Peginterferon alfa2 a 40 KD PEGASYS in combination

Peginterferon alfa-2 a (40 KD) (PEGASYS®) in combination with ribavirin (RBV): efficacy and safety results from a phase III, randomized, double-blind, multicentre study examining effect of duration of treatment and RBV dose Professor Stephanos J. Hadziyannis Department of Medicine and Hepatology Henry Dunant Hospital Athens, Greece Hadziyannis SJ et al. EASL. 2002.

Background • Previous clinical studies with pegylated IFNs only investigated 48 weeks • No prospective studies of the efficacy and safety of ribavirin doses have been conducted • Therefore no evidence-based recommendation could be made Hadziyannis SJ et al. EASL. 2002.

Study Aims • To compare the efficacy and safety of the combination of PEGASYS® and ribavirin given for 24 weeks vs. 48 weeks • To compare the efficacy and safety of two different daily doses of ribavirin (low dose 800 mg vs. ‘standard’ dose of 1000/1200 mg) taken with PEGASYS® Hadziyannis SJ et al. EASL. 2002.

Study Design (1) Total 1284 patients randomized and treated A: PEGASYS® 180 µg sc qw + ribavirin 800 mg qd, 24 weeks B: PEGASYS® 180 µg sc qw + ribavirin 1000/1200 mg qd, 24 weeks C: PEGASYS® 180 µg sc qw + ribavirin 800 mg qd, 48 weeks D: PEGASYS® 180 µg sc qw + ribavirin 1000/1200 mg qd, 48 weeks Treatment-free follow-up of 24 weeks for all patients Hadziyannis SJ et al. EASL. 2002.

Study Design (2) • Randomization stratified by HCV genotype (1 vs. non-1) and viral titre (low vs. high, defined as or >2 million copies/m. L, respectively) and by geographic region • Pre-planned distribution of genotypes – Genotype non 1 and 1 LVT 1: 1: 1: 1 – Genotype 1 HVT 1: 1: 4: 4 • • Treatment duration blinded until week 24 Ribavirin dose blinded throughout study Hadziyannis SJ et al. EASL. 2002.

Primary Endpoint Undetectable serum HCV RNA at the end of a 24 -week treatment-free follow -up period (COBAS AMPLICOR® HCV Test v 2. 0, sensitivity 50 IU/m. L) Hadziyannis SJ et al. EASL. 2002.

Main Inclusion Criteria • Quantifiable HCV RNA in serum (AMPLICOR HCV MONITOR® Test, v 2. 0) • Elevated serum ALT levels • Liver biopsy consistent with chronic HCV infection • No previous interferon or ribavirin treatment Hadziyannis SJ et al. EASL. 2002.

