PDT 152 MATERIALS CHEMISTRY Course Outlines 3 number

PDT 152 MATERIALS CHEMISTRY Course Outlines (3 number of unit/Core course) Prepared by: WAN MASTURA BINTI WAN IBRAHIM Department of Mechanical Engineering Technology Faculty of Engineering Technology Universiti Malaysia Perlis (Uni. MAP)

Course synopsis • PROVIDES an introduction to basic concepts and methods of materials chemistry and a broad overview of how chemistry is applied in development and production of advanced materials such as ceramics, polymers and metals. • COVERS basics principles and methods for synthesis of materials, nanomaterials, biomaterials and environmental application of materials. • • GIVES knowledge on the reaction kinetics such as effect of reactants and products concentration, effect of temperature, determination of order and velocity constant of reaction, electrochemistry and interface phenomenon.

Course outcomes C 01 IDENTIFY • Able to IDENTIFY the basic solid state chemistry and how structure properties relationships are important in materials chemistry. C 02 CLASSIFY • Able to CLASSIFY the inorganic materials, nano-material, synthetic strategies, physical bonding and properties (optical, electronic, conduction) in solid, and environmental application. C 03 DEMONSTRATE • Able to DEMONSTRATE the effect of reactant and products concentration, effect of temperature and diffusion in solid state. C 04 ANALYZE • Able to ANALYZE electrochemistry, surface energy, interfacial energy expect gas/liquid interfacial, absorption and colloid.

Course content • OVERVIEW AND INTRODUCTION Issues of design and bonding. Explain the basic bonding (Valence bond/molecular orbital) • SOLID STATE CHEMISTRY Describe the metal, ceramic and polymers. Explain the crystalline structure, amorphous state and symmetric. Identify the polymer physics and application. Explain the polymer additive. • NANOMATERIALS (NANOCHEMISTRY) Describe the synthesis, dimensionality, growth mechanism and nucleation process • BIOMATERIALS Explain and Identify the bio inspiration and toxicology. Describe the environmental application of materials. • SEMICONDUCTORS AND APPLICATIONS Describe and Identify thermoelectric, electronics and optics properties. • CHEMICAL KINETICS Explain and illustrate concepts of reactant and product concentration in the kinetic. Describe the effect of temperature, pressure and concentration in the kinetic of materials. Determine the order and velocity constant of reaction. • ELECTROCHEMISTRY & INTERFACE PHENOMENA Identify and calculate the electrochemistry, surface energy and interfacial energy expect gas/liquid interface • ADSORPTION Explain the adsorption theoretical and adsorbent, identify and interpret and illustrate concepts of the BET and Langmuir Analysis.

List of experiments 1. Determination of palm oil viscosity using viscometer 2. Determination of waste engine oil viscosity using viscometer 3. Determination of waste cooking oil viscosity using viscometer 4. Preparation of poly(vinyl chloride)/potash feldspar filler films 5. Preparation of alcohol solubility 6. Synthesis of conductive material from epoxy/tyre dust conductive polymer 7. Preparation of Cu. O/Zn. O alloy 8. Synthesis of conductive material from expoxy/graphite-Cu 2 O conductive composites
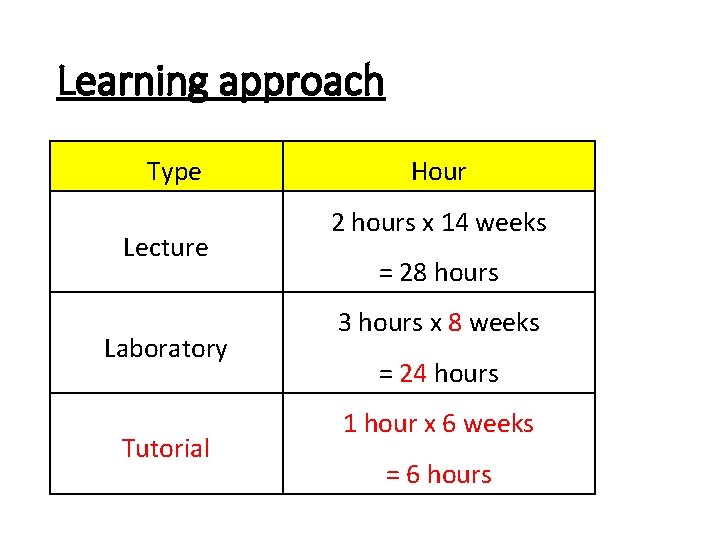
Learning approach Type Lecture Laboratory Tutorial Hour 2 hours x 14 weeks = 28 hours 3 hours x 8 weeks = 24 hours 1 hour x 6 weeks = 6 hours

Evaluation contribution Examination (60%) Continual Assessment (40%) Mid term examination (20%) Assignment (10%) Final examination (40%) Test (10%) Laboratory report (10%)

References 1. Lesley E. Smart and Elain A. Moore, 3 rd Edition, CRC press Taylor and Frances Group, 2005. 2. Silbey, Alberty and Bowendy, Physical Chemistry, Fourth Edition, John Wiley & Sons. Inc. USA, 2005. 3. R. C Srivastava, Subit K. Saha, Abhay K. Jain, Thermodynamics, Third Edition, Prentice. Hall of India, New Delhi, 2007. 4. Bradley D. Fahiman, Materials Chemistry, 2 nd Edition, Springer 2001. 5. Raymond C. & John W. T. (2014). Physical Chemistry for the Chemical Sciences. Publisher Royal Society of Chemistry. Publication City/Country Cambridge, United Kingdom. 6. Cengel Y. A. (2015). Thermodynamics : an engineering approach / Yunus A. Cengel, University of Nevada, Reno, Michael A. Boles, North Carolina State University. Eighth edition. 7. Peter A. (2015). Chemistry: A very short introduction. Oxford University Press. 8. Klaus F. , Gennady E. Z. & Haghi A. K. (2016). Materials Chemistry : A Multidisciplinary Approach to Innovative Methods. 07 Apr 2016. Publisher Apple Academic Press Inc. Publication City/Country Oakville, Canada.
- Slides: 8