Pembrolizumab Versus Chemotherapy for Microsatellite InstabilityHighMismatch Repair Deficient

Pembrolizumab Versus Chemotherapy for Microsatellite Instability-High/Mismatch Repair Deficient Metastatic Colorectal Cancer: The Phase 3 KEYNOTE-177 Study Thierry André, 1 Kai-Keen Shiu, 2 Tae Won Kim, 3 Benny Vittrup Jensen, 4 Lars Henrik Jensen, 5 Cornelis Punt, 6 Denis Smith, 7 Rocio Garcia-Carbonero, 8 Manuel Benavides, 9 Peter Gibbs, 10 Christelle de la Fouchardiere, 11 Fernando Rivera, 12 Elena Elez, 13 Johanna Bendell, 14 Dung T. Le, 15 Takayuki Yoshino, 16 Ping Yang, 17 Mohammed Farooqui, 18 Patricia Marinello, 18 and Luis A. Diaz Jr 19 1 Sorbonne Université and Hôpital Saint Antoine, Paris, France; 2 University College Hospital, NHS Foundation Trust, London, United Kingdom; 3 Asan Medical Center, University of Ulsan, Seoul, Republic of Korea; 4 Herlev and Gentofte Hospital, Herlev, Denmark; 5 University Hospital of Southern Denmark, Vejle, Denmark; 6 Amsterdam University Medical Centers, University of Amsterdam, Netherlands; 7 Bordeaux University Hospital, Bordeaux, France; 8 Hospital Universitario 12 de Octubre, Imas 12, CNIO, UCM, Madrid, Spain; 9 Hospital Regional Universitario de Malaga, Spain; 10 Western Health, St Albans, Australia; 11 Léon Bérard Center, Lyon, France; 12 Hospital Universitario Marques de Valdecilla, IDIVAL, Santander, Spain; 13 Vall d’Hebron Institute of