Pediatric Patellar Instability Developmental Anatomy Sean Keyes DO

Pediatric Patellar Instability: Developmental Anatomy Sean Keyes DO Florida Hospital for Children Orlando, FL Shital Parikh MD Cincinnati Children’s Medical Center Cincinnati, OH

Overview • Definition • Embryology • Patellar Development • Trochlear Development • Medial Patellofemoral Ligament (MPFL)
Introduction • Patella instability is defined by the dislocation or subluxation of the patella from the trochlear groove • This can be debilitating to growing children leading to falls, osteochondral fractures, loose bodies and arthritis. • The stability to the patellofemoral joint relies on a complex interplay between the morphology of the distal femur and trochlear groove, shape of the patella, ligamentous status, lower extremity alignment, rotational profile and patients’ neuromuscular development. • The purpose of this presentation is to provide an overview for the developmental anatomy for the lower extremity and patellofemoral joint.
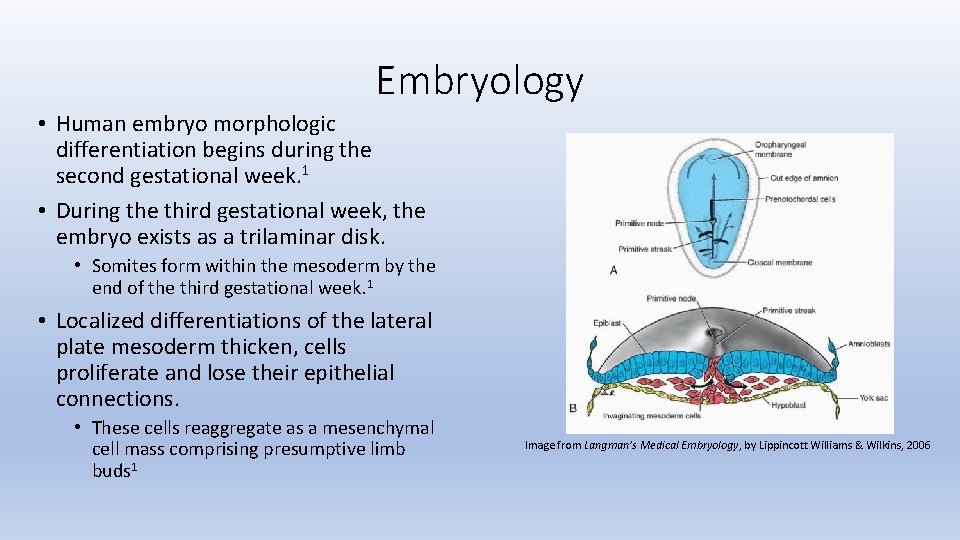
Embryology • Human embryo morphologic differentiation begins during the second gestational week. 1 • During the third gestational week, the embryo exists as a trilaminar disk. • Somites form within the mesoderm by the end of the third gestational week. 1 • Localized differentiations of the lateral plate mesoderm thicken, cells proliferate and lose their epithelial connections. • These cells reaggregate as a mesenchymal cell mass comprising presumptive limb buds 1 Image from Langman’s Medical Embryology, by Lippincott Williams & Wilkins, 2006
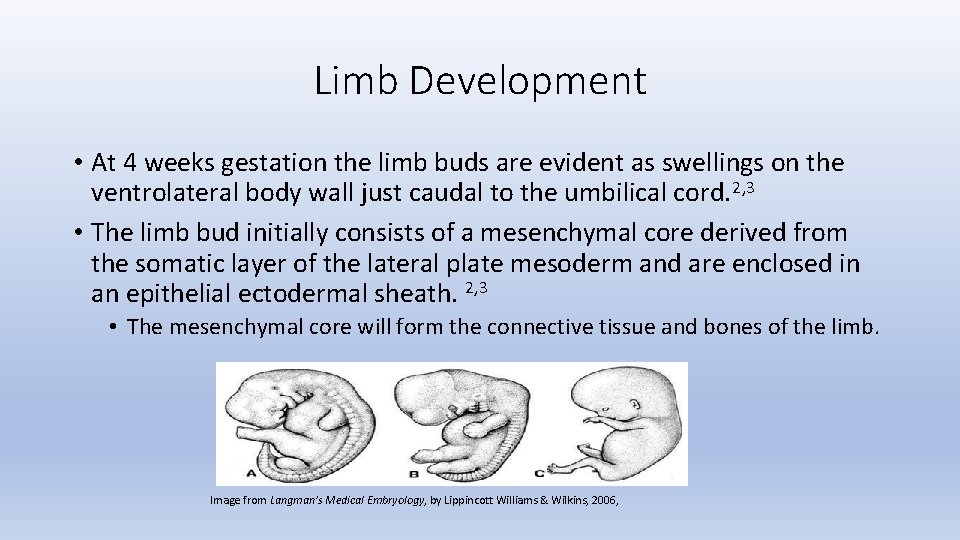
Limb Development • At 4 weeks gestation the limb buds are evident as swellings on the ventrolateral body wall just caudal to the umbilical cord. 2, 3 • The limb bud initially consists of a mesenchymal core derived from the somatic layer of the lateral plate mesoderm and are enclosed in an epithelial ectodermal sheath. 2, 3 • The mesenchymal core will form the connective tissue and bones of the limb. Image from Langman’s Medical Embryology, by Lippincott Williams & Wilkins, 2006,

