PEDIATRIC ENDOCRINOLOGY Diabetes and Thyroid Pearls Minu Mathew

PEDIATRIC ENDOCRINOLOGY Diabetes and Thyroid Pearls Minu Mathew George, MD Medical Director of Clinical Services, Pediatric Endocrinology and Diabetes Attending Physician, OU Children's Physicians/Children’s Hospital Assistant Professor, OU College of Medicine, Dept. of Ped.

Outline: • Pediatric Diabetes Mellitus Type 1 Diabetes Mellitus Type 2 Diabetes Mellitus Other types of Diabetes Mellitus • Pediatric Thyroid Disorders Congenital Hypothyroidism Hashimoto’s Thyroiditis, leading to Hypothyroidism Graves Disease, leading to Hyperthyroidism Thyroid nodules, masses or cancer

Learning Objectives: Learn about the various types of Diabetes in Children q Focus on Type 1 Diabetes, q New technology & State of the art management Learn about Hypothyroidism and reason for it’s prompt treatment in children. q. Congenital Hypothyroidism q. Hashimoto’s thyroiditis Learn about hyperthyroidism including Graves’ disease and an acute thyroid storm and its treatment in children.

DIABETES

Diagnosis • Diagnosis of diabetes mellitus is rarely difficult. • Almost always present at diagnosis: • A history • Polyuria (increased urination) • Polydipsia (increased thirst) • Polyphagia (increased hunger) • Weight loss • Clinical findings • Glucosuria • Ketonuria • Hyperglycemia • A simple urine dipstick for ketones and glucose, available in any clinical setting, is usually adequate for making the diagnosis.

Standards of Clinical Practice: 2018

Other Causes of Diabetes • • • Neonatal Diabetes Genetic Diabetes ( MODY) Pancreatic damage due to trauma CFRD Surgical causes Recurrent Pancreatitis Toxins High-dose glucocorticoids Chemotherapeutic regimens Iron overload Variety of congenital syndromes (rubella) Maternally-inherited mitochondrial diabetes with deafness

Type 1 Diabetes

Type 1 Diabetes • Previously called juvenile-onset diabetes mellitus/insulin-dependent diabetes mellitus • Pancreatic b-cells are progressively destroyed, leading to an absolute deficiency of insulin • Dependence on insulin to prevent ketosis, preserve life • Association with certain HLA phenotypes ( high risk: DR 3 and DQ-2) • Presence of circulating antibodies (GAD 65 Ab, Islet Ab, Insulin Ab, Zinc Transporter 8 Ab) • Co-existence with other autoimmune disorders (Hashimoto’s thyroiditis, Grave’s disease, Addison’s disease, Celiac disease, myasthenia gravis, vitiligo)

Pathogenesis of Type 1 Diabetes • The pathogenesis of type 1 diabetes mellitus is still not fully known • A genetic predisposition compounded by environmental factors • Progressive autoimmune-mediated beta cell destruction • Although some viral infections (mumps, congenital rubella, cox-sackie) have been shown to cause beta cell destruction, a viral association has not been demonstrated conclusively in most cases!

Autoimmune Disease Process Lancet 358, July 21, 2001

Pathogenesis cont’d: • The primary pathologic lesion in type 1 diabetes mellitus is autoimmune destruction of the pancreatic beta cells by • lymphocytic infiltration • T-cell mediated destruction • This process usually begins >1 yr prior to clinical symptoms • At diagnosis, ~90% of the beta cell mass already has been destroyed by the autoimmune process

Prevalence of T 1 DM Gan, MJ 2012

Hb. A 1 c Correlation with BG:
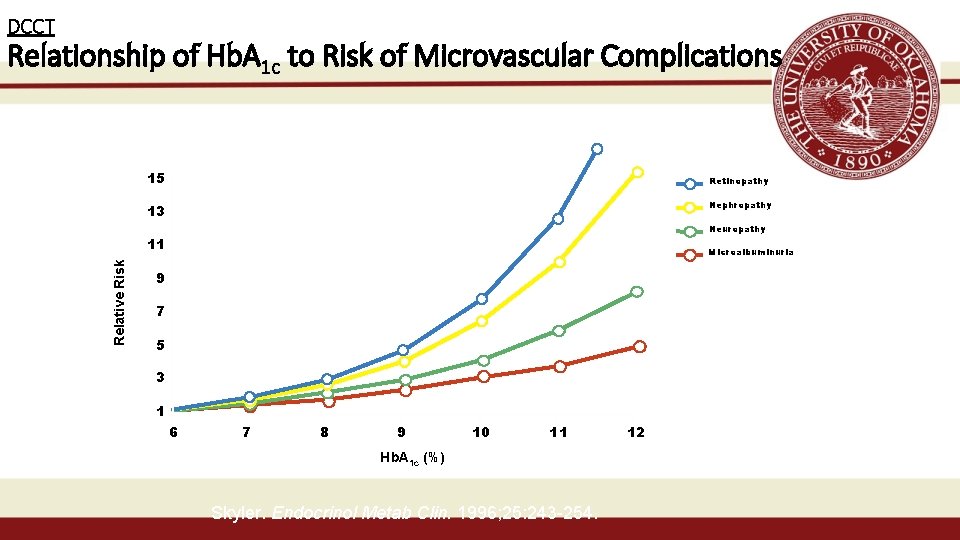
DCCT Relationship of Hb. A 1 c to Risk of Microvascular Complications 15 Retinopathy Nephropathy 13 Neuropathy Relative Risk 11 Microalbuminuria 9 7 5 3 1 6 7 8 9 10 11 Hb. A 1 c (%) Skyler. Endocrinol Metab Clin. 1996; 25: 243 -254. 12

DKA: Insulin-o-penia: • DKA is defined as an absolute or relative deficiency of insulin, resulting in • Hyperglycemia (glucose usually >300 mg/d. L) • Metabolic acidosis • p. H <7. 3 • Serum bicarbonate <15 mmol/L • Accumulation of ketone bodies • Should be considered a medical emergency, especially if the child has not been previously diagnosed with diabetes and/or is severely • Hyperglycemic • Hyperosmolar • Profoundly acidotic

Type 1 Diabetes Treatment

Overall Management Goals • Normal growth and development (including psychological adjustment to a chronic disease) • A 1 c as close to target without hypoglycemia • Best accomplished by a comprehensive multi-disciplinary team • A 1 c goal, although it must be individualized, is now at 7. 5% for all pediatric patients • Glucose goal before meals: 90 -130 mg/d. L; bedtime: 90 -150 mg/d. L • Minimize acute (hypoglycemia, hyperglycemia/DKA) and chronic complications • Monitor and treat concurrent medical conditions associated with diabetes

