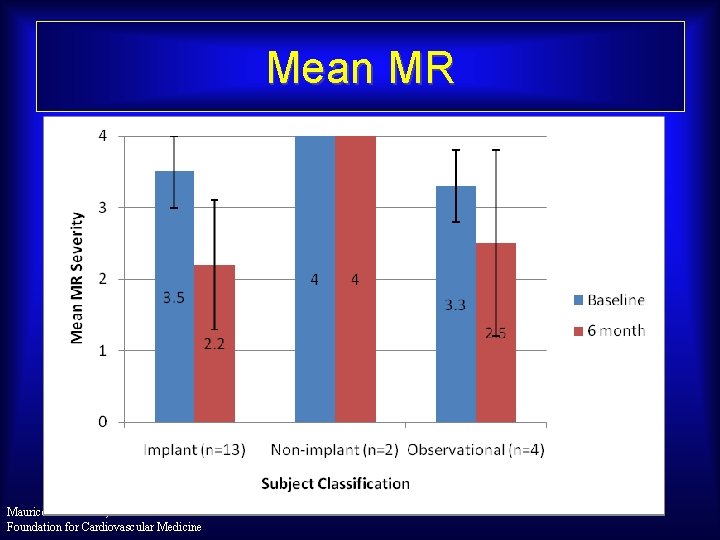
Mean MR Maurice Buchbinder, MD Foundation for Cardiovascular Medicine

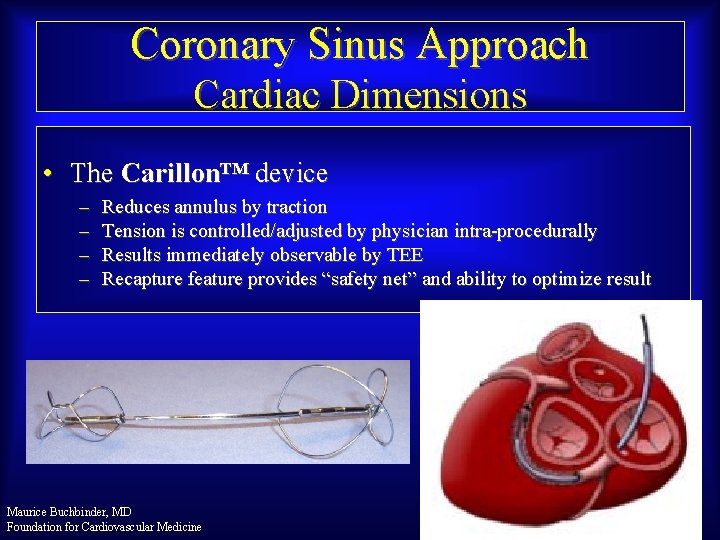
Coronary Sinus Approach Cardiac Dimensions • The Carillon™ device – – Reduces annulus by traction Tension is controlled/adjusted by physician intra-procedurally Results immediately observable by TEE Recapture feature provides “safety net” and ability to optimize result Maurice Buchbinder, MD Foundation for Cardiovascular Medicine

Study Designs • AMADEUS – Prospective, single-arm, 30 patient multi-center trial – Evaluate patients with HF and FMR @ 1 & 6 months (CARILLON® XE implant) • TITAN – Prospective, single-arm, 36 patient multi-center trial – Evaluate patients with HF and FMR @ 1, 6, 12, 18 & 24 months (CARILLON® XE 2 implant) Maurice Buchbinder, MD Foundation for Cardiovascular Medicine

AMADEUS™ & TITAN™ Patient Enrollment • CAD requiring intervention • Inadequate Vein Size • Dissection / Perforation on Access • Insufficient MR Reduction • Coronary Compromise • 1 st Gen Device Anchor Slip Maurice Buchbinder, MD Foundation for Cardiovascular Medicine