Limb Development • The ectodermal sheath at the end of the limb bud thickens, through cellular proliferation, and forms the apical ectodermal ridge (AER). • AER is responsible for proximal-todistal differentiation within the bud. • The AER exerts an inductive influence on the mesenchymal layer just beneath, progress zone (PZ), causing the cells to remain undifferentiated and rapidly dividing. 3 • Proliferation within the PZ leads to limb outgrowth. Image from: http: //www. mun. ca/biology/desmid/brian/BIOL 3530/DB_09/fig 9_4. jpg
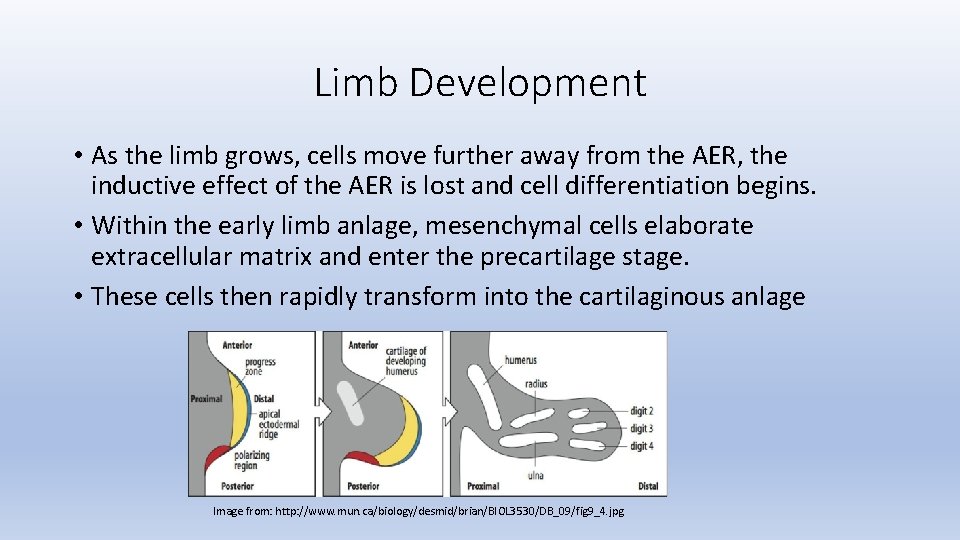
Limb Development • As the limb grows, cells move further away from the AER, the inductive effect of the AER is lost and cell differentiation begins. • Within the early limb anlage, mesenchymal cells elaborate extracellular matrix and enter the precartilage stage. • These cells then rapidly transform into the cartilaginous anlage Image from: http: //www. mun. ca/biology/desmid/brian/BIOL 3530/DB_09/fig 9_4. jpg
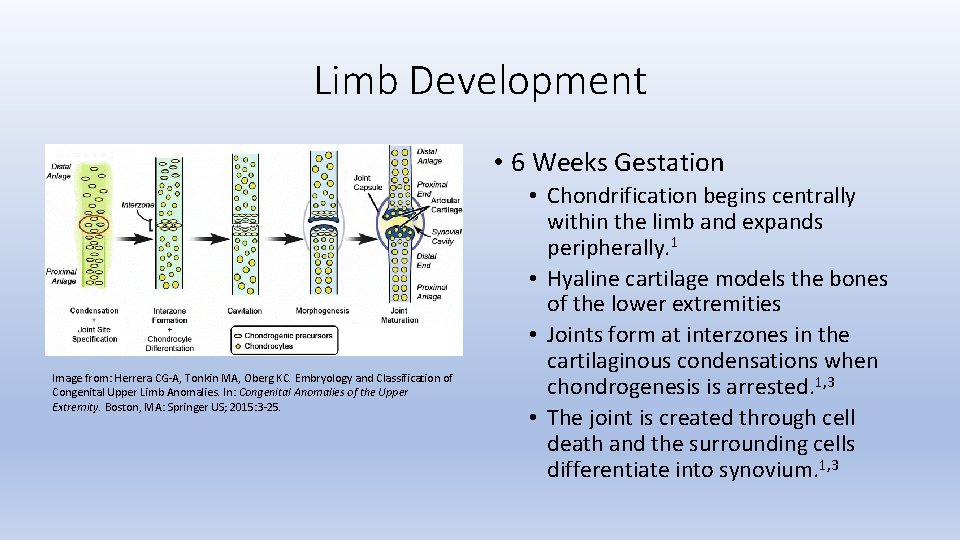
Limb Development • 6 Weeks Gestation Image from: Herrera CG-A, Tonkin MA, Oberg KC. Embryology and Classification of Congenital Upper Limb Anomalies. In: Congenital Anomalies of the Upper Extremity. Boston, MA: Springer US; 2015: 3 -25. • Chondrification begins centrally within the limb and expands peripherally. 1 • Hyaline cartilage models the bones of the lower extremities • Joints form at interzones in the cartilaginous condensations when chondrogenesis is arrested. 1, 3 • The joint is created through cell death and the surrounding cells differentiate into synovium. 1, 3
Limb Development • The perichondrium at the periphery differentiates and establishes continuity with the cartilaginous layers of the interzones. 1 • This continuity is lost when the tissue becomes fibroblastic and differentiates into surrounding ligaments and joint capsule. 1 • The joint capsule differentiates from the intermediate and deep mesoderm • Intermediate and deep mesoderm give rise to perichondrium and periosteum

