Pediatric Case Management By Dr Rachel Gast MD

Pediatric Case Management By Dr. Rachel Gast MD & Dr. Apryle Funderburk MD March 2 nd 2010
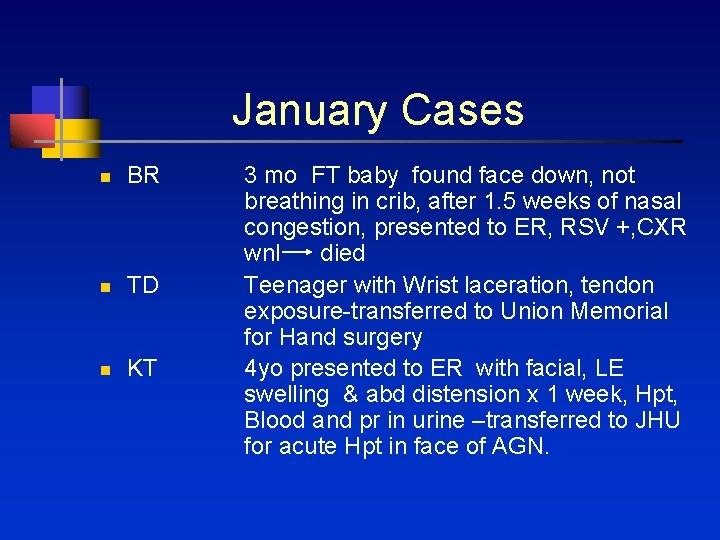
January Cases n BR n TD n KT 3 mo FT baby found face down, not breathing in crib, after 1. 5 weeks of nasal congestion, presented to ER, RSV +, CXR wnl died Teenager with Wrist laceration, tendon exposure-transferred to Union Memorial for Hand surgery 4 yo presented to ER with facial, LE swelling & abd distension x 1 week, Hpt, Blood and pr in urine –transferred to JHU for acute Hpt in face of AGN.

HPI n n n 15 year old male Headache x 2 days Pain in middle of back on morning of admission radiating to chest Progressive weakness in L. E. bilaterally Patient First → St. Joseph’s E. D.

Past History n n n PMH ADHD, exercised-induced asthma, depression, migraines Meds Atomoxetine, Trazadone Allergy Shellfish, wheat, soybeans, peanuts Vacc UTD, received H 1 N 1 (I. M. ) one month prior Fam Hx Mother-COPD, arthritis, Father Hepatitis B & cysts in brain, “misalignment” in spine, healthy siblings Social: Attends 10 th grade, lives with mom & step-dad and cats, mom smokes

Physical Exam – St. Joseph E. D. n n Pulse = 58, B. P. = 160/100 General: unable to walk, nausea with emesis x 1 n n n n n Moving legs per ED physician; absent reflex in right LE and diminished in RE Weak grasp ↑ tone in upper extremities with intact reflexes WBC = 8. 6 Hgb = 14. 8/Hematocrit = 43. 8 Platelets = 275 Electrolytes and coags normal Toxicology screen = normal CXR/Head CT/ECG = normal

n n Solu-medrol 125 mg, IV Labetolol Zofran Transferred to Sinai PICU

Case #2 n n 7 yr. old male with juvenile-onset D. M. type 2, in usual state of health Flu-like symptoms week prior to presentation n n Day prior to presentation – at school – left leg weakness, limping, gradual loss of motor function n n “Bronchitis” – prednisone, clarithromycin, albuterol No tingling, numbness nor loss of sensation No incontinence ED – progression of weakness, loss of function, areflexia; symptoms starting in opposite leg Admitted to PICU for further work-up

P. E. /Labs n n P. E. – Flaccid left LE, strength 0/5, intact sensation and vibratory sense, ↓ tone in right LE, motor 0 -1/5, able to dorsi-/plantar flex right foot, absent DTRs, negative Babinski Labs – n n n CBC WBC 13. 7 ( N 51 L 36, M 9. 2, E 2. 2, B 0. 2) CSF WBC 168 (N 38, L 50, M 12 RBC = 59, Glu 95, Pr 59 CSF (-)GS/Cx Stool Cx (+) for heavy Candida albicans EBV Ig. M 2. 3 , VCA Ab Ig. G positive, EBNA Ab Ig. G positive Anticardiolipin Ab Ig. M positive

