PEARL II Study ombitasvirparitaprevirritonavir dasabuvir ribavirin for HCV
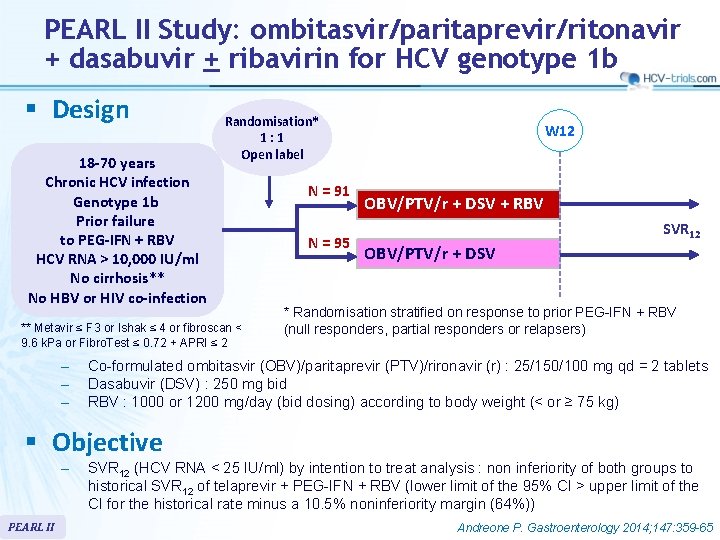
PEARL II Study: ombitasvir/paritaprevir/ritonavir + dasabuvir + ribavirin for HCV genotype 1 b § Design 18 -70 years Chronic HCV infection Genotype 1 b Prior failure to PEG-IFN + RBV HCV RNA > 10, 000 IU/ml No cirrhosis** No HBV or HIV co-infection Randomisation* 1: 1 Open label ** Metavir ≤ F 3 or Ishak ≤ 4 or fibroscan < 9. 6 k. Pa or Fibro. Test ≤ 0. 72 + APRI ≤ 2 – – – N = 91 N = 95 W 12 OBV/PTV/r + DSV + RBV SVR 12 OBV/PTV/r + DSV * Randomisation stratified on response to prior PEG-IFN + RBV (null responders, partial responders or relapsers) Co-formulated ombitasvir (OBV)/paritaprevir (PTV)/rironavir (r) : 25/150/100 mg qd = 2 tablets Dasabuvir (DSV) : 250 mg bid RBV : 1000 or 1200 mg/day (bid dosing) according to body weight (< or ≥ 75 kg) § Objective – PEARL II SVR 12 (HCV RNA < 25 IU/ml) by intention to treat analysis : non inferiority of both groups to historical SVR 12 of telaprevir + PEG-IFN + RBV (lower limit of the 95% CI > upper limit of the CI for the historical rate minus a 10. 5% noninferiority margin (64%)) Andreone P. Gastroenterology 2014; 147: 359 -65
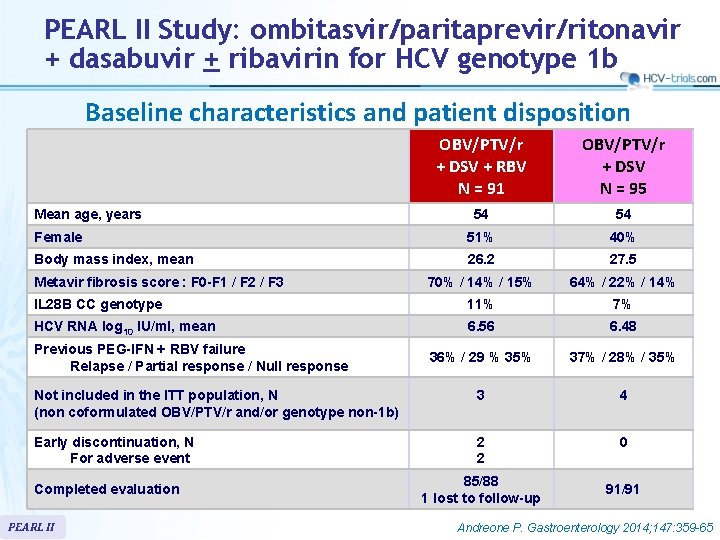
PEARL II Study: ombitasvir/paritaprevir/ritonavir + dasabuvir + ribavirin for HCV genotype 1 b Baseline characteristics and patient disposition OBV/PTV/r + DSV + RBV N = 91 OBV/PTV/r + DSV N = 95 54 54 Female 51% 40% Body mass index, mean 26. 2 27. 5 70% / 14% / 15% 64% / 22% / 14% IL 28 B CC genotype 11% 7% HCV RNA log 10 IU/ml, mean 6. 56 6. 48 36% / 29 % 35% 37% / 28% / 35% Not included in the ITT population, N (non coformulated OBV/PTV/r and/or genotype non-1 b) 3 4 Early discontinuation, N For adverse event 2 2 0 Mean age, years Metavir fibrosis score : F 0 -F 1 / F 2 / F 3 Previous PEG-IFN + RBV failure Relapse / Partial response / Null response Completed evaluation PEARL II 85/88 1 lost to follow-up 91/91 Andreone P. Gastroenterology 2014; 147: 359 -65
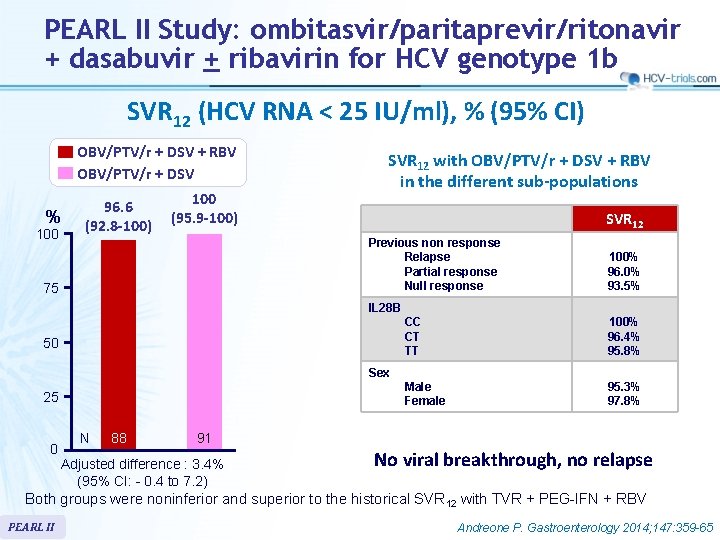