Limb Development • The interzone has 3 layers: 2 parallel chondrogenic layers and an intermediate, less dense, layer. 1 • Blood vessels penetrate the evolving joint capsule to reach the synovium. 1 • The intra-articular structures, menisci and cruciate ligaments, appear as further cellular condensations within the intermediate mesenchyme. 1

Limb Development • Early in the 8 th gestational week, the cartilage anlage of the lower limb skeletal elements are present, except for the toes. • The basic knee anatomy now resembles that of an adult with the chondroepiphysis forming the articular surfaces. 1 • At the same time, the extensor mechanism begins to function, facilitating motion and joint modelling. 1 • The final joint contours are determined by postnatal joint motion and joint reactive forces. 1

Limb Development • Blastemal cells differentiate within the epiphysis to become articular cartilage. 1 • After birth the distal femur and patella grow through endochondral ossification. 4, 5

Patellofemoral Development • 4 weeks gestation: The patellofemoral joint (PF) is an ectodermal sack filled with mesenchyme of somatic mesoderm. 1 • 4 -5 weeks gestation: mesenchymal condensations appear and chondrification begins within the patella and distal femur. 1

Patellofemoral Development • The cartilaginous patellar and distal femoral condyles are present at 7 weeks gestation. 1 • The patella begins as a dense consolidation of cells within the quadriceps mechanism. 6, 7 • The patellar anlage becomes distinguishable within the tendon near 7. 5 weeks gestation. 6

Patellofemoral Development • Before motor function begins, the knee develops in a 90⁰ position and the cartilaginous patella conforms to the distal femur. 1 • After motion begins, the patella morphology molds and/or remodels in response to mechanical stresses as the knee begins to move. 1

Patellofemoral Development • The patella increases in relative size during the first 6 months of gestation, then proportionately with the rest of the bones. 1 • The patella develops facet predominance, usually lateral facet by 23 weeks and resembles that of the adult patella. 1, 8 • This final shape can be influenced negatively through abnormal contact stresses on the articular surface during skeletal immaturity. 1

Patellofemoral Development • The secondary ossification center of the distal femoral epiphysis forms around 36 weeks gestation. 1 • Ossification for the patella begins around 3 years of age in females and 4 -5 years in males. 8, 9 • The ossification center may appear irregular as it rapidly expands centrifugally. 1, 8 • Ossification extends from proximal to distal. 8 • Patellar height measurements may demonstrate pseudo-patella alta due to incomplete distal ossification. • Full ossification of the patella is reached by 16 -18 years of age. 1 Initial irregular ossification of the patella
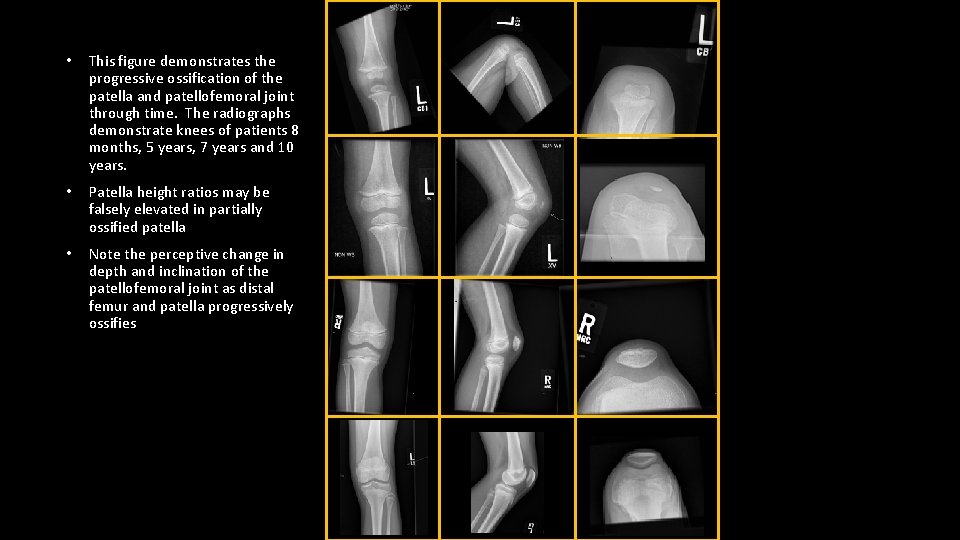
• This figure demonstrates the progressive ossification of the patella and patellofemoral joint through time. The radiographs demonstrate knees of patients 8 months, 5 years, 7 years and 10 years. • Patella height ratios may be falsely elevated in partially ossified patella • Note the perceptive change in depth and inclination of the patellofemoral joint as distal femur and patella progressively ossifies