Objectives n n To discuss updates on current influenza activity To discuss neuronal injury from influenza / influenza vaccine To discuss adverse events from H 1 N 1 2009 influenza strain vs. those from H 1 N 1 vaccine To discuss current CDC statement concerning H 1 N 1 vaccine
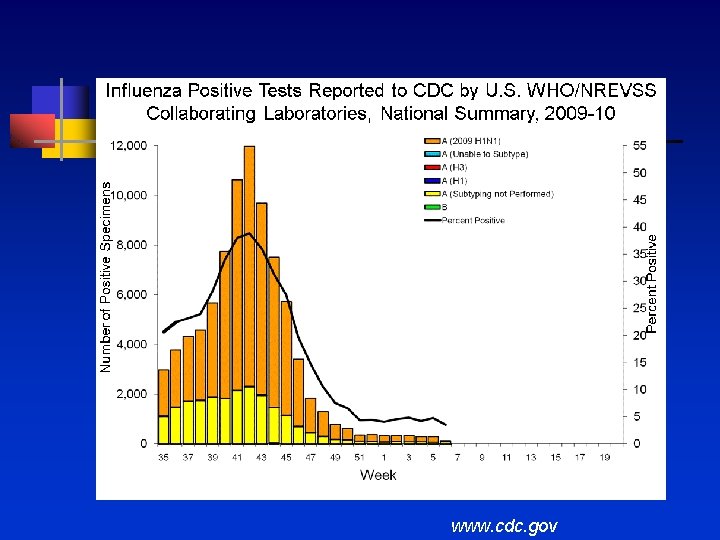
www. cdc. gov
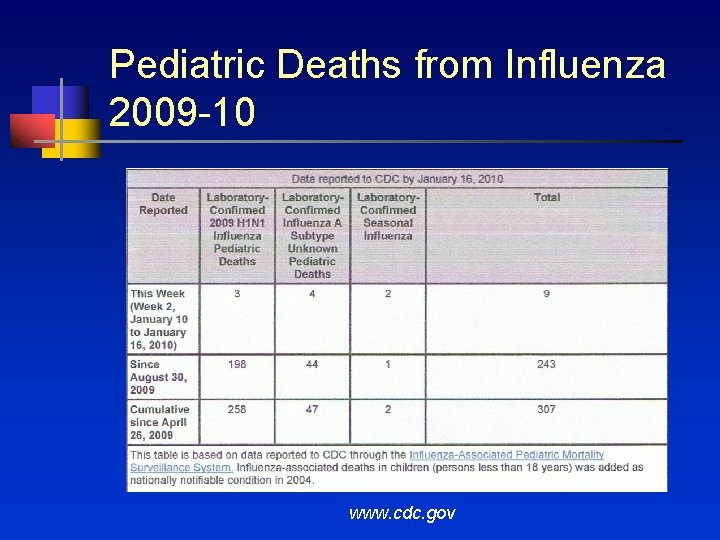
Pediatric Deaths from Influenza 2009 -10 www. cdc. gov

Influenza Vaccine n Seasonal Flu – trivalent inactivated, live attenuated – 3 virus strains n A n n n B n n H 1 N 1 H 3 N 2 For the 2009 --10 influenza season, the influenza B vaccine virus strain was changed to B/Brisbane/60/2008, a representative of the B/Victoria lineage, compared with the 2008 --09 season. The influenza A, H 1 N 1 and H 3 N 2 vaccine virus strains were not changed “Swine” Flu – n A/California/07/2009

Mechanism of neuronal Injury • • Vaccinations may induce autoimmune process n Influenza vaccines made in chicken eggs which are endemically infected with Campylobactor n Antibodies cross-react against peripheral-nerve antigen However, the immunologic process that leads to GBS or other neuronal injury is largely unknown

Vaccine Adverse Event Reporting System n n National reporting system jointly administered by CDC Immunization Safety Office and FDA n reports submitted voluntarily by people who believe an adverse event occurred after vaccination May be submitted healthcare providers, patients, or family members VAERS staff follow-up on all serious and other selected adverse event reports Data does not infer causality