Main Exclusion Criteria • • • Decompensated liver disease • Significant co-morbid medical conditions Coinfection with HIV or HBV Anaemia or expected inability to tolerate anaemia Hadziyannis SJ et al. EASL. 2002.
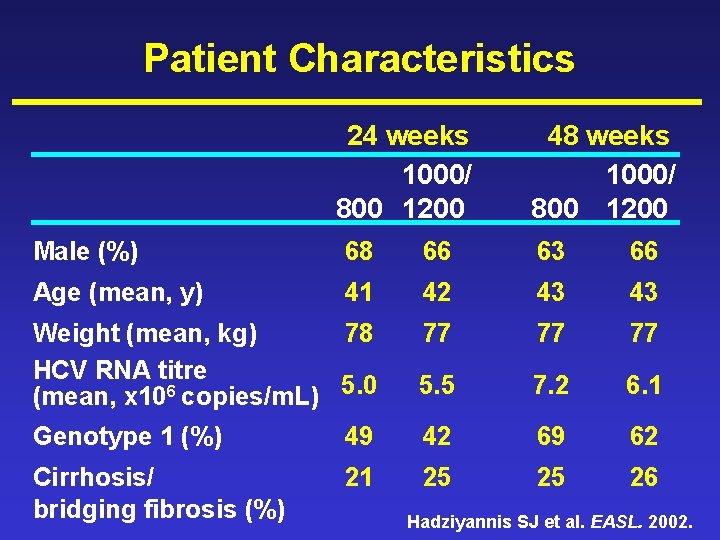
Patient Characteristics 24 weeks 1000/ 800 1200 48 weeks 1000/ 800 1200 Male (%) 68 66 63 66 Age (mean, y) 41 42 43 43 Weight (mean, kg) 78 HCV RNA titre (mean, x 106 copies/m. L) 5. 0 77 77 77 5. 5 7. 2 6. 1 Genotype 1 (%) 49 42 69 62 Cirrhosis/ bridging fibrosis (%) 21 25 25 26 Hadziyannis SJ et al. EASL. 2002.
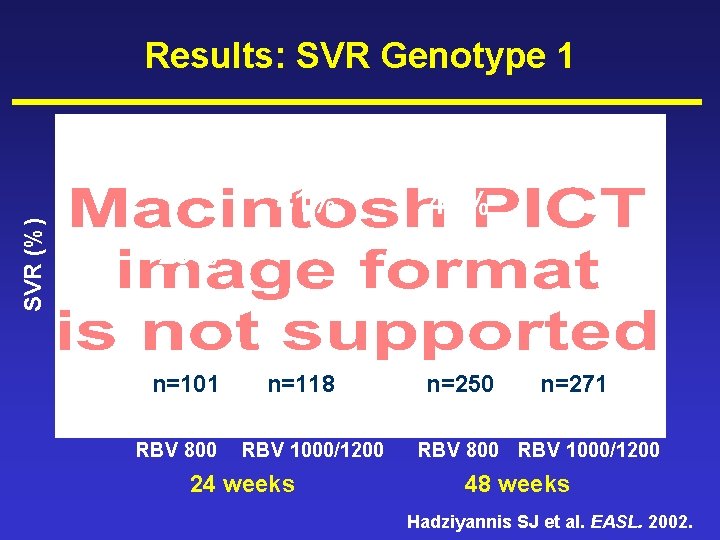
Results: SVR Genotype 1 SVR (%) 51% 40% n=118 n=250 29% n=101 PEGASYS® RBV 800 RBV 1000/1200 24 weeks n=271 PEGASYS® RBV 800 RBV 1000/1200 48 weeks Hadziyannis SJ et al. EASL. 2002.
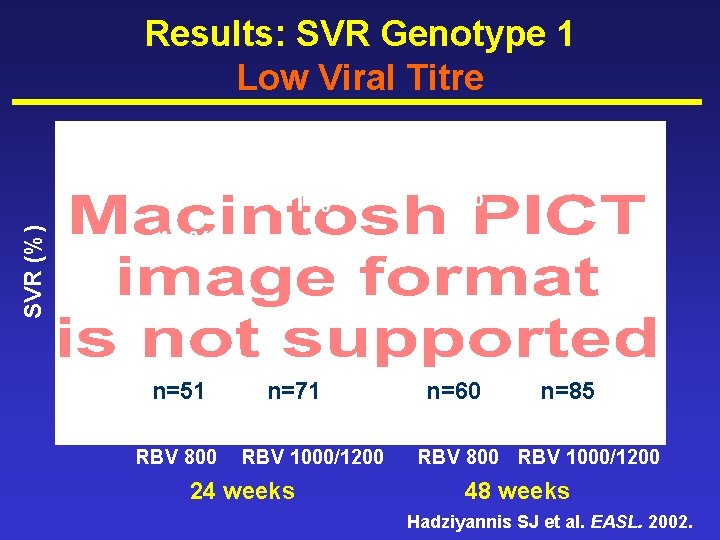
SVR (%) Results: SVR Genotype 1 Low Viral Titre 51% 53% n=71 n=60 61% 41% n=51 PEGASYS® RBV 800 RBV 1000/1200 24 weeks n=85 PEGASYS® RBV 800 RBV 1000/1200 48 weeks Hadziyannis SJ et al. EASL. 2002.

Results: SVR Genotype 1 High Viral Titre 46% SVR (%) 35% 26% 16% n=50 n=47 PEGASYS® RBV 800 RBV 1000/1200 24 weeks n=190 n=186 PEGASYS® RBV 800 RBV 1000/1200 48 weeks Hadziyannis SJ et al. EASL. 2002.
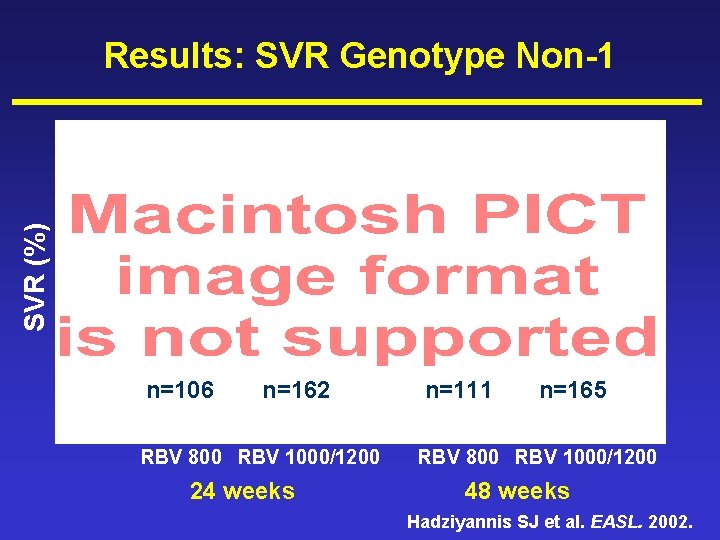
Results: SVR Genotype Non-1 78% n=106 n=162 73% 77% n=111 n=165 SVR (%) 78% PEGASYS® RBV 800 RBV 1000/1200 24 weeks PEGASYS® RBV 800 RBV 1000/1200 48 weeks Hadziyannis SJ et al. EASL. 2002.