Trochlear Groove Development • Ultrasound measurements have demonstrated trochlear groove morphology to be present early and becomes more radiographically apparent as distal femoral ossification completes. 10 • An ultrasound study looking at the trochlear grooves in patients 12 -18 years demonstrated the cartilaginous sulcus angle remained constant throughout all age groups. 11 • Anatomic dissections of aborted fetuses have verified that the morphology of the trochlear groove is the same for both fetus and adult. 12 • Morphologic changes in the patellofemoral joint noted on radiographs are representative of progressive ossification of the cartilaginous anatomy that was present as a fetus
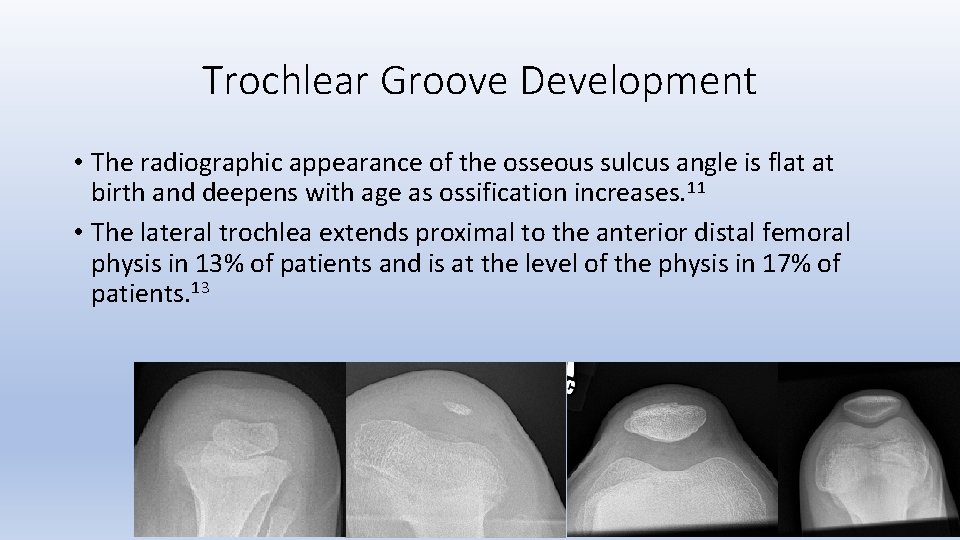
Trochlear Groove Development • The radiographic appearance of the osseous sulcus angle is flat at birth and deepens with age as ossification increases. 11 • The lateral trochlea extends proximal to the anterior distal femoral physis in 13% of patients and is at the level of the physis in 17% of patients. 13

Trochlear Groove Development • The distance between the trochlea and physis increases with age. 13 • The linear measurements for trochlear dysplasia in the distal femur (condylar height, trochlear bump) increase with age but the shape of the trochlear dysplasia does not change with age. 14
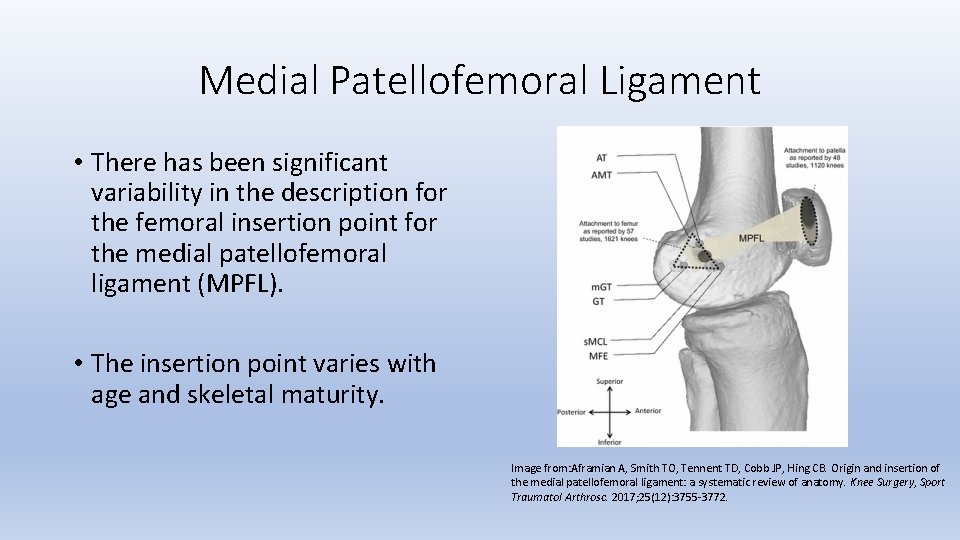
Medial Patellofemoral Ligament • There has been significant variability in the description for the femoral insertion point for the medial patellofemoral ligament (MPFL). • The insertion point varies with age and skeletal maturity. Image from: Aframian A, Smith TO, Tennent TD, Cobb JP, Hing CB. Origin and insertion of the medial patellofemoral ligament: a systematic review of anatomy. Knee Surgery, Sport Traumatol Arthrosc. 2017; 25(12): 3755 -3772.