Addressing parents’ concerns: do vaccines contain harmful preservatives, adjuvants, additives or residuals? Offit, Paul A. , Jew, Rita K. Pediatrics, 2003 n n n Preservatives Adjuvants Additives Residuals Antibiotics Cellular residuals
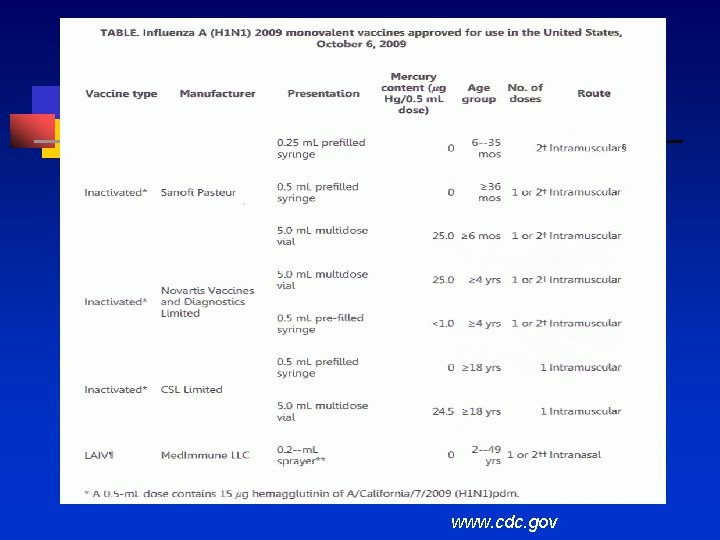
www. cdc. gov
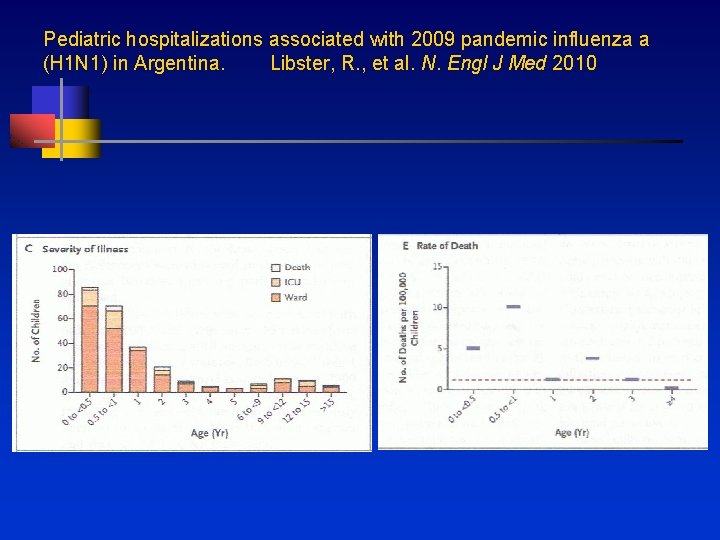
Pediatric hospitalizations associated with 2009 pandemic influenza a (H 1 N 1) in Argentina. Libster, R. , et al. N. Engl J Med 2010
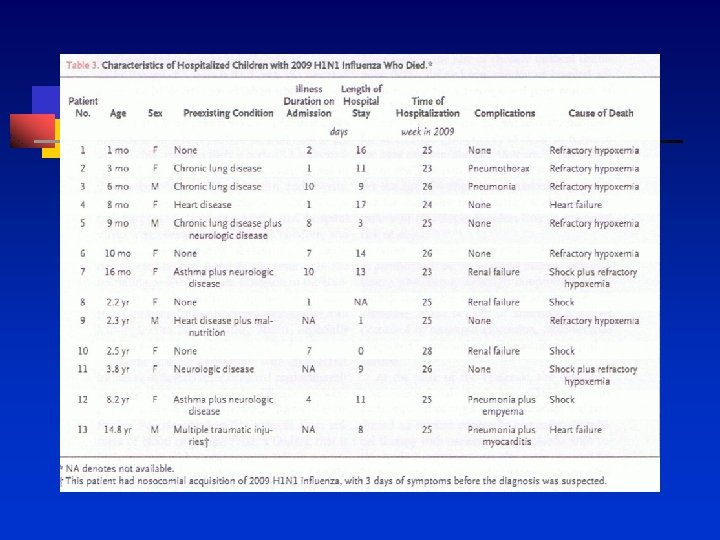

Vaccine 27 (2009) 2114 -2120 n n n 1990 -2005 Adults > 18 years of age 747. 1 million doses of TIV Event reporting rate to VAERS of 24. 4 per million TIV 18, 245 (14%) were classified as serious events
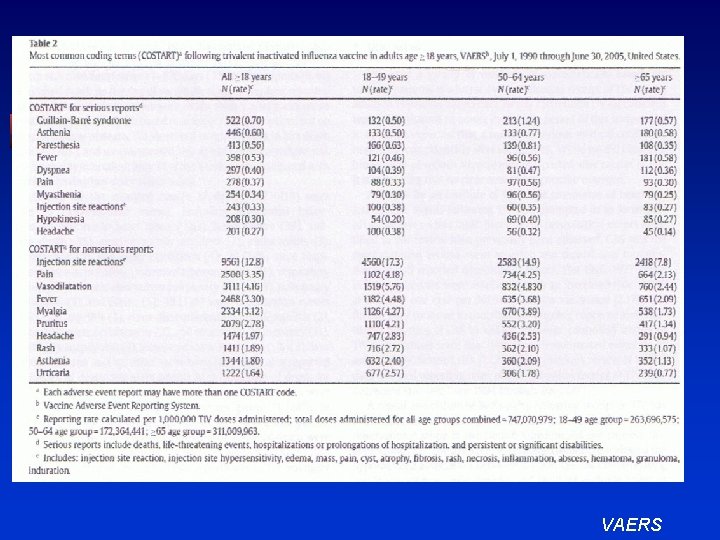
VAERS
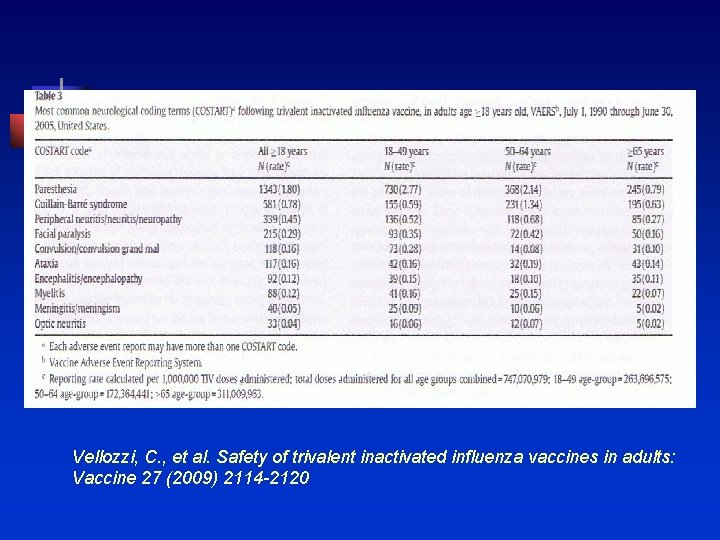
Vellozzi, C. , et al. Safety of trivalent inactivated influenza vaccines in adults: Vaccine 27 (2009) 2114 -2120