Insulin Regimens • Traditional/Conventional Insulin Management: • NPH & rapid or short-acting insulin pre breakfast and pre dinner • Intensive Insulin Management • Basal/Bolus with carbohydrate (CHO) counting • Basal: Long-acting (Lantus® or Levemir ®) • Bolus: Rapid-Acting • CSII (Pump)
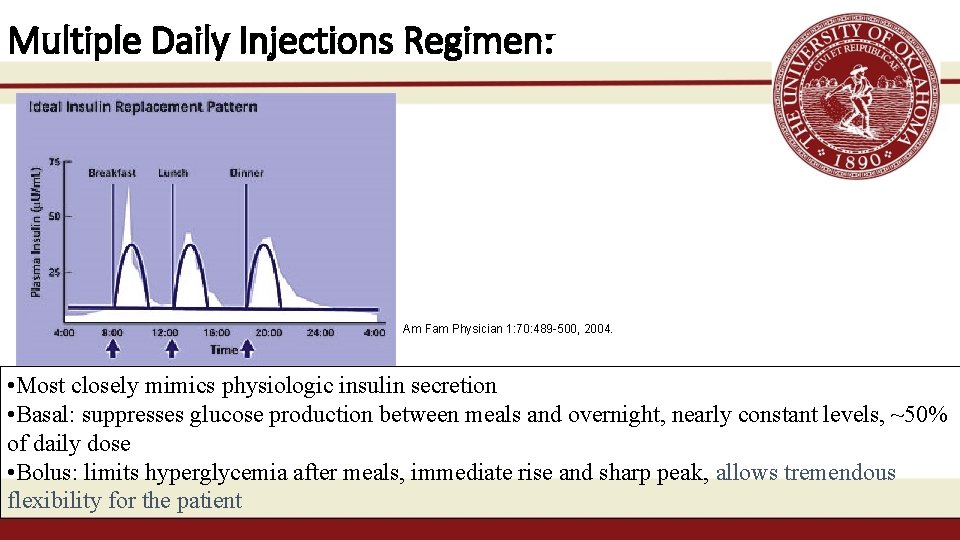
Multiple Daily Injections Regimen: Am Fam Physician 1: 70: 489 -500, 2004. • Most closely mimics physiologic insulin secretion • Basal: suppresses glucose production between meals and overnight, nearly constant levels, ~50% of daily dose • Bolus: limits hyperglycemia after meals, immediate rise and sharp peak, allows tremendous flexibility for the patient
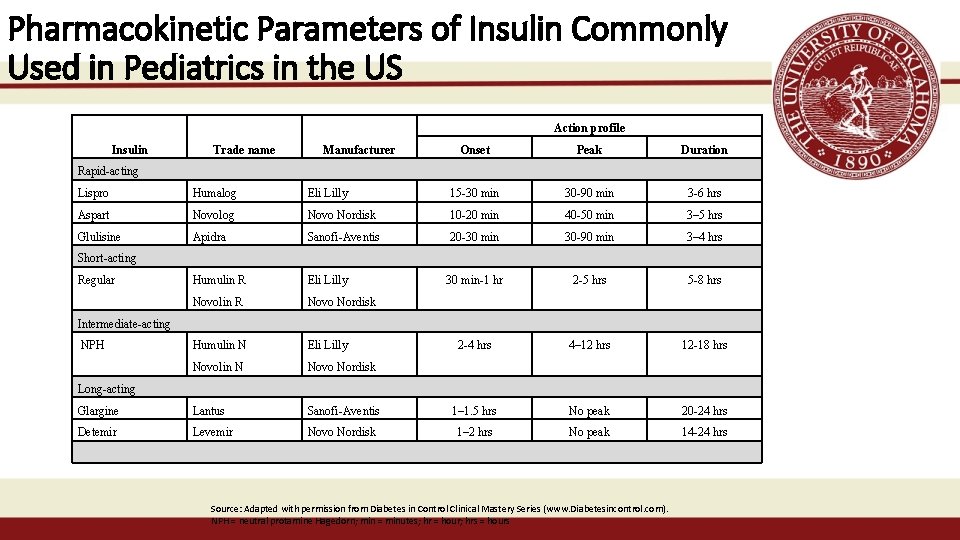
Pharmacokinetic Parameters of Insulin Commonly Used in Pediatrics in the US Action profile Insulin Trade name Manufacturer Onset Peak Duration Rapid-acting Lispro Humalog Eli Lilly 15 -30 min 30 -90 min 3 -6 hrs Aspart Novolog Novo Nordisk 10 -20 min 40 -50 min 3– 5 hrs Glulisine Apidra Sanofi-Aventis 20 -30 min 30 -90 min 3– 4 hrs Humulin R Eli Lilly 30 min-1 hr 2 -5 hrs 5 -8 hrs Novolin R Novo Nordisk Humulin N Eli Lilly 2 -4 hrs 4– 12 hrs 12 -18 hrs Novolin N Novo Nordisk Glargine Lantus Sanofi-Aventis 1– 1. 5 hrs No peak 20 -24 hrs Detemir Levemir Novo Nordisk 1– 2 hrs No peak 14 -24 hrs Short-acting Regular Intermediate-acting NPH Long-acting Source: Adapted with permission from Diabetes in Control Clinical Mastery Series (www. Diabetesincontrol. com). NPH = neutral protamine Hagedorn; min = minutes; hr = hour; hrs = hours

Insulin Pen Devices

Basal Insulin • Lantus® (glargine) or Levemir® (detemir) or newer options • Usually ONCE daily, at bedtime • Even if the patient is not eating or skips a meal or the blood sugar is within target at bedtime: Basal insulin given once daily • DO NOT mix with other insulins • MUST check 2 -3 AM blood sugars the first few days on therapy

Calculating Insulin Dosing: Total Daily Dose (TDD) • Starting doses: • 9 months-2 years: 0. 25 -0. 5 Units/kg/day • 1 yr-6 yr: 0. 5 Units/kg/day • 7 yr-puberty: 0. 75 -1. 0 Units/kg/day • Adolescence: 1 -1. 5 Units/kg/day • Basal insulin: ~50% of total daily insulin dose • Insulin to Carb Ratio: Rule of 500 • Insulin to Correction Factor: Rule of 1800

Insulin to Carbohydrate Ratio • Use only Humalog, Novolog or Apidra • Prior to meals and snacks when using the carbohydrate ratio • Only exception to giving insulin after meals is with toddlers. Orders should be written to dose post-meal.

Insulin Calculation: Rule of 500 • Multiple Daily Injections • Rule of 500: Used to calculate CHO ratio • 500/TDD = 1 unit of insulin to cover “x” number of carbohydrates (CHO) • Example: 500/20 (TDD) = 25 • Give 1 unit of insulin for every 25 g of CHO

Insulin Calculations: Rule of 1800 • Multiple Daily Injections • Rule of 1800: Used to calculate Correction Factor (CF) • 1800/TDD = 1 unit of insulin drops bloods sugar approximately “x” mg/d. L • Example: 1800/20 (TDD) = 90 • Giving 1 unit of insulin will drop blood glucose about 90 mg/d. L

Correction Factor: Basic Concepts • When newly diagnosed • Generally used at: • meal-times; not snacks • do not correct at bedtime

Insulin Dose Calculation: Example • 9 year old, 34 kg pre-pubertal patient • 0. 75 units/kg/day = 25 • 25 units total of insulin per day (TDD) • Basal insulin (Lantus/Levemir) is ½ TDD or 12 units/day • CHO: • 500/TDD • 500/25 = 20 units (CHO ratio 1: 20) • CF: • 1800/TDD • 1800/25 = ~ 70 • Given to correct blood sugar back into target range • i. e. 1: 70 > 150 • (may give CF dose even if blood sugar is not 210 mg/d. L – especially is younger children --- we teach families to do the math calculation and determine each time)

Diabetes Education* • Diabetes Team Assessment/Family Education • Basic review of pathophysiology • Target blood glucose values/use of a blood glucose meter and when to monitor blood glucose values • Insulin (action, how to draw up, administer, rotate sites, etc. ) • CHO Counting • Effects of exercise • Signs/Symptoms of hypoglycemia and treatment • Glucagon emergency use • Signs/Symptoms of hyperglycemia, treatment and sick day management • Importance of DM identification bracelet, necklace, etc. • All diabetes related prescriptions, school forms, HIPAA, etc. • Family support – child-life (inpatient) and psychology services (in and outpatient settings) *Remainder of education will occur as an outpatient

Glucometers:

Standard Monitoring Times • Before breakfast • Before lunch • Before dinner • Bedtime • 2 am (in hospital and at home for at least the first few day and with any insulin dose changes • Symptomatic – hyper or hypoglycemia

Symptoms of Hypoglycemia § Shaking/trembling § Sweating § Excessive hunger § Irritability, crying § Headache § Dizziness § Confusion § Drowsiness § Appears dazed

Treatment of Hypoglycemia : Rule of 15 Eat or drink 15 grams of quick-acting carbohydrates (CHO). (4 oz. juice, 8 oz. skim milk, 3 -4 glucose tablets) Recheck blood sugar in 15 minutes. Repeat steps 1 and 2, if necessary, until blood sugar is greater than 80. Once blood sugar is greater than 80, if a mealtime, provide insulin accordingly based on ratio. If it is not a mealtime, give a 15 gram CHO and protein snack (examples: ½ meat sandwich, peanut butter crackers, etc).