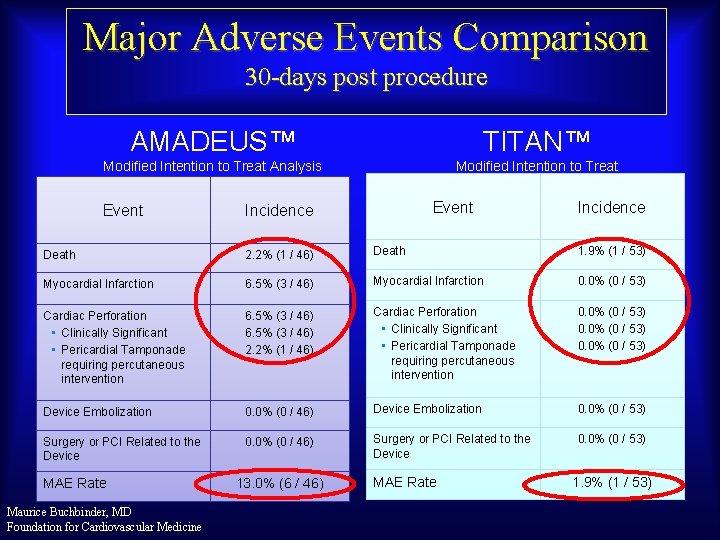
Major Adverse Events Comparison 30 -days post procedure AMADEUS™ TITAN™ Modified Intention to Treat Analysis Event Incidence Death 2. 2% (1 / 46) Death 1. 9% (1 / 53) Myocardial Infarction 6. 5% (3 / 46) Myocardial Infarction 0. 0% (0 / 53) Cardiac Perforation • Clinically Significant • Pericardial Tamponade requiring percutaneous intervention 6. 5% (3 / 46) 2. 2% (1 / 46) Cardiac Perforation • Clinically Significant • Pericardial Tamponade requiring percutaneous intervention 0. 0% (0 / 53) Device Embolization 0. 0% (0 / 46) Device Embolization 0. 0% (0 / 53) Surgery or PCI Related to the Device 0. 0% (0 / 46) Surgery or PCI Related to the Device 0. 0% (0 / 53) MAE Rate Maurice Buchbinder, MD Foundation for Cardiovascular Medicine 13. 0% (6 / 46) MAE Rate 1. 9% (1 / 53)

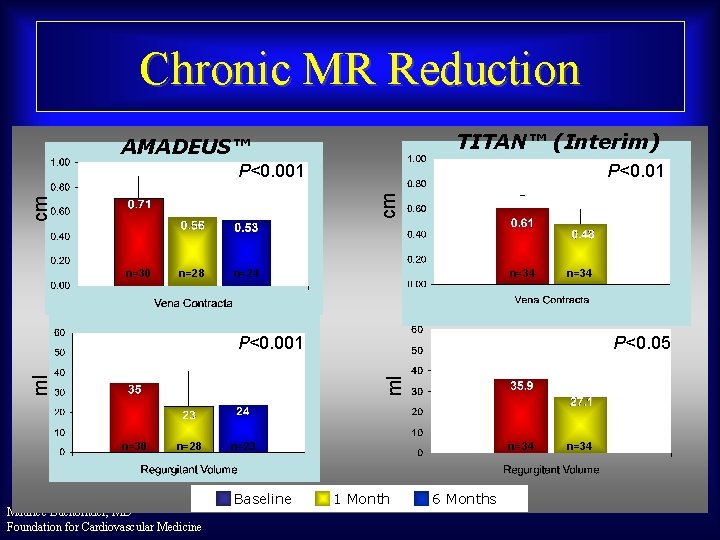
Chronic MR Reduction TITAN™ (Interim) AMADEUS™ P<0. 01 cm cm P<0. 001 n=30 n=28 n=24 n=34 P<0. 05 ml ml P<0. 001 n=30 n=28 Maurice Buchbinder, MD Foundation for Cardiovascular Medicine n=23 Baseline n=34 1 Month 6 Months n=34