Oncology, Barcelona, Spain; 14 Sarah Cannon Research Institute, Nashville, TN, USA; 15 Sidney Kimmel Comprehensive Cancer Center at Johns Hopkins, Baltimore, MD, USA; 16 National Cancer Center Hospital East, Kashiwa, Japan; 17 MSD China, Beijing, China; 18 Merck & Co. , Inc. Kenilworth, NJ, USA; 19 Memorial Sloan Kettering Cancer Center, New York, NY, USA Thierry Andre, MD

Pembrolizumab in MSI-H Metastatic CRC • Microsatellite instability: occurs in cells with high levels of mutation in short, repeated DNA sequences caused by deficiency in mismatch repair system Germline mutation in Lynch syndrome; epigenetic mutation in sporadic case • Patients with MSI-H CRC represent 5% of all patients with CRC • Mismatch-repair status predicted clinical benefit of immune checkpoint blockade with pembrolizumab and other anti-PD-1 therapies 1 -4 • In phase 2 studies, pembrolizumab demonstrated durable antitumor activity with an acceptable safety profile in previously treated MSI-H m. CRC 1 -3 FDA approved pembrolizumab for adults and pediatric patients with previously treated MSI-H metastatic tumors regardless of tumor type or site 3 • We report results from the first phase 3 study of pembrolizumab versus standard of care (chemotherapy ± bevacizumab or cetuximab) as first-line therapy in MSI-H m. CRC 1. Le DT et al. N Engl J Med. 2015; 2. Le DT et al. J Clin Oncol 2020; 3. Pembrolizumab: US Prescribing Information 