Symptoms of Hyperglycemia § Increased thirst § Increased going to the bathroom § Increased hunger § Feeling sleepy § Visual disturbance § Stomach pain § Vomiting

Hyperglycemia : If FSBS > 300 mg/d. L ü Check ketones. If NEGATIVE, TRACE, OR SMALL URINE KETONES: 1. Have patient drink at least 8 oz. water every hour. 2. Give correction factor at mealtimes 3. Recheck blood sugar in 2 hours. 4. Repeat above until blood sugar less than 300 mg/d. L.

Hyperglycemia Treatment • If ketones MODERATE TO LARGE: ü Use correction factor if at least 2 hours since last correction regardless of time of day. ü Have patient drink at least 8 oz. of water every hour. ü Recheck blood sugar and ketones in 2 hours. ü Continue to treat until blood sugar less than 300 mg/d. L AND ketones are small or less.

Insulin Pumps

Repeat DKA and Pump Therapy • Treat DKA/other concurrent illness • Problem-solve (concurrent illness/missed insulin, etc. ) • Pumps: • If you suspect patient is not giving insulin boluses (insulin omission) • Have patient show you their bolus history • Blood sugars remain high. • Suspect a problem with the insertion site. Give correction bolus with a syringe not the pump. • Have patient change the insertion site, insulin, tubing, etc. • If an insulin pump is removed, SQ insulin must be given: All patients with type 1 diabetes need basal insulin without delays. • If a patient resumes pump therapy – watch the timing: Do not accidentally overdose.

Continuous Glucose Monitors (CGM) • CGM measures glucose in the interstitial fluid every 5 minutes • Sensor information converted to a glucose value displayed on receiver and/or pump • Lag time of ~15 minutes between FSBS & interstitial fluid • Patients must still test blood sugar as directed • For device calibrations • All treatment decisions still based on FSBS
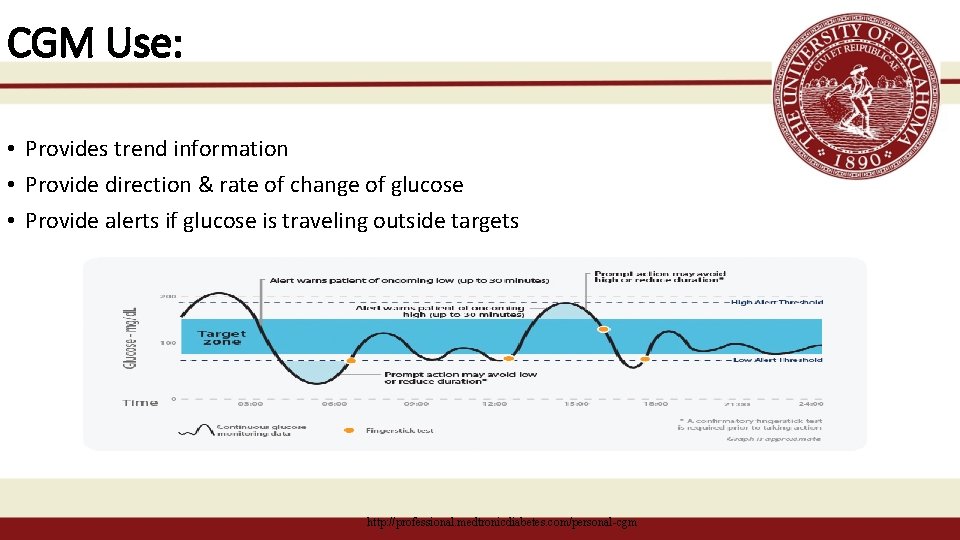
CGM Use: • Provides trend information • Provide direction & rate of change of glucose • Provide alerts if glucose is traveling outside targets http: //professional. medtronicdiabetes. com/personal-cgm

CGM Products • Personal • • Medtronic Guardian® REAL-Time CGM System Medtronic Minimed Paradigm® REAL-Time REVEL™ System Medtronic Minimed® 530 G with Enlite® Dexcom G 5™ Platinum • Professional • Medtronic i. Pro® 2 • Dexcom® SEVEN® PLUS-when set to “Blinded Mode”
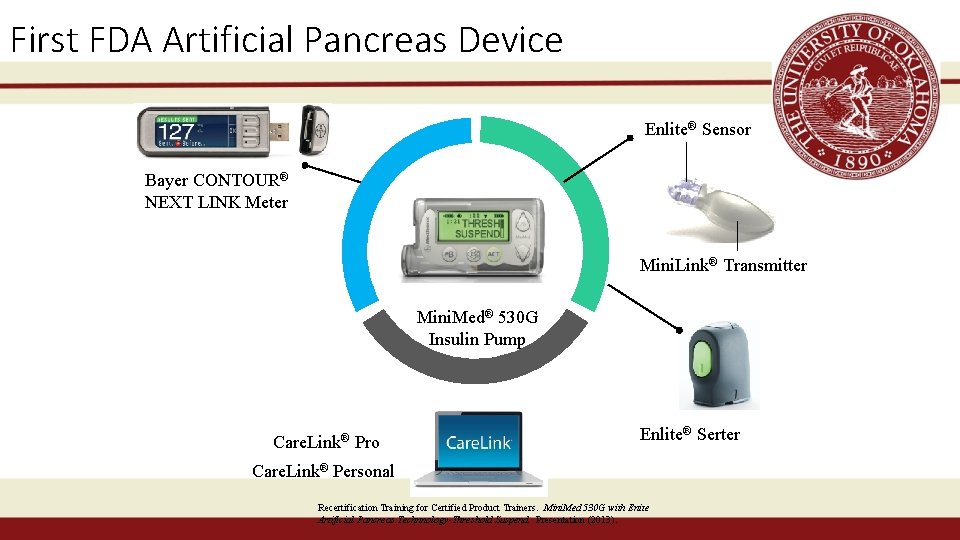
First FDA Artificial Pancreas Device Enlite® Sensor Bayer CONTOUR® NEXT LINK Meter Mini. Link® Transmitter Mini. Med® 530 G Insulin Pump Care. Link® Pro Enlite® Serter Care. Link® Personal Recertification Training for Certified Product Trainers. Mini. Med 530 G with Enite Artificial Pancreas Technnology-Threshold Suspend. Presentation (2013).