Lancet 2010; 375: 49 -55 • Pandemic vaccine – Fluval P – monovalent vaccine with 6 υg haemagglutinin per 0. 5 ml content and aluminum phosphate gel adjuvent (n = 178) • Seasonal vaccine – Fluval AB - trivalent inactivated whole-virion (n = 177)
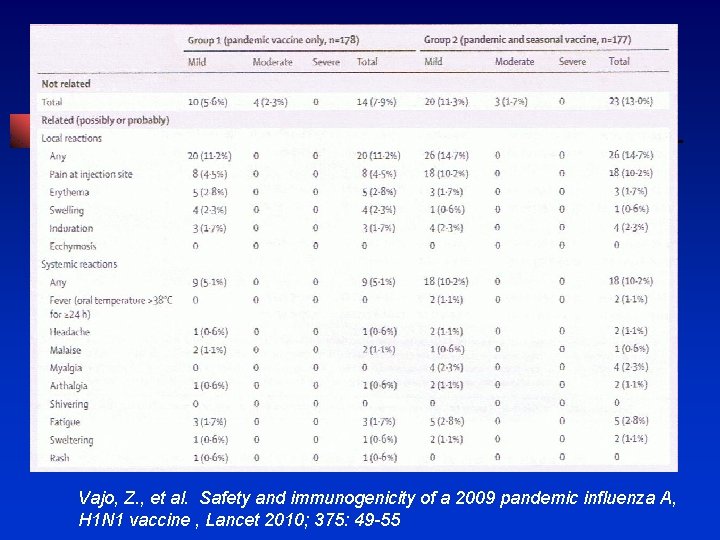
Vajo, Z. , et al. Safety and immunogenicity of a 2009 pandemic influenza A, H 1 N 1 vaccine , Lancet 2010; 375: 49 -55

VAERS

GBS and H 1 N 1 vaccination n n October 1997 – the Advisory Committee on Immunization Practices of the U. S. Public Health Service recommendations noted: “ Among persons who received the swine influenza vaccine in 1976, the rate of Gullain-Barre syndrome that exceeded the background rate was slightly less than 10 cases per million vaccinated. Even if Guillain-Barre syndrome were a true side effect in subsequent years, the estimated risk for Guillain-Barre syndrome of 1 -2 cases per million persons vaccinated is substantially less than that for severe influenza…”

CDC Statement n n As of November 24, VAERS had received 10 reports of Guillain-Barré syndrome, and two additional reports of possible Guillain-Barré syndrome were identified by medical officers reviewing other reports to VAERS describing neurologic events After chart review, four of these 12 reports met Brighton Collaboration criteria for Guillain -Barré syndrome, four did not meet the criteria, and four are under review

References n n n n Libster, R. , et al. Pediatric hospitalizations associated with 2009 pandemic influenza a (h 1 n 1) in argentina. N Engl J Med 2010; 362: 45 -55 Vellozzi, C. , et al. Safety of trivalent inactivated influenza vaccines in adults: background for pandemic influenza vaccine safety monitoring. Vaccine 27 (2009) 2114 -2120 Kerr, Douglas A. , Ayetey, Harold. Immunopathogenesis of acute transverse myelitis. Current Opinion in Neurology 2002, 15: 330 -347 Mossad, Sherif B. The resurgence of swine-origin influenza a (h 1 n 1). Cleveland Clinic Journal of Medicine Volume 76 Number 6 June 2009 Haber, P. , et al. Guillain-barre syndrome following influenza vaccination. JAMA, November 24, 2004—Vol 292, No. 20 Scheibner, Viera. Adverse effects of adjuvents in vaccines. Nexus Dec 2000 (Vol 8, No 1) & Feb 2001 (Vol 8, Number 2) Vajo, Z. , et al. Safety and immunogenicity of a 2009 pandemic influenza a h 1 n 1 vaccine when administered alone or simultaneously with the seasonal influenza vaccine for the 2009 -10 influenza season: a multicentre, randomised controlled trial. Lancet 2010; 375: 49 -55. MMWR. “Update on Influenza A (H 1 N 1) 2009 Monovalent Vaccines. ” October 9 th, 2009. http: //www. cdc. gov/mmwr/preview/mmwrhtml/mm 5839 a 3. htm
HPI n C. A n Day 1 n n Day 2 -Taken to PMD n n n Severe HA, b/L hip pain, low grade fever, generalized malaise/ mylagias, H/o visit to Nigeria 3 weeks prior. Congestion /T-103 with HA dx as acute sinusitis Sent home on Bactrim Day 3 - Taken to first OHS n n n Symptoms worsen with persistent fevers Chest-xray –negative/Rapid strep positive Sent home with Pen VK (no h/o sore throat)
HPI n Day 3 n n Symptoms persisted plus onset of abdominal pain/vomiting Taken to 2 nd OHS ER n n n CT-negative for acute abdominal process/patchy infiltrate of LLL- on chest x-ray Thrombocytopenia/elevated LFT’s/hyponatremia/ febrile Patient admitted for w/u of poor clinical status and abnormal labs

