Pelvic Organ Prolapse Working Group September 7 2018

Pelvic Organ Prolapse Working Group September 7, 2018 Cate Bradley and Kathleen Kobashi

Overview 1 Year 1 goals of the working group 2 What has been accomplished thus far 3 Short-term and long-term goals for the core minimum dataset 4 REDCap demonstration of core minimum dataset 5 Next steps for the working group
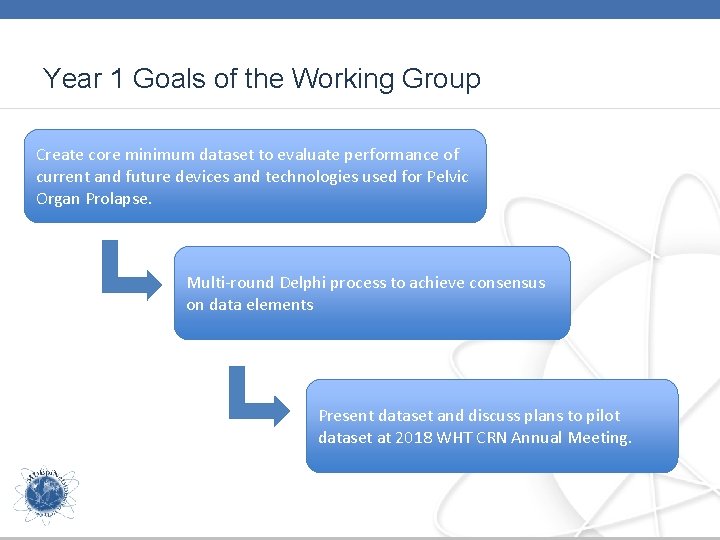
Year 1 Goals of the Working Group Create core minimum dataset to evaluate performance of current and future devices and technologies used for Pelvic Organ Prolapse. Multi-round Delphi process to achieve consensus on data elements Present dataset and discuss plans to pilot dataset at 2018 WHT CRN Annual Meeting.

What has been Accomplished Thus Far October-December 2017 • Data elements were compiled by FDA using (1) PFDR, (2) existing literature and (3) WHT-CRN elements suggested at September 2017 kick-off meeting January-March 2018 • ● POP working group members selected and convened • ● Conference call to kick-start the POP working group, review the Delphi process • ● Round 1 of the Delphi process initiated April-June 2018 July-September 2018 • ● Round 1 Delphi Survey completed • ● Results analyzed by Cornell team and discussed in conference calls with co-chairs • ● Working group solicited applications for patient partners

Working Group Members Clinical Chairs: • Catherine Bradley – American Urogynecologic Society (AUGS) • Kathleen Kobashi – Society of Urodynamics, Female Pelvic Medicine & Urogenital Reconstruction (SUFU) FDA Chair: Tony Du – Center for Devices and Radiological Health (CDRH) Working Group Members: • • Lenore Ackerman (SUFU) Matt Barber (AUGS) Diane Brinza (Coloplast) Maude Carmel (SUFU) Bilal Chughtai (Weill Cornell Medicine) Elisabeth Erekson (Dartmouth-Hitchcock) Michael Flynn (UMass Memorial) Donna Gardner (Boston Scientific) • • Alex Gomelsky (SUFU) Douglass Hale (IU Health) Gary Lemack (SUFU) Pamela Moali (AUGS) Victor Nitti (SUFU) Amy O'Boyle (AUGS) Priya Padmanabhan (SUFU) Samantha Pulliam (UNC) • • Charles Rardin (AUGS) Stuart Reynolds (AUGS) Holly Richter (AUGS) Eric Rovner (SUFU) Yahir Santiago-Lastra (SUFU) Colleen Skau (AUGS) Sherry Xiao (Patients Like Me) Other Members: • FDA – CDRH – Danica Marinac-Dabic, Nilsa Loyo-Berrios, Mary Jung – Office of Device Evaluation (ODE) – Sharon Andrews, Jacqueline Cunkelman • Cornell – Art Sedrakyan, Courtney Baird, Molly Nowels, Nadia Kim 5

Patient Partner’s Role • Working group meets monthly via teleconference. • Role of the patient partner is to: • Serve for one-year in a volunteer capacity • Provide input to working group on data elements • Help identify priority questions for CRN of interest to patients making decisions on various treatment options for pelvic organ prolapse • Participate in monthly meetings/calls (one hour each) 6

POP Patient Partner: Sherrie Palm • Sherrie Palm is: • Founder/CEO of the Association for Pelvic Organ Prolapse Support (APOPS) • POP Key Opinion Leader • Global women's pelvic health advocate • Multi-published author of award-winning book Pelvic Organ Prolapse: The Silent Epidemic • National and international speaker on POP QOL • Sherrie’s points of focus are: • Generating global POP awareness • Developing guidance and support structures for women navigating POP • Bridge-building within POP healthcare, research, academic, industry, and policy sectors toward evolution of POP directives 7
Survey Monkey Sample 8