2020. Merck Sharp & Dohme Corp. , Whitehouse Station, NJ, USA; 4. Overman MJ et al. Lancet Oncol 2017. d. MMR, DNA mismatch repair deficient; m. CRC, metastatic colorectal cancer; MSI-H, microsatellite instability-high. Thierry Andre, MD
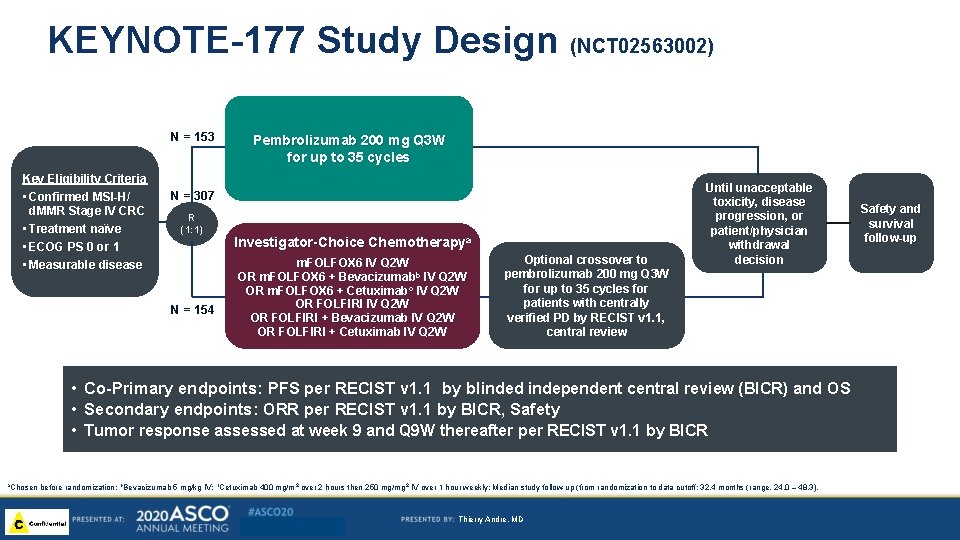
KEYNOTE-177 Study Design (NCT 02563002) N = 153 Key Eligibility Criteria • Confirmed MSI-H/ d. MMR Stage IV CRC • Treatment naïve Pembrolizumab 200 mg Q 3 W for up to 35 cycles N = 307 R (1: 1) • ECOG PS 0 or 1 • Measurable disease N = 154 Investigator-Choice Chemotherapya m. FOLFOX 6 IV Q 2 W OR m. FOLFOX 6 + Bevacizumabb IV Q 2 W OR m. FOLFOX 6 + Cetuximabc IV Q 2 W OR FOLFIRI + Bevacizumab IV Q 2 W OR FOLFIRI + Cetuximab IV Q 2 W Optional crossover to pembrolizumab 200 mg Q 3 W for up to 35 cycles for patients with centrally verified PD by RECIST v 1. 1, central review Until unacceptable toxicity, disease progression, or patient/physician withdrawal decision • Co-Primary endpoints: PFS per RECIST v 1. 1 by blinded independent central review (BICR) and OS • Secondary endpoints: ORR per RECIST v 1. 1 by BICR, Safety • Tumor response assessed at week 9 and Q 9 W thereafter per RECIST v 1. 1 by BICR a. Chosen before randomization; b. Bevacizumab 5 mg/kg IV; c. Cetuximab 400 mg/m 2 over 2 hours then 250 mg/mg 2 IV over 1 hour weekly; Median study follow-up (from randomization to data cutoff: 32. 4 months (range, 24. 0 – 48. 3). Thierry Andre, MD Safety and survival follow-up