Medial Patellofemoral Ligament • The patellar insertion for the MPFL has been studied in cadaveric dissections with specimens ages 2 -11 years. The MPFL was noted to be 12 mm in length with the midpoint being 5 mm superior to the mid -pole of the patella. 16 • Several cadaveric studies have verified the femoral insertion of the MPFL to be below the level of the physis. 17– 19
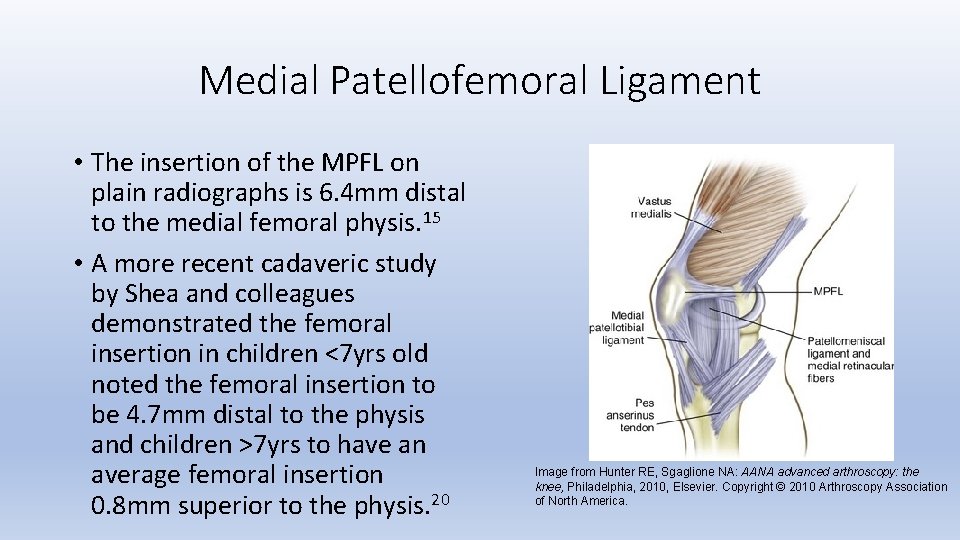
Medial Patellofemoral Ligament • The insertion of the MPFL on plain radiographs is 6. 4 mm distal to the medial femoral physis. 15 • A more recent cadaveric study by Shea and colleagues demonstrated the femoral insertion in children <7 yrs old noted the femoral insertion to be 4. 7 mm distal to the physis and children >7 yrs to have an average femoral insertion 0. 8 mm superior to the physis. 20 Image from Hunter RE, Sgaglione NA: AANA advanced arthroscopy: the knee, Philadelphia, 2010, Elsevier. Copyright © 2010 Arthroscopy Association of North America.