Delphi Survey Results • Round 1 to 2 • • • Start to End of Round 2 • • • Response rate: 20/21 working group members Results: 99 (88%) data elements had ≥ 50% consensus Response rate: 15/21 working group members Results: 153 (94%) data elements had ≥ 50% consensus Below are the number of data elements by round of the Delphi process. Start of Round 1: 1. Patient factors a. pre-operative (10) b. medical history (11) c. surgical history (14) d. peri-operative (29) e. post-operative (25) 2. Device factors (9) 3. Surgeon factors (14) TOTAL: 112 data elements Start of Round 2: 1. Patient factors a. pre-operative i. medical history (17) ii. surgical history (12) iii. examination (5) b. peri-operative i. procedure (38) ii. discharge (3) c. post-operative i. 0 -30 days of follow-up (26) ii. 31 -90 days of follow-up (23) iii. >90 days of follow-up (23) 2. Device factors (4) 3. Surgeon factors (12) End of Round 2: 1. Patient factors a. pre-operative i. medical history (15) ii. surgical history (12) iii. examination (3) b. peri-operative i. procedure (33) ii. discharge (3) c. post-operative i. 0 -30 days of follow-up (25) ii. 31 -90 days of follow-up (22) iii. >90 days of follow-up (23) 2. Device factors (4) 3. Surgeon factors (11) TOTAL: 163 data elements TOTAL: 151 data elements 9

ST and LT Goals for the Core Minimum Dataset Short Term Goals • Establish core minimum dataset to assess procedure quality and performance of POP devices through a Delphi Method • Incorporate POP core minimum dataset as a new module into AQUIRE (AUGS Urogynecology Quality Registry). • AQUIRE at present includes a module on SUI but is capable of including other modules for different urogynecological procedures, such as POP. Long Term Goal • Support establishment of a coordinated network that links existing registries and data networks to address clinical questions related to devices and technologies for the treatment of pelvic organ prolapse. • Provide platform for potential future modular expansion: premarket investigation of POP devices, POP quality measure testing, additional research questions

REDCap Demonstration of Dataset

REDCap Disclaimer • • REDCap is a HIPAA-compliant data collection platform that is used to collect data for single and multi-site research studies. We may or may not use REDCap when we finally pilot the datasets in practice – right now, we are just using REDCap to demonstrate what it would look like to actually fill out a form to collect one registry’s data elements in practice. • • As you know, we will be working with the NIH CDE Repository, which supports form building that can be used in REDCap and other settings. One of the goals for a minimum dataset is that it be as easy as possible for the physician to complete. What you’ll find is that data collection tools, like REDCap, can lessen physician burden through tools like branching logic and automatic data capture. This is not a final form by any means. For example, not all of the questions would need to be asked at every encounter, and we still need to work with YOU and the informatics team to finalize the data element definitions, harmonizable data elements, collection methodologies, etc. REDCap Survey Link
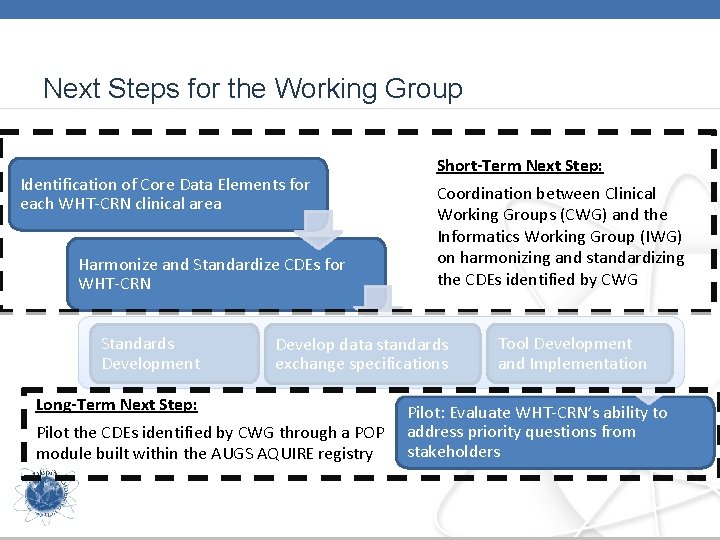
Next Steps for the Working Group Identification of Core Data Elements for each WHT-CRN clinical area Harmonize and Standardize CDEs for WHT-CRN Standards Development Short-Term Next Step: Coordination between Clinical Working Groups (CWG) and the Informatics Working Group (IWG) on harmonizing and standardizing the CDEs identified by CWG Develop data standards exchange specifications Long-Term Next Step: Pilot the CDEs identified by CWG through a POP module built within the AUGS AQUIRE registry Tool Development and Implementation Pilot: Evaluate WHT-CRN’s ability to address priority questions from stakeholders
Thank you.
- Slides: 14