HPI n Day 4 -5 n HD 2 -3 n n Worsening thrombocytopenia, onset of anemia and hyponatremia with elevated creatinine Persistent fevers n n n CMV neg, EBV positive, Hepatitis panel neg, Mono spot neg, Urine cx / Blood cx neg HD 2 -Malaria Smear obtained HD 3 - Worsening labs/ Smear positive for P. falciparum n n Obtained first dose of atovaquine-proguanil Hypotensive to 80/50’s with heart rate 100’s/dizzy/ decreased UOP transferred to Sinai PICU

Physical Examination, Day 5 n Day 5, Transfer to Sinai PICU n n n n n VS: General: Skin: HEENT: Wt-74. 6 kg, B. P-100/50, H. R-90’s, R. R-18 100% on 1 LNC Tired appearing but interactive No lesion or rashes No oropharyngeal erythema, PERRL, anicteric, no nystagmus Neck: Normal ROM with some tenderness CV: RRR, S 1 S 2 normal. No murmurs, rubs, gallops. cap refill < 2 sec Lungs: Occasional course BS with adequate/equal air entry Abdomen: Tender diffusely, RUQ most tender, No guarding or rebound, No HSM with normal BS Neuro: CN II-XII intact with motor/sensation intact throughout. Alert/oriented X 3 MS: Pain on passive extension/flexion of hip. Negative hip roll. No effusions or tenderness of joints. 3/5 strength of b/l lower extremities , 5/5 strength of upper extremities

Labs Remarkable for n n n Creatinine of 1. 13 T/D. bilirubin of 7. 4/5. 8, AST of 164, ALT of 140 and CBC with Hgb / Hct of 9. 8/29. 6 and Platelets of 28 Slightly abnormal coagulapathy with INR of 1. 2, D-dimer of 35. 2, PT of 12. 3, PTT of 38. 7 Low haptoglobulin Negative urine dip and normal VBG Normal: Hgb electrophoresis, G-6 PD study

PICU Course n Day 5 -8 n n Continue on atovaquine-proguanil with improvement in fever curve after 24 hours Anemia persisted but all other labs improved Blood smears collected every 12 hours, parasite density fell from 13% to 4% in 24 hours and less than 1% prior to floor transfer Day 8 -12 n On the floor improving symptoms except on Day of illness 11 n n Double vision and headache reoccurred with dizziness CT of head negative Transfused with PRBCS for Hgb of 5. 8 with resolution of symptoms D/C home after 3 negative smears, Course of 3 days of oral anti -malarial and improved fever