Lower Extremity Alignment • The alignment of the lower extremity changes through the course of growth. • Staheli and colleagues described the normative values of a lower extremity rotational profile including femoral version and tibial torsion. 21 • When toddlers begin standing their feet are wider apart and knees in genu varum. 21
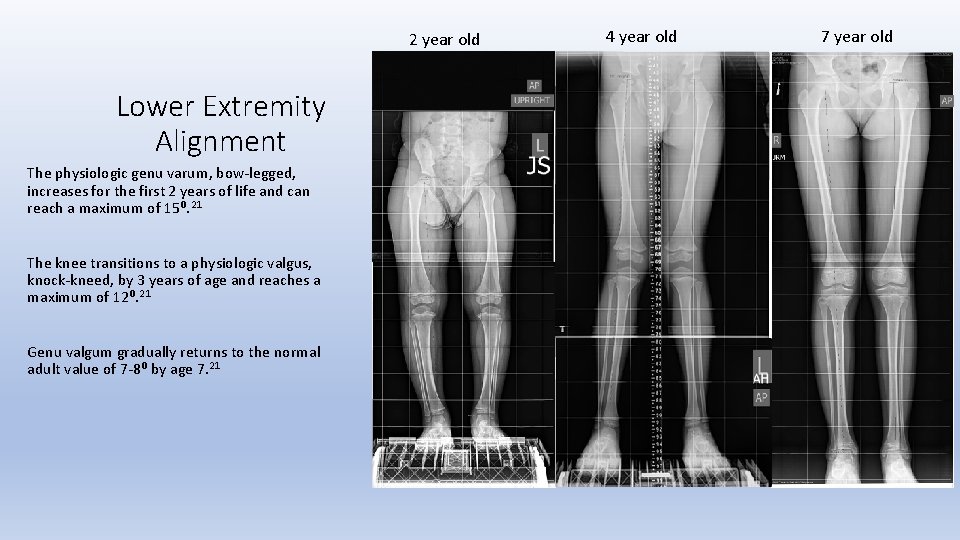
2 year old Lower Extremity Alignment The physiologic genu varum, bow-legged, increases for the first 2 years of life and can reach a maximum of 15⁰. 21 The knee transitions to a physiologic valgus, knock-kneed, by 3 years of age and reaches a maximum of 12⁰. 21 Genu valgum gradually returns to the normal adult value of 7 -8⁰ by age 7. 21 4 year old 7 year old

Rotational Profile • During the 4 th week of gestation the limb bud begins to grow with the great toe in the preaxial position. 22 • During the 7 th week the entire limb rotates medially, bringing the great toe to the midline. 22 • During the remainder of the intrauterine life and through childhood the lower limb rotates laterally. 22

Rotational Profile • Children have increased femoral anteversion that decreases with growth and increasing weight bearing. 23 • The mean femoral anteversion in boys trends from 30⁰ at age 3 years to 10⁰ by age 10 years. 24 • The mean femoral anteversion in girls trends from 40⁰ at age 3 years to 10⁰ by age 20 years. 24

References 1. Wasserlauf BL, Paletta GA. Developmental Anatomy of the Pediatric and Adolescent Knee. In: Micheli LJ, Kocher MS, eds. The Pediatric and Adolescent Knee. 1 st ed. Elsevier; 2006: 27 -32. 2. Morcuende JA, Sanders JO. Embryology and Development of the Neuromuscular Apparatus. In: Weinstein SL, Flynn JM, eds. Lovell and Winter’s Pediatric Orthopaedics. 