Sustained Virologic Response: Effect of Cirrhosis 61% 65% SVR (%) 50% n=436 n=321 n=115 All patients Non-cirrhotics Cirrhotics Treatment: PEGASYS® + RBV 1000/1200 mg for 48 weeks Hadziyannis SJ et al. EASL. 2002.
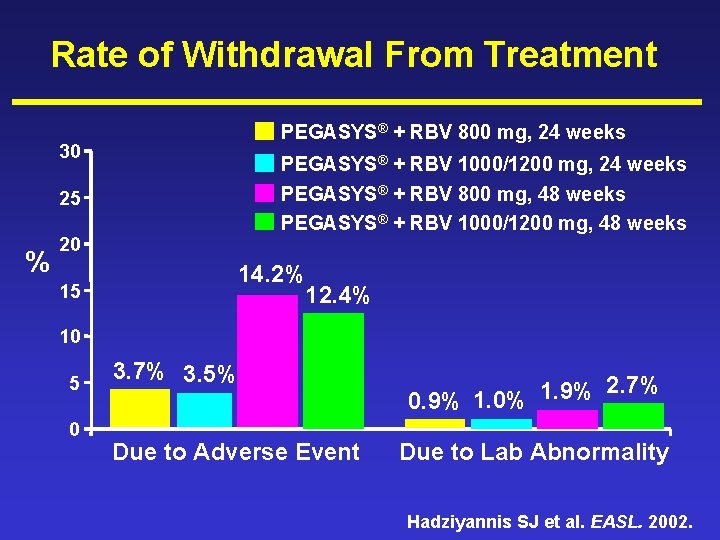
Rate of Withdrawal From Treatment PEGASYS® + RBV 800 mg, 24 weeks 30 PEGASYS® + RBV 1000/1200 mg, 24 weeks PEGASYS® + RBV 800 mg, 48 weeks PEGASYS® + RBV 1000/1200 mg, 48 weeks 25 % 20 15 14. 2% 12. 4% 10 5 0 3. 7% 3. 5% Due to Adverse Event 2. 7% 1. 9% 0. 9% 1. 0% Due to Lab Abnormality Hadziyannis SJ et al. EASL. 2002.

Discontinuations (%) Ribavirin Discontinuations for Adverse Events / Lab Abnormalities 18% 7% 19% 6% PEGASYS® RBV 800 RBV 1000/1200 24 weeks PEGASYS® RBV 800 RBV 1000/1200 48 weeks Hadziyannis SJ et al. EASL. 2002.

Serious Adverse Events 9% 10% 7% % 3% 1% 3% PEGASYS® RBV 800 RBV 1000/1200 24 weeks Treatment-related SAEs 4% 3% PEGASYS® RBV 800 RBV 1000/1200 48 weeks Hadziyannis SJ et al. EASL. 2002.

Summary (1) • Overall SVR of 61% in patients treated for 48 weeks with PEGASYS® and RBV 1000/1200 mg • Overall safety profile similar to previous studies Hadziyannis SJ et al. EASL. 2002.

Summary (2) • Genotype 1 – 51% SVR achieved with 48 weeks treatment, 1000/1200 mg RBV – Shorter duration and/or lower RBV dose leads to reduction in efficacy Hadziyannis SJ et al. EASL. 2002.

Summary (3) • Genotype non-1 – SVR 78% 24 weeks with 800 mg RBV – Increasing duration and/or dose of RBV gave no increase in efficacy • Shorter treatment associated with fewer SAEs and withdrawals for safety • Lower dose RBV associated with fewer – SAEs (24 weeks) – RBV dose modifications – Large decreases in haemoglobin Hadziyannis SJ et al. EASL. 2002.

Study Countries Norway UK Sweden Canada Ireland Denmark Belgium Finland USA Netherlands France Mexico Germany Taiwan Brazil Portugal Spain Australia Italy New Zealand Greece 21 Countries Hadziyannis SJ et al. EASL. 2002.
- Slides: 21