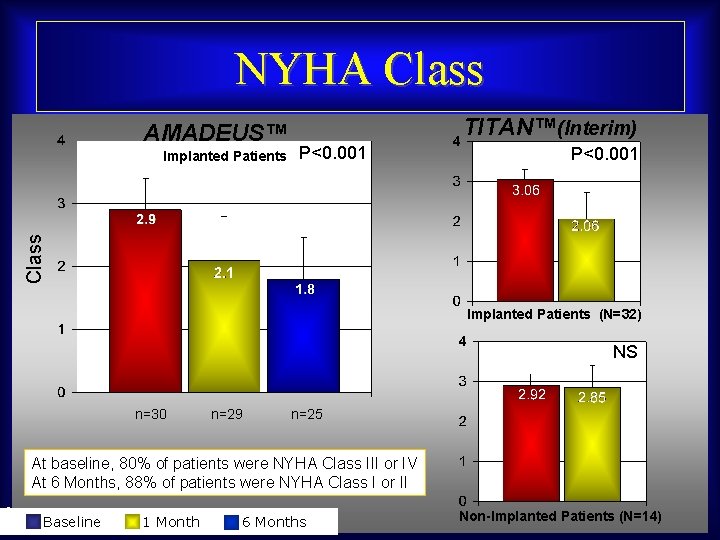
NYHA Class AMADEUS™ P<0. 001 Class Implanted Patients TITAN™(Interim) Implanted Patients (N=32) NS n=30 n=29 n=25 At baseline, 80% of patients were NYHA Class III or IV At 6 Months, 88% of patients were NYHA Class I or II Maurice Buchbinder, MD Baseline 1 Month Foundation for Cardiovascular Medicine 6 Months Non-Implanted Patients (N=14)

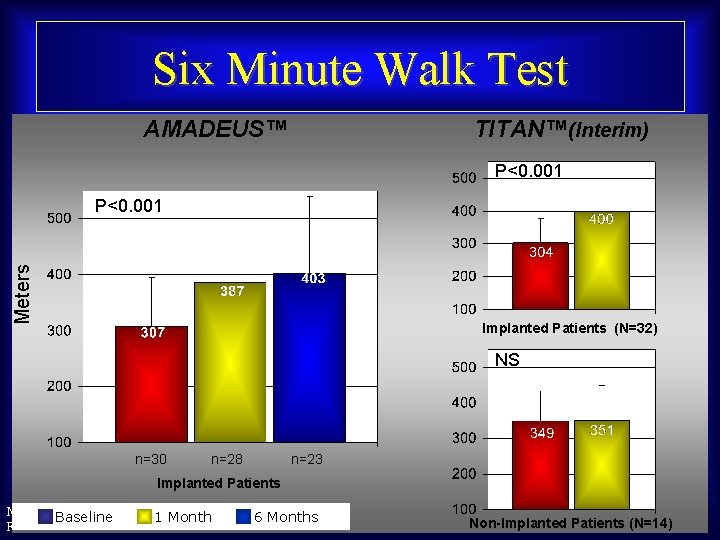
Six Minute Walk Test AMADEUS™ TITAN™(Interim) P<0. 001 Meters P<0. 001 Implanted Patients (N=32) NS n=30 n=28 n=23 Implanted Patients Maurice Buchbinder, MD Baseline 1 Month Foundation for Cardiovascular Medicine 6 Months Non-Implanted Patients (N=14)

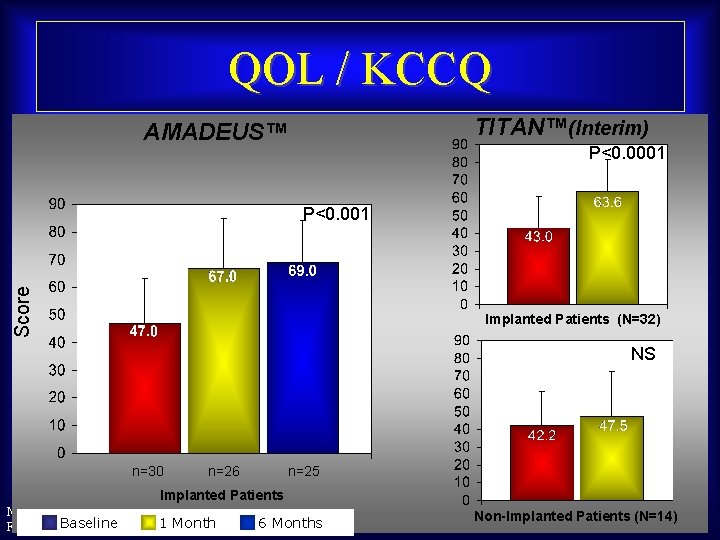
QOL / KCCQ TITAN™(Interim) AMADEUS™ P<0. 0001 Score P<0. 001 Implanted Patients (N=32) NS n=30 n=26 n=25 Implanted Patients Maurice Buchbinder, MD Baseline 1 Month Foundation for Cardiovascular Medicine 6 Months Non-Implanted Patients (N=14)