7 th ed. Philadelphia: Lippincott Williams & Wilkins; 2014: 1 -40. 3. Sadler TW, Thomas W, eds. Skeletal System. In: Langman’s Medical Embryology. 10 th ed. Baltimore: Lippincott Williams & Wilkins; 2006: 125 -142. 4. Sarin VK, Carter DR. Mechanobiology and joint conformity regulate endochondral ossification of sesamoids. J Orthop Res. 2000; 18(5): 706712. 5. Maes C, Kronenberg HM. Postnatal Bone Growth: Growth Plate Biology, Bone Formation, and Remodeling. In: Pediatric Bone. ; 2012: 55 -82. 6. Walmsley R. The development of the patella. J Anat. 1940; 74(Pt 3): 360 -368. 3. 7. Fulkerson J, Hungerford D. Disorders of the Patellofemoral Joint. In: Normal Anatomy: . 2 nd ed. Baltimore: Williams & Wilkins; 1990: 1 -24. 8. Ogden JA. Radiology of postnatal skeletal development. X. Patella and tibial tuberosity. Skeletal Radiol. 1984; 11(4): 246 -257. 9. Birch JG. Growth and Development. In: Herring J, ed. Tachdjian’s Pediatric Orthopedics. 4 th ed. Saunders/Elsevier; 2007: 3 -22. 10. Nietosvaara Y, Aalto K. The cartilaginous femoral sulcus in children with patellar dislocation: an ultrasonographic study. J Pediatr Orthop. 1997; 17(1): 50 -53. 11. Nietosvaara Y. The femoral sulcus in children. An ultrasonographic study. J Bone Joint Surg Br. 1994; 76(5): 807 -809. 12. Glard Y, Jouve J-L, Garron E, Adalian P, Tardieu C, Bollini G. Anatomic study of femoral patellar groove in fetus. J Pediatr Orthop. 2005; 25(3): 305 -308. 13. Parikh SN, Rajdev N. Relationship between Trochlear Dysplasia and the Anterior Distal Femoral Physis in Pediatric and Adolescent Patients with Patellar Instability. In: 36 th Annual Meething AANA. Denver; 2017. 14. Parikh SN, Rajdev N, Sun Q. The Growth of Trochlear Dysplasia During Adolescence. J Pediatr Orthop. March 2018: 1.