Labs Remarkable for n n n Creatinine of 1. 13 T/D. bilirubin of 7. 4/5. 8, AST of 164, ALT of 140 and CBC with Hgb / Hct of 9. 8/29. 6 and Platelets of 28 Slightly abnormal coagulapathy with INR of 1. 2, D-dimer of 35. 2, PT of 12. 3, PTT of 38. 7 Low haptoglobulin Negative urine dip and normal VBG Normal: Hgb electrophoresis, G-6 PD study
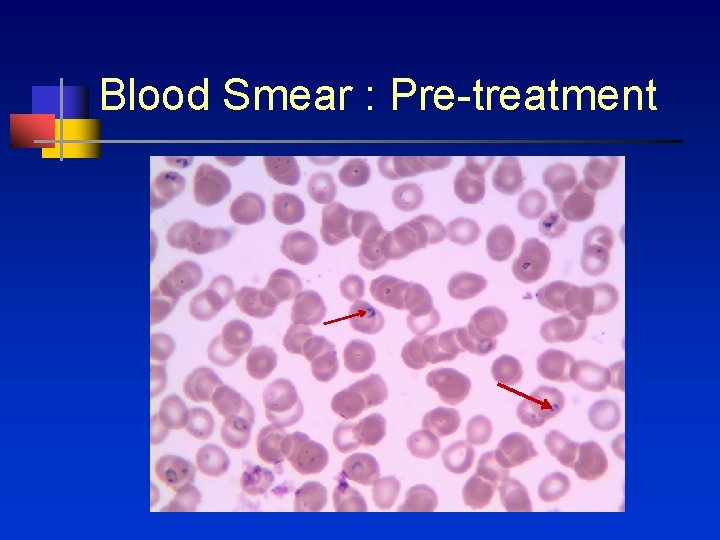
Blood Smear : Pre-treatment
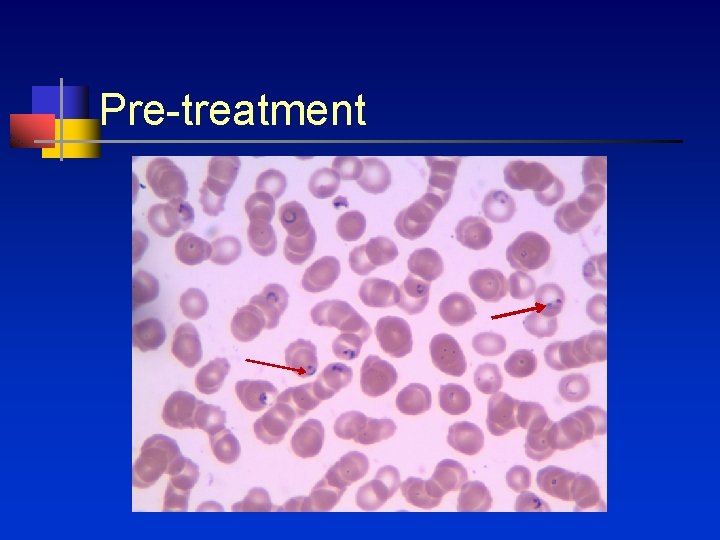
Pre-treatment
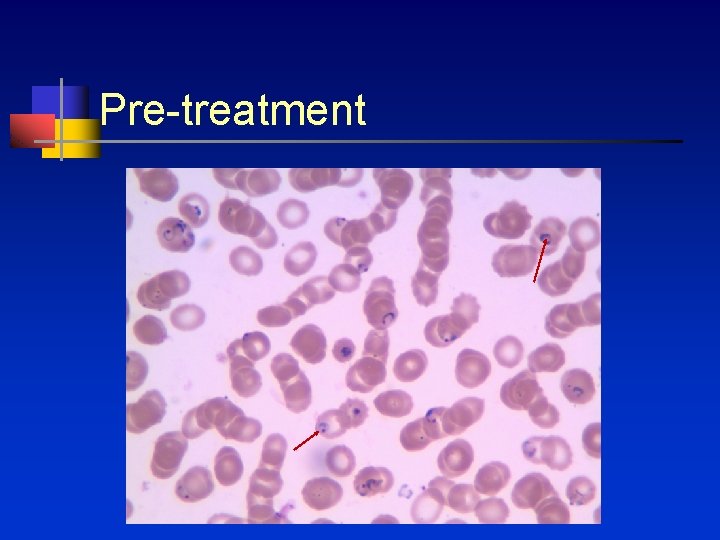
Pre-treatment
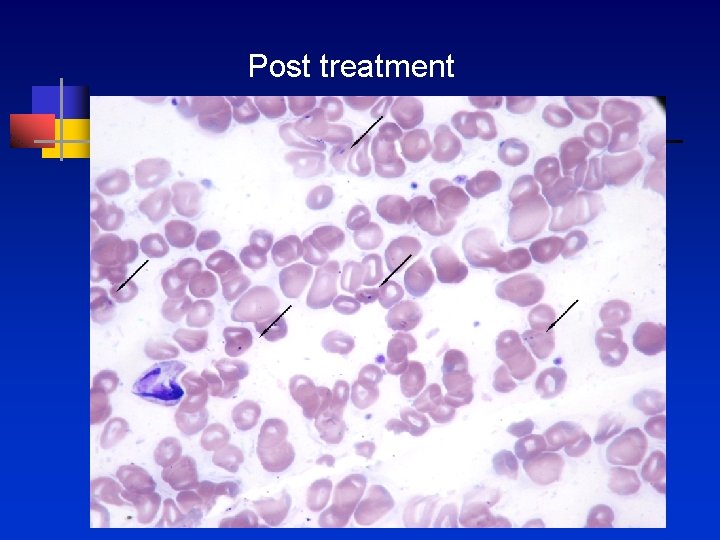
Post treatment
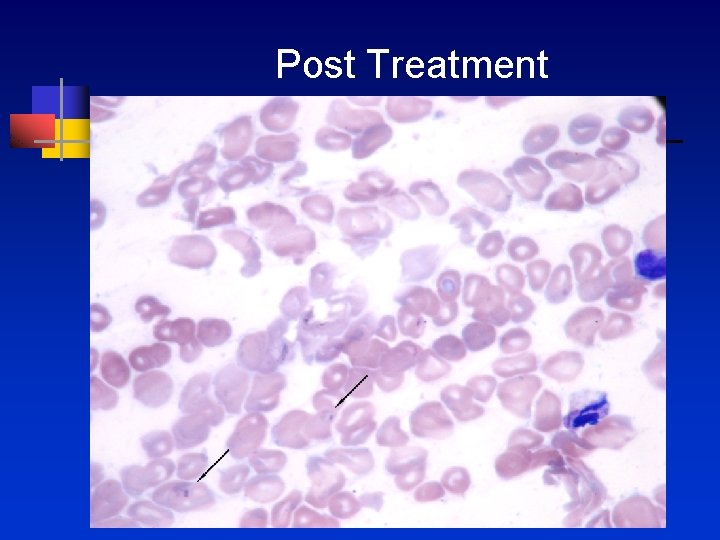
Post Treatment

Objectives n n n Discuss Malaria and its Presentation Review Differential Diagnosis of Febrile Traveler Study CDC Guidelines for chemoprophylaxis of malaria Present CDC Treatment Guidelines Investigate recent studies on Treatment of Severe Anemia caused by malaria

Definition n Parasitic infection with Plasmodium protozoa Transmitted by vector female Anopheles mosquito 4 species to cause infection in humans n n n P. falciparum P. vivax P. ovale P. malariae Plasmodium knowlesis recently identified to cause human infection