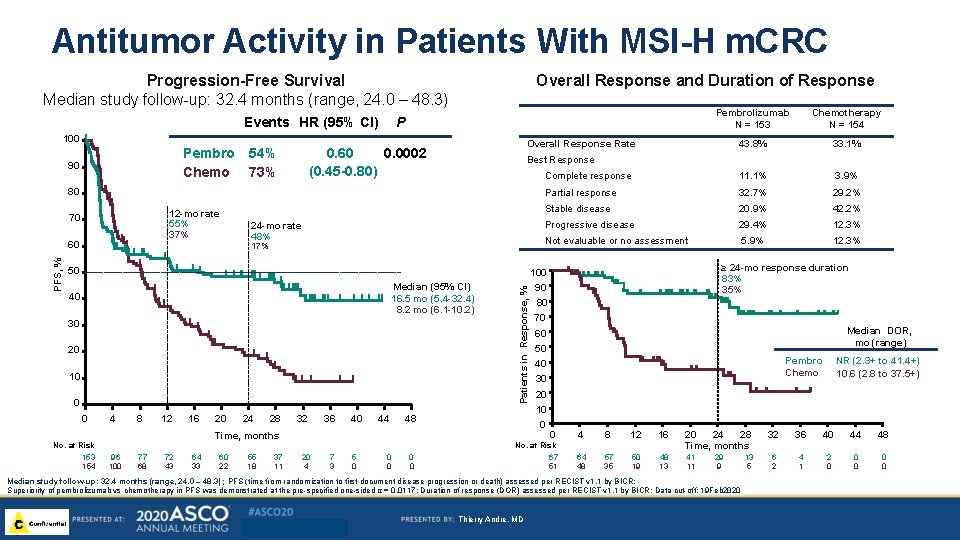
Antitumor Activity in Patients With MSI-H m. CRC Overall Response and Duration of Response Progression-Free Survival Median study follow-up: 32. 4 months (range, 24. 0 – 48. 3) Events HR (95% CI) 100 Pembro Chemo 90 54% 73% 12 -mo rate 55% 37% 43. 8% 33. 1% Complete response 11. 1% 3. 9% Partial response 32. 7% 29. 2% Stable disease 20. 9% 42. 2% Progressive disease 29. 4% 12. 3% Not evaluable or no assessment 5. 9% 12. 3% Best Response 24 -mo rate 48% 17% 50 ≥ 24 -mo response duration 83% 35% 100 Median (95% CI) 16. 5 mo (5. 4 -32. 4) 8. 2 mo (6. 1 -10. 2) 40 30 20 10 0 0 No. at Risk 153 154 4 8 12 16 20 24 28 32 36 40 44 48 Time, months 96 100 77 68 72 43 64 33 60 22 55 18 37 11 Patients in Response, % PFS, % 60 Chemotherapy N = 154 Overall Response Rate 0. 60 0. 0002 (0. 45 -0. 80) 80 70 Pembrolizumab N = 153 P 90 80 70 Median DOR, mo (range) 60 50 Pembro Chemo 40 30 20 10 0 0 4 8 12 16 20 24 28 Time, months 64 48 57 35 50 19 48 13 41 11 No. at Risk 20 4 7 3 5 0 0 0 67 51 29 9 Median study follow-up: 32. 4 months (range, 24. 0 – 48. 3) ; PFS (time from randomization to first document disease progression or death) assessed per RECIST v 1. 1 by BICR; Superiority of pembrolizumab vs chemotherapy in PFS was demonstrated at the pre-specified one-sided α = 0. 0117; Duration of response (DOR) assessed per RECIST v 1. 1 by BICR; Data cut-off: 19 Feb 2020 Thierry Andre, MD NR (2. 3+ to 41. 4+) 10. 6 (2. 8 to 37. 5+) 13 5 32 36 40 44 48 6 2 4 1 2 0 0 0