OUCP Pediatric Diabetes Team: § Team Members: 6 BC-MD’s, PNP, Pharm. D, Psychology, RD/LD, 5 CDE’s, RN’s, LPN’s and MA’s and Clinic team § New onset gets full diabetes education (~8 hours) § All patients are provided 24/7 emergency pager access to the team § All diabetes prescriptions and other necessary supplies taken care of by the diabetes team § All patients will have follow-up with the outpatient DM team (the diabetes team will coordinate this appointment time)

Distinguishing Characteristics between Type 1 and Type 2 Diabetes in Youth in 2018 Type 1 Type 2 Usually childhood, but some dx well into Usually adult, but some dx in youth adulthood Short to long course Slow course Usually thin, but may be obese with recent weight loss Usually obese; always obese in youth Often present with ketoacidosis Up to 33% of youth with type 2 DM have ketonuria at dx; 5 -25% present in DKA Increased incidence of other Increases in PCOS, HTN, TG elevated, autoimmune dx; thyroid, adrenal, celiac LDL low, AN (up to 90%) dz Caucasians predominate NA, AA, Latino, Asian, Pacific Islander

Type 2 Diabetes (not the same at Type 2 in adults) “A whole different animal” TODAY STUDY

Type 2 Diabetes • Risk Factors • Obesity • Sedentary lifestyle • Certain ethnic groups • Native American, Hispanic, African American • Family history • Gestational diabetes • Signs of insulin resistance • Acanthosis Nigricans

Ramifications of this New “Epidemic” of Type 2 Diabetes in Children • Family physicians care for ~90% of all diabetes cases in the US • Are aware of this new epidemic of T 2 DM in youth • Tend to treat them the same way they treat an adult with T 2 DM • Almost all type 1 diabetes in the US is managed by Pediatric Diabetes & Endocrinology Centers/Teams • General pediatricians are largely untrained in the management of childhood diabetes • TODAY Study Results and Oklahoma’s Rol

Questions to ask What might help distinguish type 1 from type 2? • Blood tests? • Antibodies… “Gold Standard” for diagnosis • C-peptide or insulin levels prior to treatment? • Obese or not? • Family history • Ethnicity • Ketosis or not?

What do we know… § 30 -35% of patients newly diagnosed with type 1 diabetes mellitus (T 1 DM) are overweight or obese § Trial. Net study has revealed that some patients with T 1 DM may go months or years between a biochemical diagnosis and requirement for insulin § 3 -fold increase in family history of T 2 DM among patients with T 1 DM § Up to 33% T 2 DM cases have ketosis at disease onset

Clinical Pearls § The emergence of type 2 diabetes in youth has presented us with substantial diagnostic dilemmas and treatment challenges § Types 1 and 2 diabetes in youth initially may appear similar or identical, and the traditional factors used to distinguish type 1 from type 2 (age, obesity, ketones) may be of limited value § Therefore certain factors, including ketosis, ketoacidosis, or extreme hyperglycemia dictate immediate initiation of insulin at diagnosis in an obese child with new-onset diabetes

Distinguishing Characteristics between Type 1 and Type 2 Diabetes in Youth… 2017 Type 1 Type 2 Usually childhood, but some dx well into adulthood Usually adult, but some dx in youth Short to long course Slow course Usually thin, but may be obese with recent weight loss Usually obese; always obese in youth Often present with ketoacidosis Up to 33% of youth with type 2 DM have ketonuria at dx; 5 -25% present in DKA Increased incidence of other autoimmune Increases in PCOS, HTN, TG elevated, LDL dx; thyroid, adrenal, celiac dz low, AN (up to 90%), NAFLD Caucasians predominate NA, AA, Latino, Asian, Pacific Islander

Clinical Recommendations • Clinicians must ensure that insulin therapy is initiated for children and adolescents with T 2 DM • Who are ketotic or in diabetic ketoacidosis, • Who have venous or plasma glucose levels >250 mg/d. L • Who have hemoglobin A 1 c >9%; or • In whom the distinction between type 1 and type 2 diabetes is unclear

Summary: Type 2 Diabetes in Children/Adolescents • Recommended Reading: • Copeland KC, Silverstein J, Moore KR, Prazar GE, Raymer T, Shiffman RN, et al. • Management of newly diagnosed type 2 diabetes mellitus (T 2 DM) in children and adolescents. Pediatrics. 2013 Feb; 131(2): 364 -82. • Inpatient – if presenting in DKA – medical management may look similar to those with type 1 diabetes • Outpatient • Lifestyle changes – exercise and dietary changes • Multiple daily injections • Combination insulin and oral agents • Oral agents and aggressive off-label combination therapy to avoid complication

Diabetes Keto. Acidosis Insulin deficiency & Counter regulatory Hormones
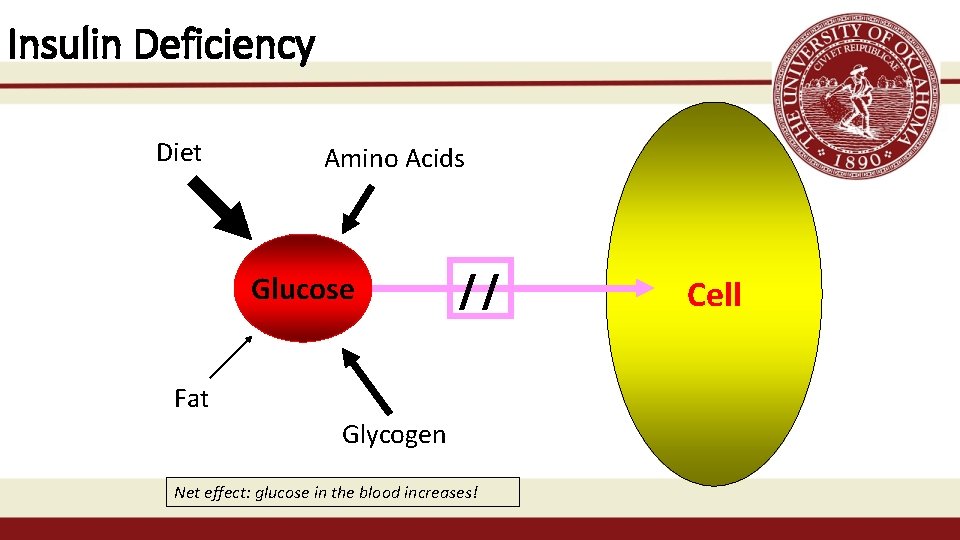
Insulin Deficiency Diet Amino Acids Glucose // Fat Glycogen Net effect: glucose in the blood increases! Cell

DKA • DKA is defined as an absolute or relative deficiency of insulin, resulting in: • D: Hyperglycemia (glucose usually >300 mg/dl) • K: Accumulation of ketone bodies • A: Metabolic acidosis • p. H <7. 30 • serum bicarbonate <15 mmol/L

DKA • This can occur in the following situations: • Untreated newly diagnosed type 1 or 2 DM. • At the time of b cell failure in a type 2 DM. • Ineffectively treated type 1 or 2 DM. • Noncompliance with insulin shots • Inadequate dose • Use of expired or inactive insulin • Insulin pump failure • Treated type 1 diabetes with stress or steroids • Illness • Trauma • Surgery

Initial Management • The following steps should be performed within the first 30 min after presentation: • Focused and rapid history and physical • Laboratory tests, bedside and to lab • Begin hydration

Treatment • If bedside tests confirm DKA, begin hydration immediately as follows: • Establish venous access through a large vein • Begin a 20 ml/kg bolus of NS (if clinically appropriate) • The bolus should be given at the maximum rate allowed by the venous catheter (usually within 30 -60 min) unless there is evidence of renal compromise (hematuria, oliguria)

Treatment • Consider establishing the following ancillary lines: • a second venous line for insulin drip and maintenance fluids • additional venous access for blood draws • if patient is unconscious or comatose, arterial line for blood gases and blood sampling • If seriously obtunded or unconscious: Foley catheter (to facilitate accurate determinations of fluid balance).

Insulin • Insulin is essential to the management of DKA, and should be started as soon as the diagnosis has been verified, the patient is in a controlled critical care environment and the initial fluid bolus is complete. • Unless long (>2 hour) delays are expected, insulin should not be administered by the referring physician at an outlying center prior to transport.