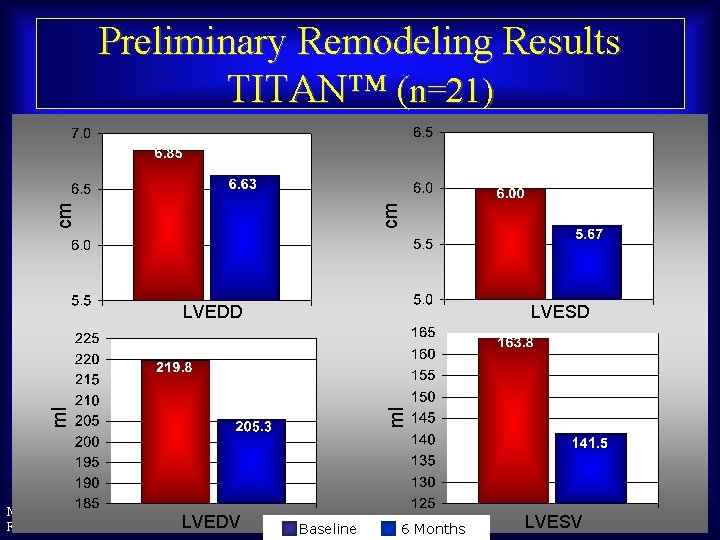
cm cm Preliminary Remodeling Results TITAN™ (n=21) LVESD ml ml LVEDD Maurice Buchbinder, MD LVEDV Foundation for Cardiovascular Medicine Baseline 6 Months LVESV

Conclusions • Low MAE rates @ 30 days, learning curve! • Permanent implantation and significant MR reduction were achieved in the majority of eligible patients. • Significant improvements in functional parameters – 6 MWT, NYHA, QOL • Maybe a mortality and LV remodeling benefit Maurice Buchbinder, MD Foundation for Cardiovascular Medicine

Coronary Sinus Mitral Repair Maurice Buchbinder, MD Foundation for Cardiovascular Medicine

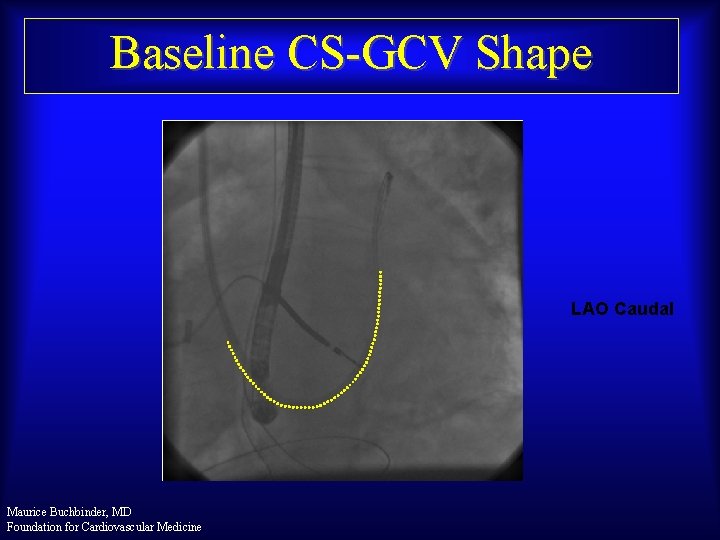
Baseline CS-GCV Shape LAO Caudal Maurice Buchbinder, MD Foundation for Cardiovascular Medicine

PTMA Implant Proximal Adjustment Maurice Buchbinder, MD Foundation for Cardiovascular Medicine

PTMA Implant System Elements • Two implant system lengths • 5 implant rod lengths: 110, 120, 130, 145, 160 mm; stiffness increased 50% over PTOLEMY-1 • 25 mm internal final position adjustment Maurice Buchbinder, MD Foundation for Cardiovascular Medicine

PTOLEMY-1 Patient Study Flow • Start: May 2006 • Close April 2008 Maurice Buchbinder, MD Foundation for Cardiovascular Medicine