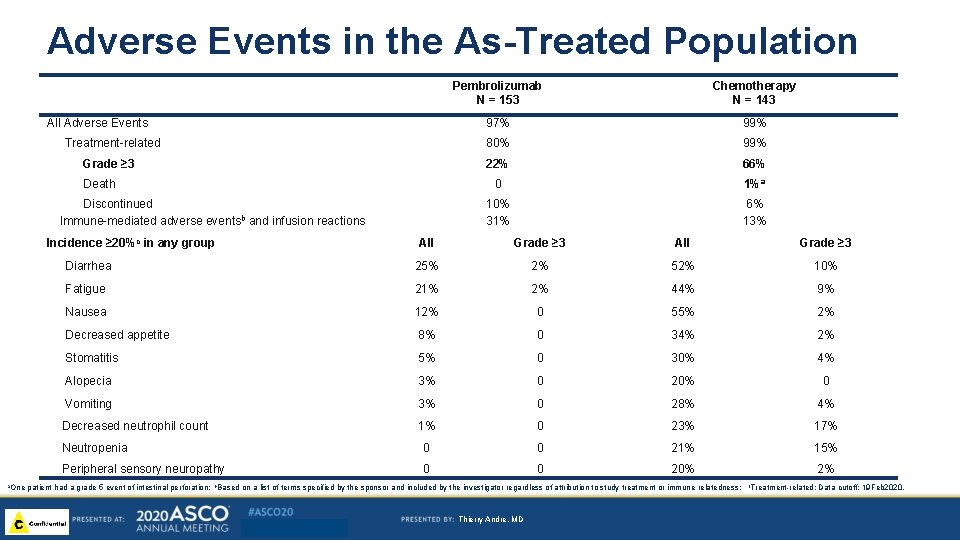
Adverse Events in the As-Treated Population All Adverse Events Treatment-related Grade ≥ 3 Death Discontinued Immune-mediated adverse eventsb and infusion reactions Incidence ≥ 20%c in any group Chemotherapy N = 143 97% 99% 80% 99% 22% 66% 0 1%a 10% 31% 6% 13% All Grade ≥ 3 Diarrhea 25% 2% 52% 10% Fatigue 21% 2% 44% 9% Nausea 12% 0 55% 2% Decreased appetite 8% 0 34% 2% Stomatitis 5% 0 30% 4% Alopecia 3% 0 20% 0 Vomiting 3% 0 28% 4% Decreased neutrophil count 1% 0 23% 17% 0 0 21% 15% 0 0 20% Neutropenia Peripheral sensory neuropathy a. One Pembrolizumab N = 153 patient had a grade 5 event of intestinal perforation; b. Based on a list of terms specified by the sponsor and included by the investigator regardless of attribution to study treatment or immune relatedness; Thierry Andre, MD 2% c. Treatment-related; Data cutoff: 19 Feb 2020.

Summary and Conclusions • Pembrolizumab provided a clinically meaningful and statically significant improvement in PFS versus chemotherapy in patients with MSI-H m. CRC Median PFS 16. 5 vs 8. 2 months (HR 0. 60, 95% CI 0. 45 -0. 80; P = 0. 0002) • Responses were more durable with pembrolizumab versus chemotherapy Response rate: 43. 8% vs 33. 1% (P = 0. 