Two Bag Method • The standard method for preparing an insulin drip is as follows: • Mix regular human insulin equal to patient's weight (in kg) into 100 ml of 0. 5 NS • If drip is mixed correctly as detailed above, 10 ml/hr = 0. 1 U/kg/hr insulin. • Flush tubing with 10 ml of drip, then piggyback into IV fluids. • Once insulin drip is prepared, run at a constant rate (0. 05 units/kg/hr for <6 years old or 0. 1 units/kg/hr for >6 years old) • Hang two bags of fluid with identical composition except for dextrose content • One containing no dextrose and the other containing 10% dextrose • Sodium, potassium and phosphate content should be based upon initial laboratory results • In the absence of hyperkalemia, K should be added after a void

Two Bag Method • The total infusion rate for both solutions should be 150% maintenance • Maintain FSBS between 200 and 300 by titrating the relative rates of the 10% dextrose and dextrose-free solutions • Insulin infusion rate should be decreased ONLY if the FSBS is below 150 with only the 10% dextrose solution running at 150% maintenance • Under this circumstance insulin infusion should be reduced to • 0. 03 units/kg/hr for < 10 years old • 0. 05 units/kg/hr for > 10 years old

What To Monitor • Electrolytes: q hour x 2, then q 4 hours • Blood glucose: q hour while on an insulin drip • Calculate and plot • anion gap (Na+K-Cl-HCO 3) • uncorrected sodium • corrected sodium (add 1. 6 meq to Na for each 100 mg/dl glucose above 100 mg/dl) • Strict I/O's and daily weights while on parenteral fluids/insulin drip or when seriously obtunded.

Cerebral Edema • Cerebral edema (although rare) is the most serious complication of DKA • When it occurs, it is rapid, fulminant, progressive, and difficult to reverse • Cerebral complications typically occur in the first 24 hrs of treatment, during a period when biochemical parameters are improving • Early symptoms that may indicate cerebral edema include new onset or intensifying headache, altered consciousness, and recurrent vomiting; ultimately progressing to increasing blood pressure, bradycardia, and opisthotonic posturing • Recent studies indicate that many children with DKA have CT evidence for mild, subclinical brain swelling during therapy; however, the great majority recovers uneventfully

What To Monitor • Neurologic Signs q hour x 12 hours, then as indicated • If neurologic signs (including headache) should appear • fluid replacement and insulin should be slowed • bicarbonate replacement stopped • mannitol ordered to bedside and given within the first 15 minutes of neurologic deterioration (0. 5 -1 gm/kg IV over 5 -10 minutes).

Cerebral Edema • Fluid Volume • Some data indicate a relationship between the volume of fluids administered during treatment of DKA and clinically significant cerebral edema. • A fluid volume of 4 L/m 2/day has been proposed as a risk boundary. • Severe Acidosis • Some data support an association between acidosis and cerebral edema • Increased with bicarbonate therapy • Elevated BUN • This may reflect greater dehydration

DKA Morbidity and Mortality • Mortality 0. 15% in US • Cerebral edema accounts for 57 -87% of all DKA deaths • Incidence of cerebral edema 0. 87%

Resolution of DKA • When acidosis is corrected • p. H >7. 3 • OR bicarbonate >15 • AND patient is able to tolerate PO • insulin drip may be discontinued and SQ insulin given

Oklahoma's Largest Group of Peds Endo’s: • If your patient has diabetes, you know it takes full-time attention • An ADA recognized this Center with its award for outstanding diabetes self-management program We provide the following services for children with type 1 and type 2 diabetes: • 24 -hour-a-day telephone access to our pediatric endocrinologists and certified diabetes educators • Prompt evaluation of new-onset pediatric diabetes cases • Daily monitoring of newly diagnosed patients over the telephone for the first week • Return clinic visits within two weeks for non-urgent medical issues • Initial and ongoing type 1 and type 2 diabetes education classes • Certified pump instructors

NOW SWITCHING TO THYROID

Thyroid Fetal Development Fetal thyroid begins as a thickening of the pharyngeal floor Descends caudally forming the thyroglossal duct The fetal bilobed shape is recognized by 7 th week of gestation Synthesis of thyroid hormones starts by 11 -12 wks of gestation During the pregnancy, transplacental passage remains an important source of fetal thyroid hormone • At birth, in response to neonatal (cold) extrauterine exposure there is an acute release of TSH (the TSH surge), followed by increase in T 4 levels at 24 to 36 hrs of life • TSH remains elevated for 3 to 5 days after birth. • • •
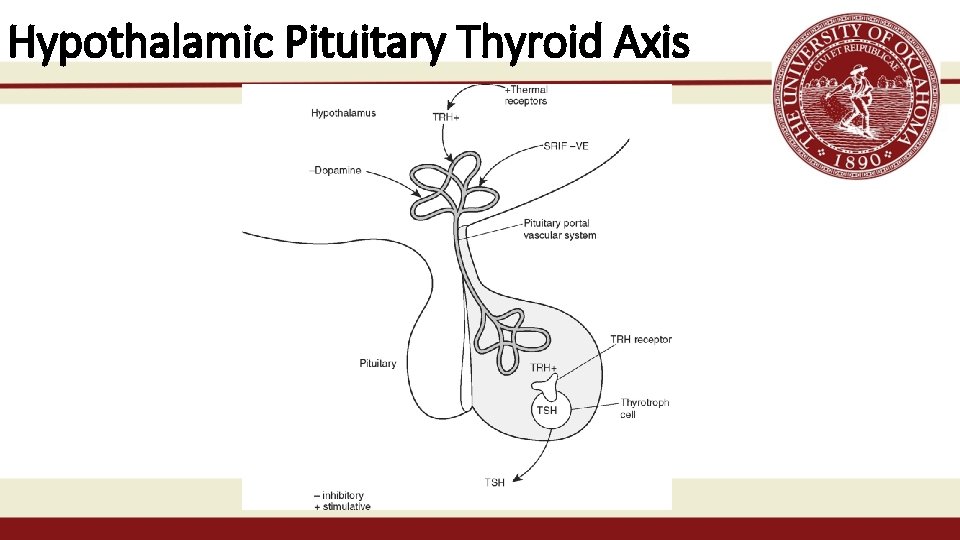
Hypothalamic Pituitary Thyroid Axis
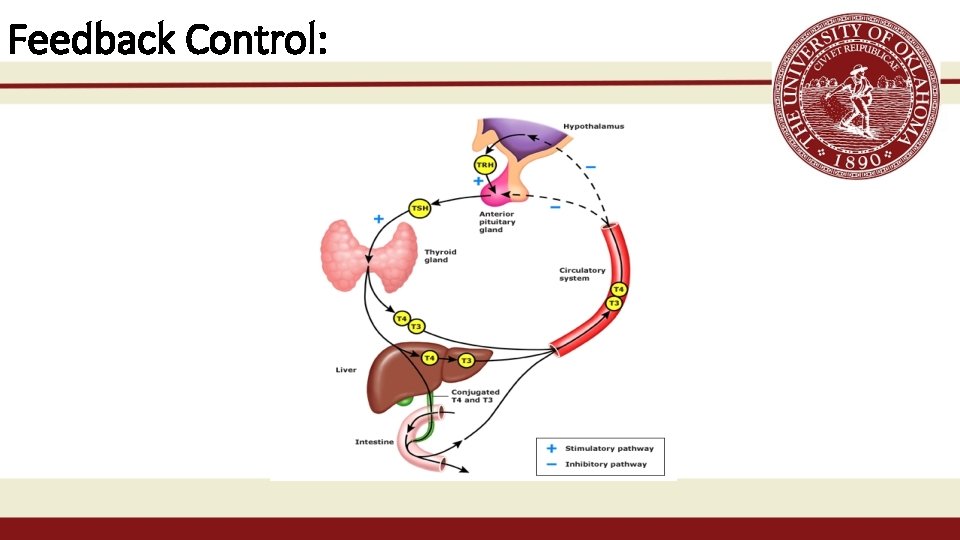
Feedback Control:
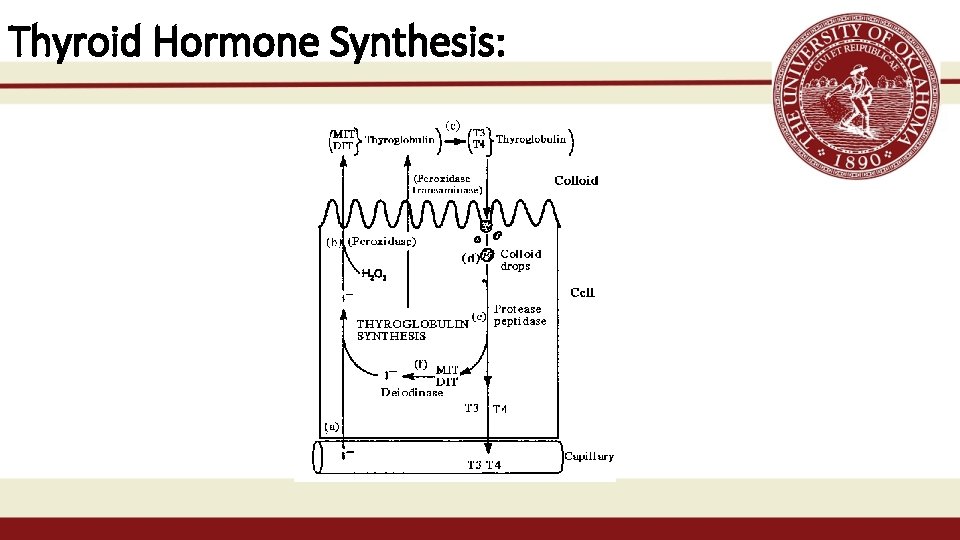
Thyroid Hormone Synthesis:
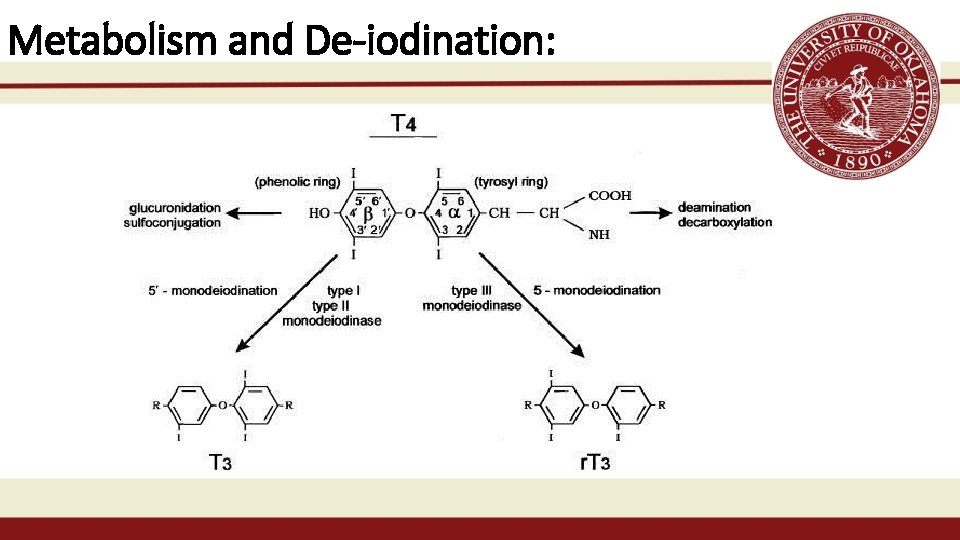
Metabolism and De-iodination:

Lab Testing: • The most sensitive test of thyroid function is the TSH • Free T 4 and TSH are the 2 most common tests ordered and the most useful combination. • T 3 is the active thyroid hormone, and reverse T 3 is the inactive metabolite. • T 4 = Total T 4 and is the sum of FT 4 + bound T 4 • TBG is one of the carriers/binding proteins for thyroid hormones in blood along with albumin.

Congenital Hypothyroidism: (CH) • Decreased T 4 production in a newborn. • #1 preventable cause of potential intellectual disability. • Incidence- approximately 1: 4000 newborns

CH: Clinical Manifestations �Prolonged Jaundice (Direct) �Feeding Difficulty �Lethargy �Umbilical Hernia �Macroglossia �Constipation �Cold or mottled skin �Hypothermia �Abnormal cry �Edema �Hypothyroid appearance �Hypotonia

CH: Newborn Metabolic Screening �Blood from a heel-prick collected on special filter paper �Collected between two and five days of age � Some programs also routinely obtain a 2 nd specimen between 2 and 6 weeks of age �Many screening programs carry out an initial TSH test �detects infants with primary hypothyroidism and can detect infants with subclinical hypothyroidism (normal T 4, elevated TSH) �misses infants with delayed TSH elevation and with hypothalamic or pituitary hypothyroidism �Other states measure levels of T 4, followed by measurement of TSH when T 4 is low �identifies infants with primary hypothyroidism, some with hypothalamic or pituitary hypothyroidism, and infants with a delayed increase in TSH levels
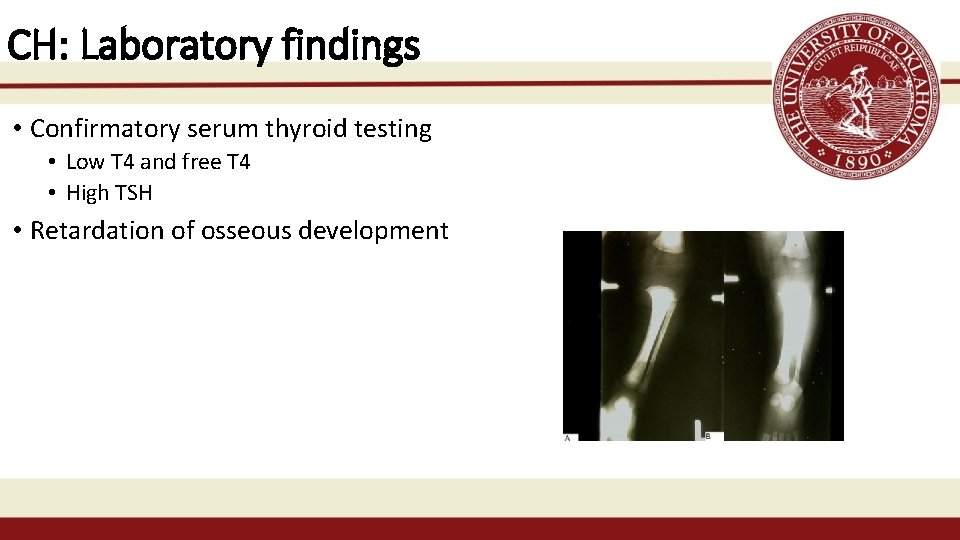
CH: Laboratory findings • Confirmatory serum thyroid testing • Low T 4 and free T 4 • High TSH • Retardation of osseous development