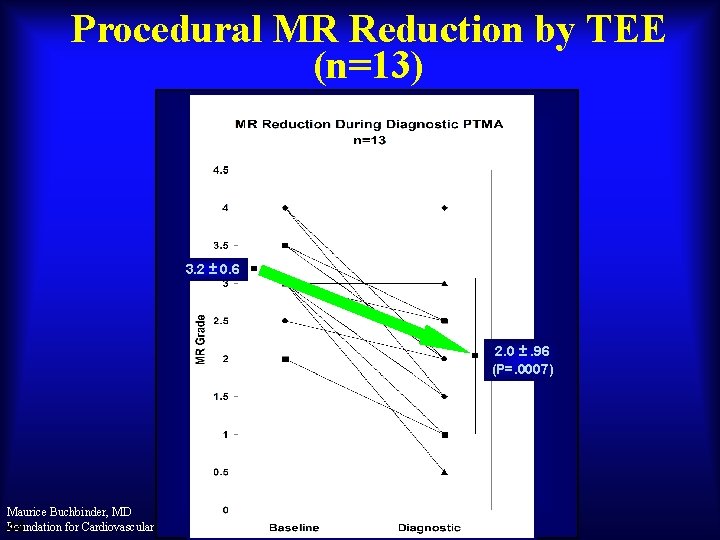
Procedural MR Reduction by TEE (n=13) 3. 2 ± 0. 6 2. 0 ±. 96 (P=. 0007) Maurice Buchbinder, MD Foundation for Cardiovascular Medicine 44

PTOLEMY 2 Trial Europe & Canada • • Indications: II + III NYHA Heart Failure quantitative echo criteria for moderate to severe MR • • • Number of Subjects: 50 enrolled, 30 implants – Europe 30 enrolled, 20 implants - Canada • • • Follow-up periods: Discharge, 1, 3, 6, & 12 mo 2 – 5 years implanted patients • • • Design: Non-randomized, parallel follow-up 8 Centers Europe 3 Centers Canada Enrollment rate = 1 pt /mo /ctr Maurice Buchbinder, MD Foundation for Cardiovascular Medicine • Endpoints: • • • • Safety – 30 days 1° Device / Procedure-Related Major Cardiac Adverse Events Efficacy – 6 months & 12 months 1° MR reduction – echo core lab 2° Quality of life: - Minnesota QOL - 6 minute walk distance Obs: EQ-5 D Obs: Left ventricular size Obs: Rehospitalization rate Obs: Mortality and morbidity

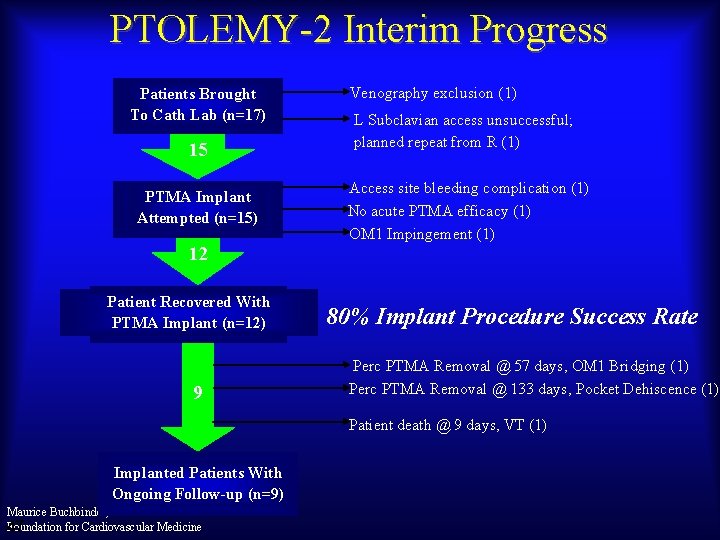
PTOLEMY-2 Interim Progress Patients Brought To Cath Lab (n=17) 15 PTMA Implant Attempted (n=15) Venography exclusion (1) L Subclavian access unsuccessful; planned repeat from R (1) Access site bleeding complication (1) No acute PTMA efficacy (1) OM 1 Impingement (1) 12 Patient Recovered With PTMA Implant (n=12) 9 80% Implant Procedure Success Rate Perc PTMA Removal @ 57 days, OM 1 Bridging (1) Perc PTMA Removal @ 133 days, Pocket Dehiscence (1) Patient death @ 9 days, VT (1) Implanted Patients With Ongoing Follow-up (n=9) Maurice Buchbinder, MD Foundation for Cardiovascular Medicine 47