0275) Median duration of response: not reached vs 10. 6 mo with pembrolizumab versus chemotherapy 24 -month progression-free survival rates of 48. 3% versus 18. 6% • Improved safety profile with pembrolizumab versus chemotherapy Lower incidence of grade ≥ 3 treatment-related events (22% vs 66%) No new toxicities observed with pembrolizumab or chemotherapy • Pembrolizumab should be considered a new standard-of-care as first-line therapy in patients with MSI-H m. CRC Thierry Andre, MD

Abstr. 4004. OS and Long-term DFS of 3 versus 6 Months of Adjuvant Oxaliplatin and Fluoropyrimidine-based Therapy for Patients with Stage III Colon Cancer: Final Results from the IDEA Collaboration AF Sobrero, T Andre, JA Meyerhardt, A Grothey, T Iveson, T Yoshino, I Souglakos, JP Meyers, R Labianca, MP Saunders, D Vernerey, T Yamanaka, I Boukovinas, E Oki, V Georgoulias, V Torri, A Harkin, J Taieb, AF Shields, Q Shi for the IDEA collaborators Sobrero et al 7
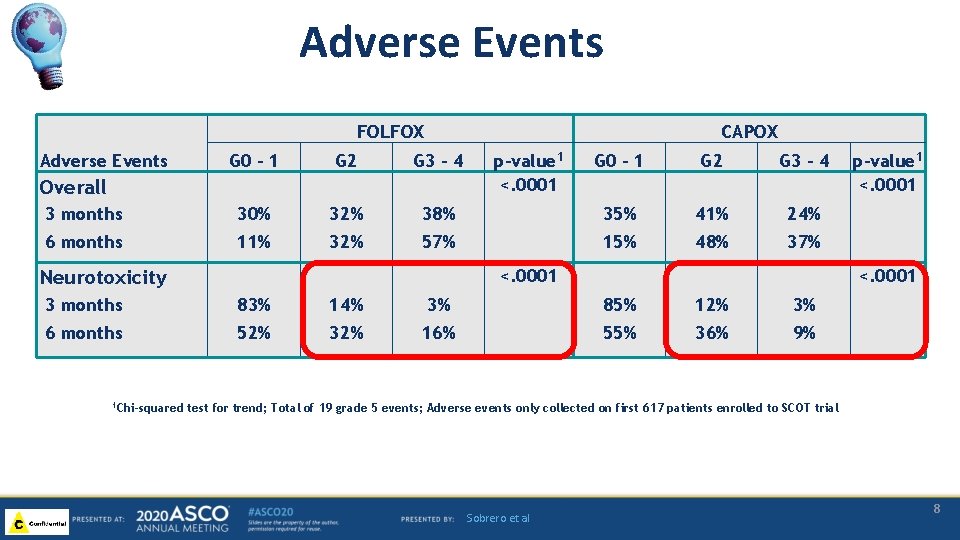
Adverse Events FOLFOX Adverse Events CAPOX G 0 – 1 G 2 G 3 – 4 G 0 – 1 G 2 G 3 - 4 3 months 30% 32% 38% 35% 41% 24% 6 months 11% 32% 57% 15% 48% 37% Overall p-value 1 <. 0001 Neurotoxicity <. 0001 3 months 83% 14% 3% 85% 12% 3% 6 months 52% 32% 16% 55% 36% 9% 1 Chi-squared p-value 1 <. 0001 test for trend; Total of 19 grade 5 events; Adverse events only collected on first 617 patients enrolled to SCOT trial Sobrero et al 8