CH: Treatment & Goals • Levothyroxine (l-thyroxine)- starting dose 10 -15 mcg/kg • Thyroid suspensions may result in unreliable dosing. • The l-thyroxine tablet should be crushed, mixed in a small volume of breast milk, formula or water and fed to the infant. • The tablet should not be mixed with soy formula as this has been shown to interfere with absorption. • Do not dose it with Calcium and Fe containing meds. • Serum Free T 4 or total T 4 should be kept in the upper range of normal during the first year of life • Serum TSH should be kept under 5 m. U/L ideally at 2 m. U/L.
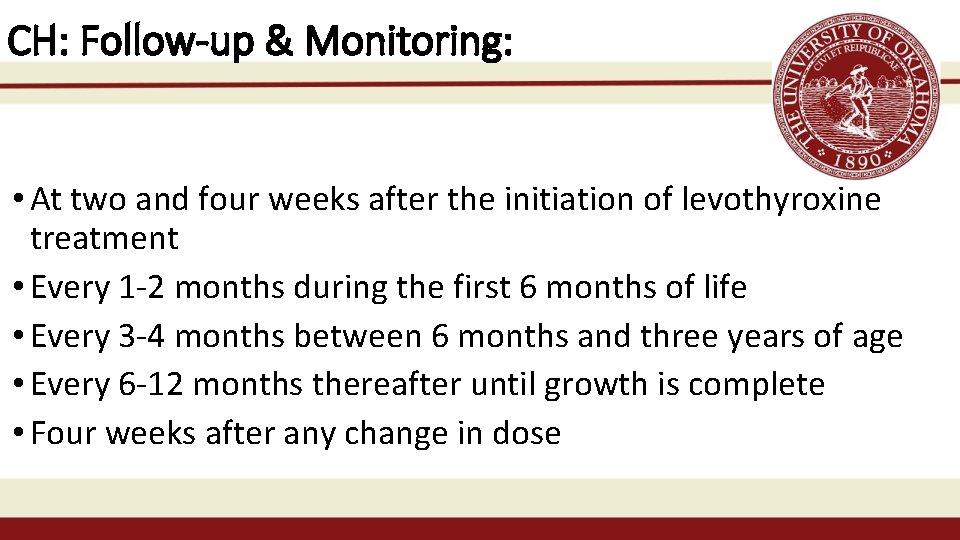
CH: Follow-up & Monitoring: • At two and four weeks after the initiation of levothyroxine treatment • Every 1 -2 months during the first 6 months of life • Every 3 -4 months between 6 months and three years of age • Every 6 -12 months thereafter until growth is complete • Four weeks after any change in dose

CH: Transient or Permanent • 10% to 20% of babies diagnosed as having congenital hypothyroidism have transient hypothyroidism. • If the diagnosis of permanent hypothyroidism is not clear, T 4 therapy can be stopped for 1 month when the child reaches 3 years of age and thyroid function retested off therapy. • Permanent hypothyroidism if the serum T 4 value is low and the TSH value is elevated, and thyroid replacement restarted. • If the T 4 and TSH values remain normal off tx, transient hypothyroidism.

Prognosis: • Babies born with congenital hypothyroidism who are treated adequately and promptly (in the first 2 postnatal weeks) grow and develop normally. • Children who are treated inadequately in the first 2 wks to 3 years after birth have IQs below those of unaffected children.

Acquired Hypothyroidism

Acquired Hypothyroidism: Primary hypothyroidism �Autoimmune- Hashimoto thyroiditis �Iatrogenic - PTU, methimazole, iodides, lithium, Amoidarone - Irradiation /Radioiodine - Thyroidectomy Central hypothyroidism (deficiency of TSH) – Craniopharyngioma and other tumors pressing on hypothalamus/pituitary – Neurosurgery/ Cranial irradiation/ Head trauma Peripheral - Resistance to thyroid hormones - Large hemangiomas of the liver

Hashimoto thyroiditis: (Chronic Lymphocytic) • The most common cause of acquired childhood hypothyroidism • More common in females • Usually occurs in early to mid-puberty • Female-to-Male ratio is 2: 1 • Incidence of Hashimoto’s during adolescence is approximately 1% to 2% • May occur by itself or in association with other autoimmune diseases; (type 1 diabetes mellitus, Addison disease, juvenile idiopathic arthritis, and systemic lupus erythematosus) • Occurs more commonly in individuals who have Down syndrome or Turner syndrome

Hashimoto’s: Clinical Presentation • Fatigue • Cold intolerance • Somnolence • Proximal muscle weakness • Delayed relaxation phase of ankle reflex • Constipation • Delayed growth • Overweight for height; not morbidly obese • Pallor • Coarse and thick skin • Enlargement of thyroid gland - diffuse and nontender • Bradycardia • Irregular menstrual cycles • Delayed puberty (occasionally precocious puberty)

AH: Growth Curves • The onset of acquired childhood hypothyroidism often is very subtle • In retrospect, it may be evident that signs and symptoms were present for a longer time, sometimes for 2 to 3 or more years • Decline in linear growth from the onset of hypothyroidism • Weight not as excessive as you would have imagined
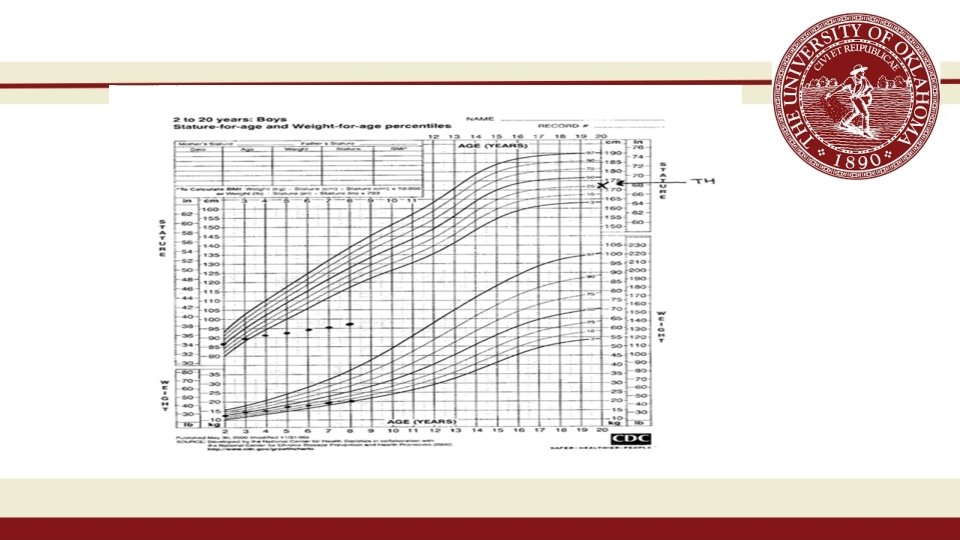

AH: Laboratory findings • low serum T 4 or low serum free T 4 • elevated serum TSH in Primary • thyroid antibody testing (thyroid peroxidase antibody, antimicrosomal antibodies, thyroglobulin antibody) • Bone age - delayed

Hashimoto: Treatment • Thyroid hormone replacement. • 6 to 12 months - 5 to 8 mcg/kg of body weight • 1 to 3 years - 4 to 6 mcg/kg • 3 to 10 years- 3 to 5 mcg/kg • 10 to 18 years - 2 to 4 mcg/kg • Serum free T 4 and TSH concentrations should be monitored periodically, preferably at 3 - to 6 -month • The goal is to keep the serum free T 4 concentration at the mid-normal range and the TSH concentration in the normal range

AH: Prognosis • The prognosis for recovering lost linear growth depends on the duration of the hypothyroidism as well as the age at which treatment is started. • If the onset of childhood hypothyroidism occurs after age 2 to 3 years, no permanent intellectual damage or neurologic deficit is likely. • Overall, the prognosis of acquired childhood hypothyroidism is good if diagnosed in time and hypothyroidism is not of longstanding duration.

Acquired Hypothyroidism: (AH) Primary hypothyroidism �Autoimmune- Hashimoto thyroiditis �Iatrogenic – PTU, methimazole, iodides, lithium, Amoidarone - Irradiation - Radioiodine - Thyroidectomy Central hypothyroidism (deficiency of TSH) – Craniopharyngioma and other tumors pressing on hypothalamus/pituitary – Neurosurgery – Cranial irradiation – Head trauma Peripheral - Resistance to thyroid hormones - Large hemangiomas of the liver

Hyperthyroidism GRAVES DISEASE �Incidence 1 : 5, 000 children �The peak incidence is in the 11 - to 15 -yr old �There is a 5 : 1 female to male ratio �Most children with Graves disease have a positive family history of some form of autoimmune thyroid disease.