Epidemiology n n n 350 -500 million cases worldwide Predominates in tropical areas Over 1 million people die n n Most young children in Sub-Saharan Africa Account 20% of childhood deaths in Africa Every 30 seconds a child dies from malaria 1200 malaria cases reported annually

Epidemiology
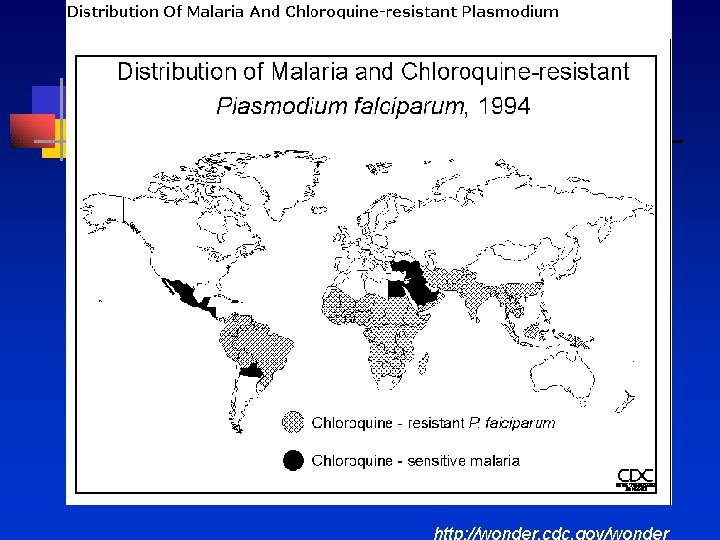
http: //wonder. cdc. gov/wonder

Sources of Infection in U. S n Imported n n Airport Malaria n n n Mosquitoes fly from endemic to non-endemic area and infect local residents Locally transmitted n n Majority of cases h/o of outbreaks in Southeast Congenital Blood Transfusions n n One case every 2 years 1 case per 4 million units of blood

Sources of Infection in U. S n 1997 to 2006 -10, 745 cases of malaria reported in the U. S n 59. 3% -sub-Saharan Africa n 13. 9% -Asia n 13. 3%-Caribbean and Central/South America n 0. 03% -Oceania n 54 fatal cases reported in the U. S n 85. 2% caused by P. falciparum n 71. 1% from sub-Saharan Africa
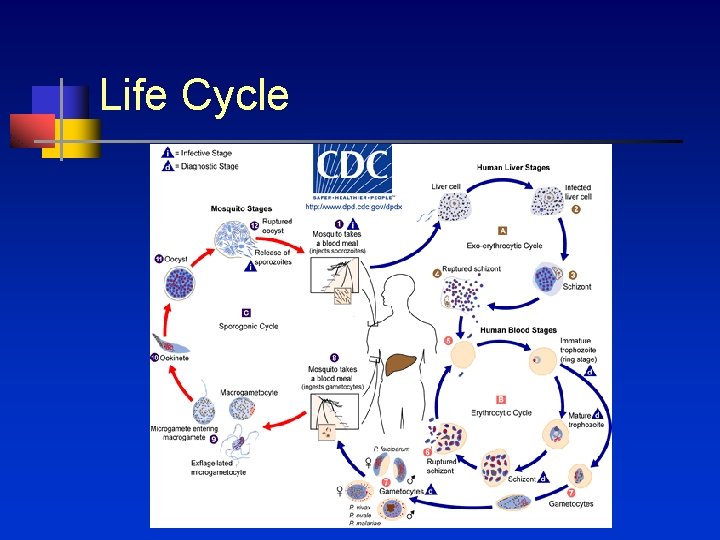
Life Cycle

Clinical Presentation n Symptoms present as early as 7 -14 days or as late as several months or longer after exposure Uncomplicated n Fever, Anemia, influenza-like symptoms, jaundice, transient HA, myalgias Severe n >5% parasite load n Mental confusion, seizures, kidney failure, acute respiratory distress syndrome, coma, death

Differential To Consider n n n Typhoid Fever Dengue Filarians Leishmanians Onchoncerciasis African trypanosomiasis

Diagnosis n Smear Microscopy gold standard n n n Rapid Diagnostic Test (RDT) n n n Thick: Identify presence of parasite Thin: Determine speciation/Parasite level FDA approved for hospital use Results in 2 -15 minutes PCR n n n Not FDA approved More sensitive than microscopy/delay in results Confirm species of parasite

CDC Chemoprophylaxis Guidelines n Areas with Limited Malaria Transmission n n Mosquito avoidance/dusk to dawn DEET repellant Insecticide covered Bed Tents Areas with Mainly P. vivax Malaria n Primaquine n n Areas with Chloroquine-Sensitive Malaria n n If Pts not G-6 PD-deficient Chloroquine Areas with Chloroquine-Resistant Malaria n n n Atovaquone/proguanil Doxycycline Mefloquine

CDC Chemoprophylaxis Guidelines n Areas with Mefloquine-Resistant Malaria n n n Atovaquone/proguanil Doxycyline Infants, Children and Adolescents n Chloroquine/mefloquine n n Doxycycline n n All weights/all ages 8 years or older Atovaquone/proguanil n n Not in infants less than 5 kg Off-label less than 11 kg