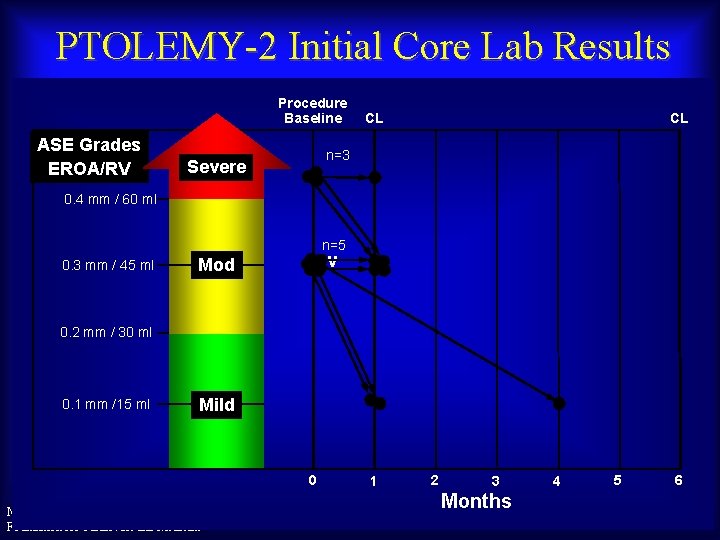
PTOLEMY-2 Initial Core Lab Results Procedure Baseline ASE Grades EROA/RV CL CL n=3 Severe 0. 4 mm / 60 ml n=5 0. 3 mm / 45 ml Mod – v 0. 2 mm / 30 ml 0. 1 mm /15 ml Mild 0 Maurice Buchbinder, MD Foundation for Cardiovascular Medicine 1 2 3 Months 4 5 6

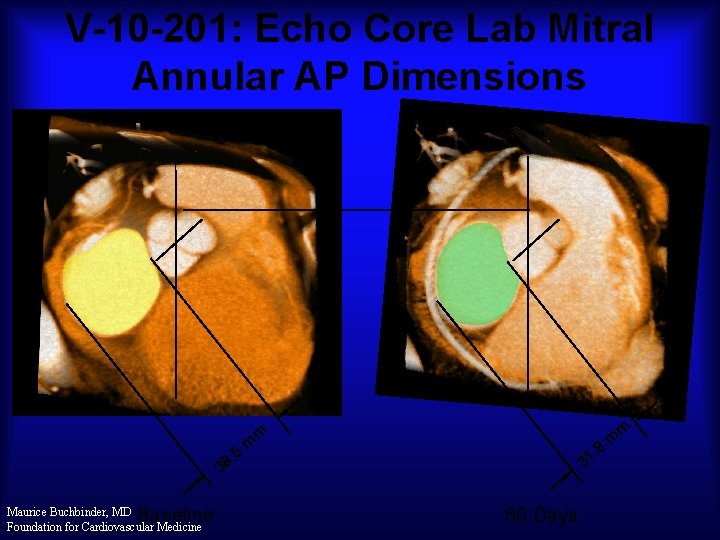
V-10 -201: Echo Core Lab Mitral Annular AP Dimensions m m . 5 8 3 Baseline Maurice Buchbinder, MD Foundation for Cardiovascular Medicine m 8 . 31 60 Days m

Edge-to-edge • e. Valve Coronary sinus annuloplasty • • ? Cardiac Dimensions Carillon • Indirect annuloplasty • • • Direct annuloplasty • Mitralign • Guided Delivery Systems • Quantum. Cor, Cordis DPA • Mi. Cardia, Mitral Solutions Mitral valve replacement • Cardi. AQ • Endovalve Maurice Buchbinder, MD • M-Valve Foundation for Cardiovascular Medicine Today’s Device Landscape: Percutaneous MV Repair