HYPER: Clinical Findings • • Emotional lability Disturbed sleep habits (night-mares) Hyperactivity and short attention span Poor school performance Increased appetite but weight loss Diarrhea and increased GI motility Heat intolerance and excessive perspiration Muscle weakness and fatigue

HYPER: Clinical Findings Goiter: Diffuse, smooth and non-tender Bruit with stethoscope bell Variable size, can be small Eyes: Proptosis (muscle and proteoglycans) Eye muscle weakness Lid lag Corneal abrasion (open during sleep)

HYPER: Clinical Findings Cardiovascular Tachycardia, wide pulse pressure Bounding precordium, sharp pulse Cardiac output increased Neurological Brisk reflexes, hyper-irritability

HYPER: Laboratory findings • Elevated, free T 4, T 3 • Low TSH • TSIG (TRAB) elevated • Antithyroglobulin, antiperoxidase antibodies present in low titers • Thyroglobulin (TG) is elevated (low in factitious) • Bone age can be advanced • 4 hour and 24 hour uptakes are elevated

HYPER: Treatment Factors influencing treatment choice �Age �Sex (R/O early pregnancy) �Severity and duration of the disease �Size of the gland �Presence of other complicating conditions �Available surgical expertise �Likelihood of compliance

HYPER: Symptomatic Initial Treatment Propranolol 2 mg/kg/day q 12 h Blocks peripheral T 4 conversion to T 3 Side-effects: Bradycardia, hypoglycemia Contraindications: � asthma, heart block

GRAVES: Radio-iodine Treatment �Long track record of safety �Outline precautions (Contamination) �R/O pregnancy �Post-radiation 1. Expect increased T 4 in first 2 weeks because of radiation thyroiditis 2. Treatment failure in severe cases 3. Hypothyroidism after 2 -4 months

HYPER: Antithyroid drugs • Toxicity (skin, marrow, liver, kidney) • Propylthiouracil is no longer recommended • Methimazole (Tapazol) is the preferred drug • Drug failure • Non-compliance • Large gland less likely to respond • Treatment required averages 4. 5 y
HYPER: Surgery �Illness or contraindication for medical Rx �Requires preparation (Lugol’s iodide and Methimazole) �Goals: Decrease T 4 for anesthesia and blood flow Decease vascularity Increase consistency Risks: �Hypoparathyroidism and recurrent laryngeal nerve palsy

Neonatal Graves Disease (NGD) q. Happens in 2% of infants born to mothers with a history of Graves �Transplacental passage of TSIG �Delayed onset: PTU: 3 -4 days TRBAb: several weeks �The disorder usually remits spontaneously within 6 -12 wk

NGD: Clinical Presentation • Tachycardia • Goiter • Irritability • Hyperthermia • Heart failure • Advanced bone age • Cranial synostosis

NGD: Treatment �Neonatal Graves Disease remits in 6 to 12 weeks �Rx: Propranolol 2 mg/kg/day 3 x daily PTU 5 -10 mg/kg/day 10% K iodide, 1 drop q 8 h Digoxin (DC inderal) � Follow gland size, free T 4 and TSIG � Taper doses after 6 to 12 weeks

Thyroid Nodules

Thyroid Nodule • Solitary nodules of the thyroid are common in children. • They are found by palpation in approximately 2% of children aged 11 -18 • Most thyroid nodules in children are benign • Percentage of nodules that harbor cancer is closer to 2%. • Children exposed to radiation have a high incidence of benign adenoma and carcinoma of the thyroid.
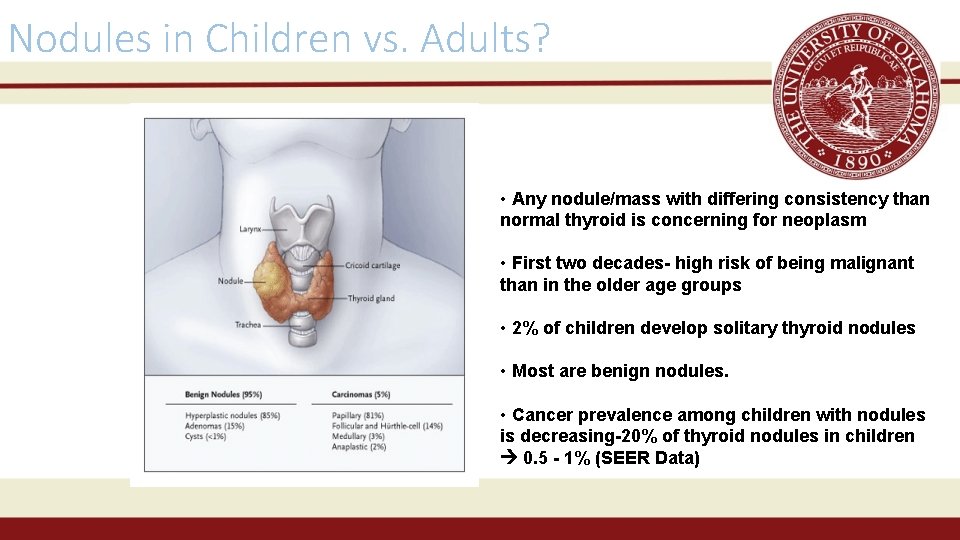
Nodules in Children vs. Adults? • Any nodule/mass with differing consistency than normal thyroid is concerning for neoplasm • First two decades- high risk of being malignant than in the older age groups • 2% of children develop solitary thyroid nodules • Most are benign nodules. • Cancer prevalence among children with nodules is decreasing-20% of thyroid nodules in children 0. 5 - 1% (SEER Data)

Nodule: Diagnostic Studies • Thyroid function tests • Antithyroid antibody • Ultrasonographic examination of the thyroid • Fine needle aspiration (FNA) • Radioiodine uptake and scan • All persons with a history of head or neck irradiation should have careful examinations of the thyroid at least every 2 yr and indefinitely
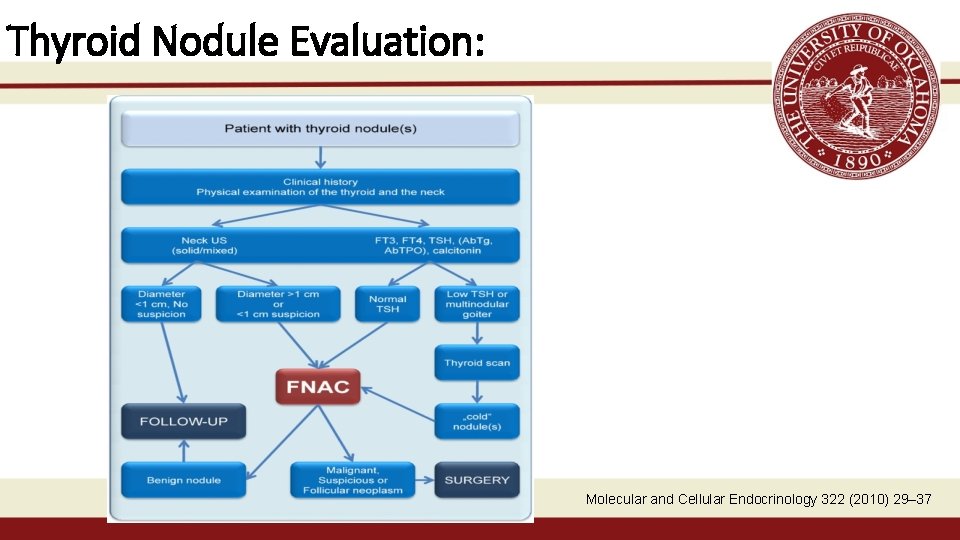
Thyroid Nodule Evaluation: Molecular and Cellular Endocrinology 322 (2010) 29– 37

Thyroid Gland Disorders
- Slides: 115