CDC Chemoprophylaxis Guidelines n Pregnancy and Breastfeeding n Chloroquine/Hydroxychloroquine n n Mefloquine n n n Limited data in 1 st trimester/safe in 2 nd and 3 rd Chloroquine resistent areas Atovaquone/proguanil n n Not been shown to have harmful effects Insufficient data Primaquine n Never use in pregnancy

CDC Chemoprophylaxis Guidelines n Atovaquone/Proguanil n n n Chloroquine and Hydroxychloroquine n n n 1 -2 days PTT, Same time daily in area, 4 wks daily post Mefloquine n n “Aralen”/“Plaquenil” 1 -2 wks PTT, Same day weekly, 4 wks post Doxycycline n n “Malarone” 1 -2 days PTT, Same day daily in area, 7 days post 1 -2 wks PTT, Same day weekly, 4 wks post Primaquine n n Primary: 1 -2 days PTT, Same time daily in area, 7 days post Anti-relapse : 14 days post

Tips for Primary Doctor n n Don’t hesitate to refer patient to local travel clinics prior to travel Preventative Measures depend on variety of factors n n Destination of country, Season of year, age of patient/underlying health conditions, itinerary of traveler Travel to malaria-endemic areas should be a part of patients chart Highest risk of infection are 1 st/2 nd generation of nonendemic visiting friends/family in endemic country Highest risk of severe infection are non-exposed individuals, infants/children, pregnant women

CDC Treatment Guidelines Clinical Diagnosis Region of Infection Adult drug Pediatric Drug Uncomplicated P. falciparum Chloroquineresistant or unknown resistance Atovaquoneproguanil Artemetherlumefantrine Quinine sulfate+ Clindamycine, Doxycycline or Tetracylcine Quinine sulfate +Clindamycin, Doxycycline or Tetracycline Mefloquine
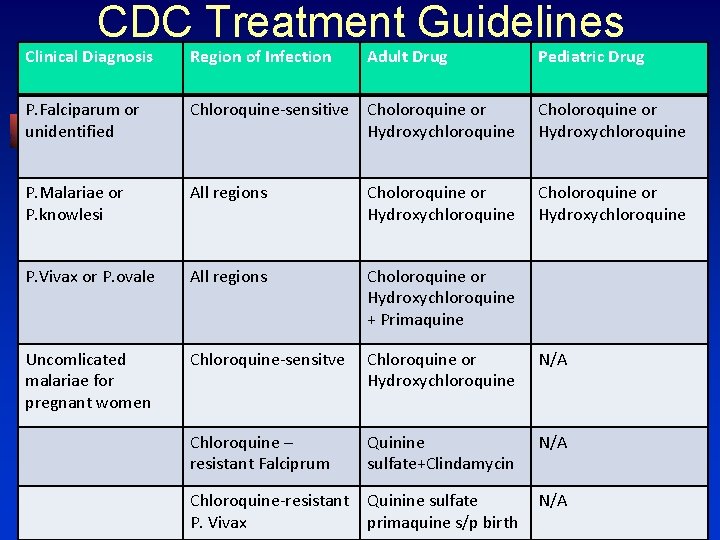
CDC Treatment Guidelines Clinical Diagnosis Region of Infection Adult Drug Pediatric Drug P. Falciparum or unidentified Chloroquine-sensitive Choloroquine or Hydroxychloroquine P. Malariae or P. knowlesi All regions Choloroquine or Hydroxychloroquine P. Vivax or P. ovale All regions Choloroquine or Hydroxychloroquine + Primaquine Uncomlicated malariae for pregnant women Chloroquine-sensitve Chloroquine or Hydroxychloroquine N/A Chloroquine – resistant Falciprum Quinine sulfate+Clindamycin N/A Chloroquine-resistant P. Vivax Quinine sulfate primaquine s/p birth N/A

CDC Treatment Guidelines Clinical Diagnosis Region of Infection Adult drug Pediatric Drug P. vivax Chloroquineresistant Quinine+Doxyclin e or Tetracycline +Primaquine Atovaquineproguanil +Primaquine Mefloquine+ Primaquine
Severe Malaria Treatment Quinine/Quinidine Artesunate/Artemisinins

EBM n Artesunate versus quinine for treating severe malaria n n Cochrane Database Syst Rev. 2007 Six trials enrolling 1938 participants n n n 1664 adults and 274 children Compared IV artesnuate vs. IV quinine for treatment of severe malaria Treatment with artesunate significantly reduced risk of death, reduced parasite clearance and incidence of hypoglycemia

References • • • www. cdc. gov/malaria/about/biology/index. html http: //wwwnc. cdc. gov/travel/yellowbook/2010/chapter 2/malaria. aspx Rosenthal P. J. Artesunate for the Treatment of Severe Falciprum Malaria. NEJM 2008: 358: 1829 -36. Clinical Review: Evidence behind the WHO Guidelines: Hospital Care for Children: Efficacy and Safety of Artemisinin Derivatives in Children with Malaria. Journal of Tropical Pediatrics 2006. 52: 1 -2. Malaria. The Red Book 2009. 1: 438. Griffith et al. Treatment of Malaria in the United States: A systematic Review. JAMA 2007. 297: 20.
- Slides: 62