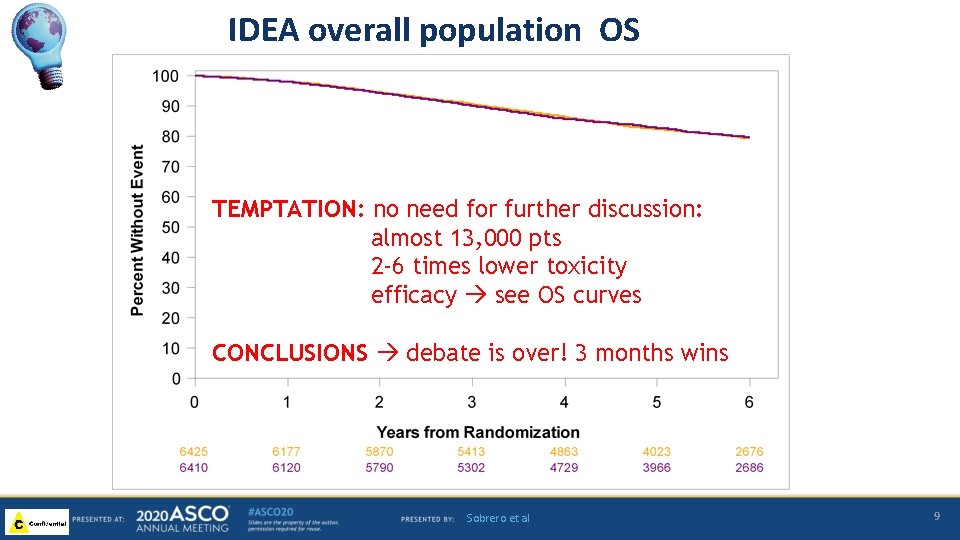
IDEA overall population OS TEMPTATION: no need for further discussion: almost 13, 000 pts 2 -6 times lower toxicity efficacy see OS curves CONCLUSIONS debate is over! 3 months wins Sobrero et al 9
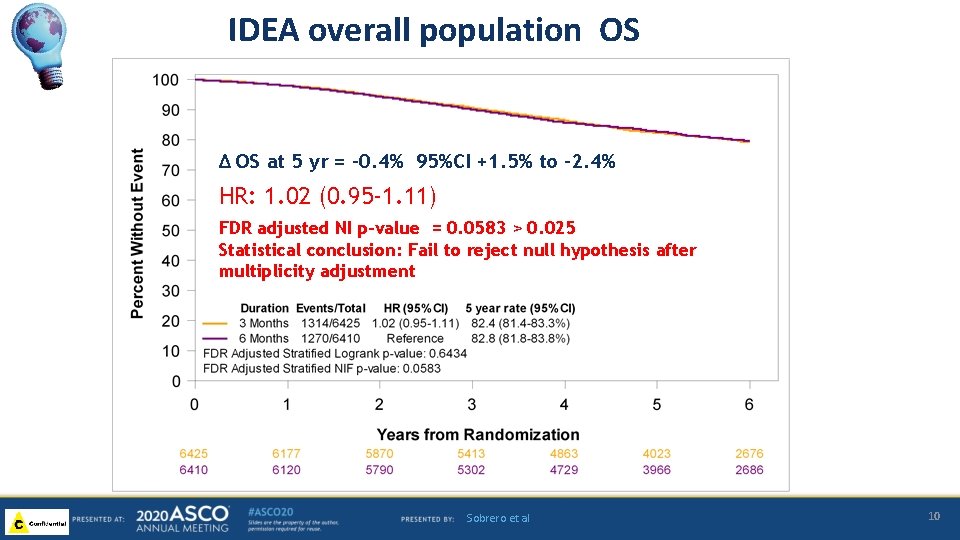
IDEA overall population OS ∆ OS at 5 yr = -0. 4% 95%CI +1. 5% to -2. 4% HR: 1. 02 (0. 95 -1. 11) FDR adjusted NI p-value = 0. 0583 > 0. 025 Statistical conclusion: Fail to reject null hypothesis after multiplicity adjustment Sobrero et al 10
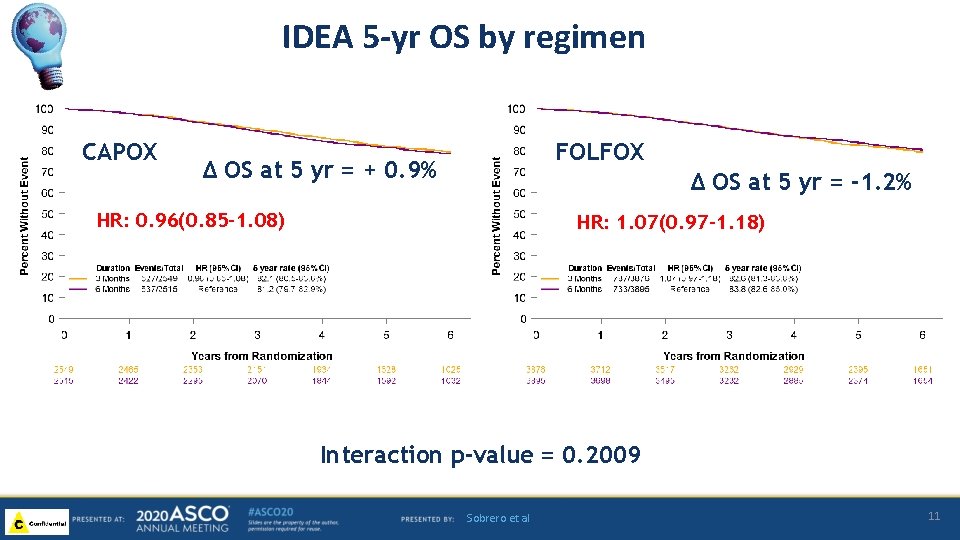
IDEA 5 -yr OS by regimen CAPOX FOLFOX ∆ OS at 5 yr = + 0. 9% ∆ OS at 5 yr = -1. 2% HR: 0. 96(0. 85 -1. 08) HR: 1. 07(0. 97 -1. 18) Interaction p-value = 0. 2009 Sobrero et al 11
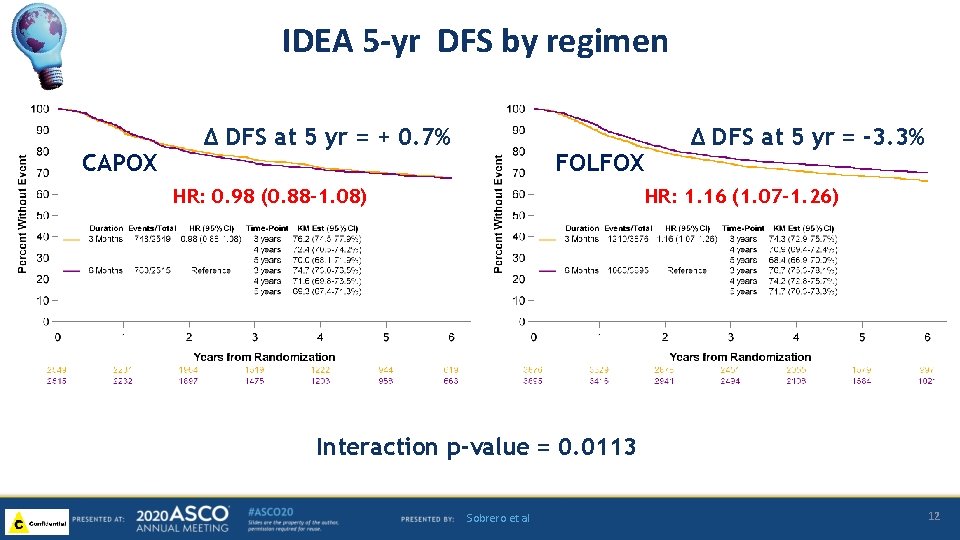
IDEA 5 -yr DFS by regimen CAPOX ∆ DFS at 5 yr = + 0. 7% FOLFOX HR: 0. 98 (0. 88 -1. 08) ∆ DFS at 5 yr = -3. 3% HR: 1. 16 (1. 07 -1. 26) Interaction p-value = 0. 0113 Sobrero et al 12
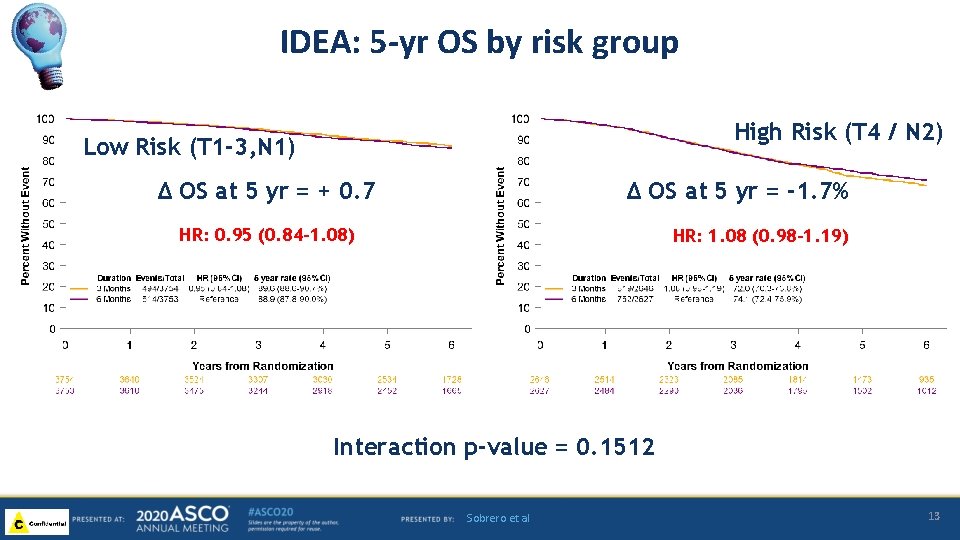
IDEA: 5 -yr OS by risk group High Risk (T 4 / N 2) Low Risk (T 1 -3, N 1) CAPOX FOLFOX ∆ OS at 5 yr = -1. 7% ∆ OS at 5 yr = + 0. 7 HR: 0. 95 (0. 84 -1. 08) HR: 1. 08 (0. 98 -1. 19) Interaction p-value = 0. 1512 Sobrero et al 13
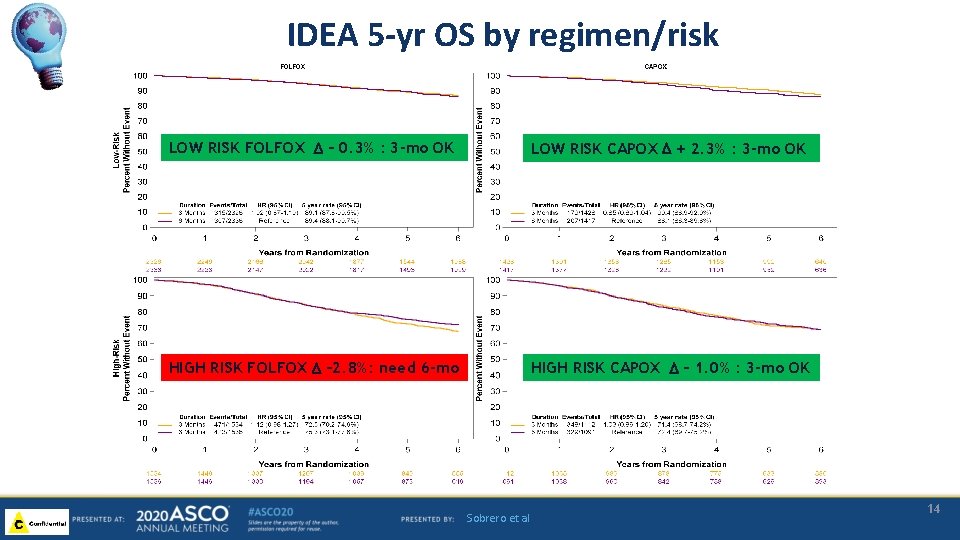
IDEA 5 -yr OS by regimen/risk LOW RISK FOLFOX D – 0. 3% : 3 -mo OK LOW RISK CAPOX D + 2. 3% : 3 -mo OK HIGH RISK FOLFOX D – 2. 8%: need 6 -mo HIGH RISK CAPOX D – 1. 0% : 3 -mo OK Sobrero et al 14