References 15. Shea KG, Polousky JD, Jacobs JC, Ganley TJ, Aoki SK, Grimm NL, Parikh SN. The Patellar Insertion of the Medial Patellofemoral Ligament in Children. J Pediatr Orthop. 2015; 35(4): e 31 --e 35. 16. Farrow LD, Alentado VJ, Abdulnabi Z, Gilmore A, Liu RW. The Relationship of the Medial Patellofemoral Ligament Attachment to the Distal Femoral Physis. Am J Sports Med. 2014; 42(9): 2214 -2218. 17. Shea KG, Polousky JD, Jacobs JC, Ganley TJ, Aoki SK, Grimm NL, Parikh SN. The Relationship of the Femoral Physis and the Medial Patellofemoral Ligament in Children. J Pediatr Orthop. 2014; 34(8): 808 -813. 18. Shea KG, Styhl AC, Jacobs JC, Ganley TJ, Milewski MD, Cannamela PC, Anderson AF, Polousky JD. The Relationship of the Femoral Physis and the Medial Patellofemoral Ligament in Children: A Cadaveric Study. Am J Sports Med. 2016; 44(11): 2833 -2837. 19. Nelitz M, Dornacher D, Dreyhaupt J, Reichel H, Lippacher S. The relation of the distal femoral physis and the medial patellofemoral ligament. Knee Surgery, Sport Traumatol Arthrosc. 2011; 19(12): 2067 -2071. 20. Shea KG, Martinson WD, Cannamela PC, Richmond CG, Fabricant PD, Anderson AF, Polousky JD, Ganley TJ. Variation in the Medial Patellofemoral Ligament Origin in the Skeletally Immature Knee: An Anatomic Study. Am J Sports Med. 2018; 46(2): 363 -369. 21. Staheli LT, Corbett M, Wyss C, King H. Lower-extremity rotational problems in children. Normal values to guide management. J Bone Joint Surg Am. 1985; 67(1): 39 -47. 22. Staheli LT. Rotational problems in children. Instr Course Lect. 1994; 43: 199 -209. 23. Staheli LT. In-toeing and out-toeing in children. J Fam Pract. 1983; 16(5): 1005 -1011. 24. Jacquemier M, Glard Y, Pomero V, Viehweger E, Jouve J-L, Bollini G. Rotational profile of the lower limb in 1319 healthy children. 2008; 28(2): 187 -193 25. Aframian A, Smith TO, Tennent TD, Cobb JP, Hing CB. Origin and insertion of the medial patellofemoral ligament: a systematic review of anatomy. Knee Surgery, Sport Traumatol Arthrosc. 2017; 25(12): 3755 -3772. 26. Herrera CG-A, Tonkin MA, Oberg KC. Embryology and Classification of Congenital Upper Limb Anomalies. In: Congenital Anomalies of the Upper Extremity. Boston, MA: Springer US; 2015: 3 -25. 27. Hunter RE, Sgaglione NA: AANA advanced arthroscopy: the knee, Philadelphia, 2010, Elsevier. Copyright © 2010 Arthroscopy Association of North America.
- Slides: 30