Percent Yield Percent yield describes the efficiency of

Percent Yield

Percent yield describes the efficiency of a chemical reaction • 100 free throws Theoretically… Actually…

Chemical reactions also have theoretical and actual outcomes • Reasons for not producing theoretical amounts: –Reactants or products might stick to container

Reasons for not producing theoretical amounts: –Reactants or products might evaporate

Reasons for not producing theoretical amounts: –Other unintended products might form

Reasons for not producing theoretical amounts: –Purification process might reduce reactant amounts

Because of these problems, scientists need to know how to gage the yield of a chemical reaction

Theoretical yield • Maximum amount of product that can be produced from a given amount of reactant N 2 + 3 H 2 → 2 NH 3

Actual yield • Amount of product produced when a chemical reaction is carried out in an experiment

Popcorn • If there are 100 kernels in a bag… –What is theoretical yield? –What is the actual yield?

Percent yield • Ratio of the actual yield to theoretical yield shown as percent

Percent yield • If there are 105 kernels in a bag of popcorn and 13 were left unpopped, what is the percent yield of that bag? 92/105 = 87. 6 %

Now apply this to chemistry… • Solid silver chromate (Ag₂Cr. O₄) forms when potassium chromate (K₂Cr. O₄) is added to a solution containing 0. 500 g of silver nitrate (Ag. NO₃). Determine theoretical yield of Ag₂Cr. O₄. Write out the equation and balance it!
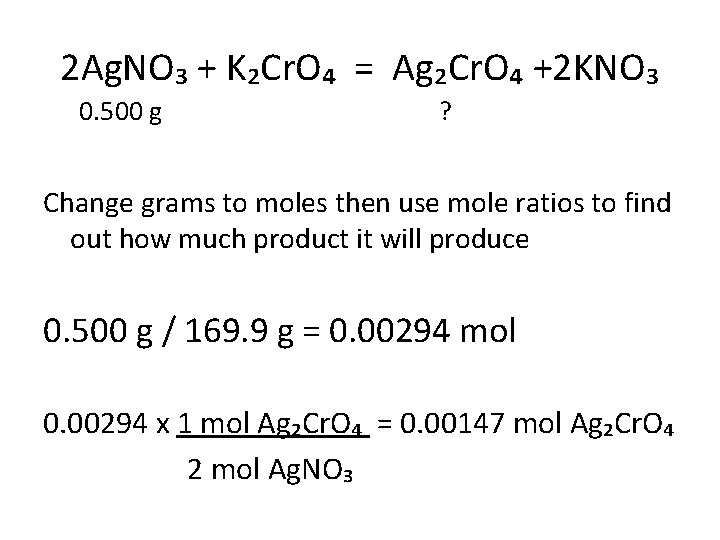
2 Ag. NO₃ + K₂Cr. O₄ = Ag₂Cr. O₄ +2 KNO₃ 0. 500 g ? Change grams to moles then use mole ratios to find out how much product it will produce 0. 500 g / 169. 9 g = 0. 00294 mol 0. 00294 x 1 mol Ag₂Cr. O₄ = 0. 00147 mol Ag₂Cr. O₄ 2 mol Ag. NO₃
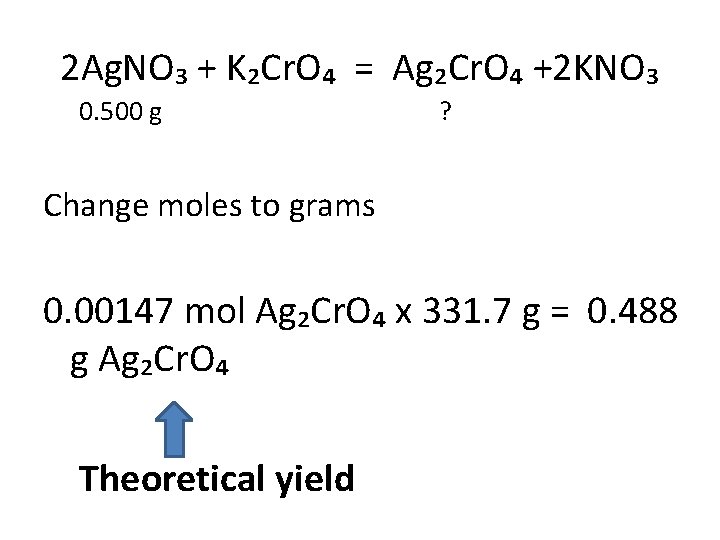
2 Ag. NO₃ + K₂Cr. O₄ = Ag₂Cr. O₄ +2 KNO₃ 0. 500 g ? Change moles to grams 0. 00147 mol Ag₂Cr. O₄ x 331. 7 g = 0. 488 g Ag₂Cr. O₄ Theoretical yield

If the experiment is performed and the actual yield is 0. 455 g Ag₂Cr. O₄, what is the percent yield? 0. 455 g 0. 488 g x 100 = 93. 2% Ag₂Cr. O₄

Popcorn time!!!
- Slides: 17