SUMMARY OF IDEA STAGE III STUDY 1. 12, 835 patients randomized 2. 2 to 6 times lower toxicities ; 3 times lower neurotoxicity grade 2+ : 14 vs 46% 3. Primary (3 yr DFS) and two secondary endpoints (5 yr OS and DFS) show minimal, clinically irrelevant differences for most patients with stage 3 colon cancer using 3 months of adjuvant therapy 4. Drug regimen effect was observed in every endpoint, although the trial was not randomized for regimen - No loss of efficacy was observed in low risk cancers with 3 m CAPOX - Minimal loss with 3 m CAPOX in high risk and 3 m FOLFOX in low risk cancers - Relevant loss with 3 m FOLFOX in high risk cancers 5. Strong correlation between DFS at 3 yrs and 5 yr OS data Sobrero et al 15

IMPLICATIONS FOR CLINICAL PRACTICE 1. 60% of patients with stage 3 colon cancer are low risk and should receive 3 month of CAPOX 2. 40% of patients are high risk. For the majority of these, the riskbenefit assessment suggests 3 months of CAPOX as well. For those unwilling to loose even 1 -2% of efficacy, 6 months of therapy is recommended. 3. For High risk cancers, novel prognostic factors including Immunoscore and/or ct. DNA as marker for MRD, may help to define the best adjuvant therapy in the future 1 -2 1 Pages F, Ann Oncol 2020; 2 Taieb J, ESMO 2019 (abstract, Ann Oncol 2019. 30, 867 -867) Sobrero et al 16
- Slides: 16