PEARL II Study: ombitasvir/paritaprevir/ritonavir + dasabuvir + ribavirin for HCV genotype 1 b SVR 12 (HCV RNA < 25 IU/ml), % (95% CI) OBV/PTV/r + DSV + RBV OBV/PTV/r + DSV % 100 96. 6 (92. 8 -100) 100 (95. 9 -100) SVR 12 with OBV/PTV/r + DSV + RBV in the different sub-populations SVR 12 Previous non response Relapse Partial response Null response 75 100% 96. 0% 93. 5% IL 28 B 50 CC CT TT 100% 96. 4% 95. 8% Male Female 95. 3% 97. 8% Sex 25 0 N 88 91 Adjusted difference : 3. 4% (95% CI: - 0. 4 to 7. 2) No viral breakthrough, no relapse Both groups were noninferior and superior to the historical SVR 12 with TVR + PEG-IFN + RBV PEARL II Andreone P. Gastroenterology 2014; 147: 359 -65
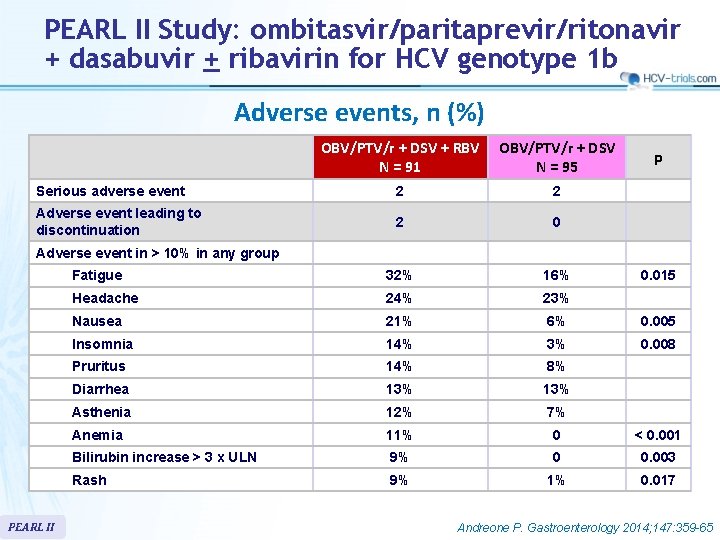
PEARL II Study: ombitasvir/paritaprevir/ritonavir + dasabuvir + ribavirin for HCV genotype 1 b Adverse events, n (%) OBV/PTV/r + DSV + RBV N = 91 OBV/PTV/r + DSV N = 95 Serious adverse event 2 2 Adverse event leading to discontinuation 2 0 Fatigue 32% 16% Headache 24% 23% Nausea 21% 6% 0. 005 Insomnia 14% 3% 0. 008 Pruritus 14% 8% Diarrhea 13% Asthenia 12% 7% Anemia 11% 0 < 0. 001 Bilirubin increase > 3 x ULN 9% 0 0. 003 Rash 9% 1% 0. 017 p Adverse event in > 10% in any group PEARL II 0. 015 Andreone P. Gastroenterology 2014; 147: 359 -65

PEARL II Study: ombitasvir/paritaprevir/ritonavir + dasabuvir + ribavirin for HCV genotype 1 b § Summary – The ITT SVR 12 rates in genotype 1 b HCV infected patients receiving the 12 -week regimen of OBV/PTV/r + DSV with or without RBV (96. 6%– 100%, respectively) were superior to the historical rate of telaprevir + PEG-IFN + RBV • No benefit of addition of RBV, with significantly more adverse events, bilirubin increase and anemia – Adverse events in both groups of 12 -week regimens were generally mild and manageable. – Overall, only 2 (1. 1%) patients discontinued treatment because of adverse events, and the 5 serious adverse events reported in 4 patients were considered to be unrelated to study drug – Limitations • open-label study design • exclusion of patients with cirrhosis, HBV or HIV co-infection PEARL II Andreone P. Gastroenterology 2014; 147: 359 -65
- Slides: 5