PDP Chemistry Acids and bases Thermochemistry energy changes

PDP Chemistry Acids and bases Thermochemistry: energy changes in chemical reactions

Topic 6: Acids and bases What is an acid Reactions of acids Reactions of bases p. H scale Indicators
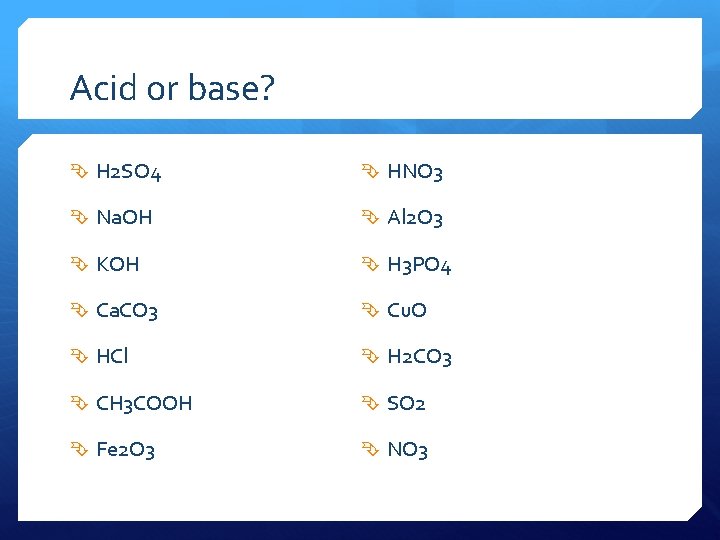
Acid or base? H 2 SO 4 HNO 3 Na. OH Al 2 O 3 KOH H 3 PO 4 Ca. CO 3 Cu. O HCl H 2 CO 3 CH 3 COOH SO 2 Fe 2 O 3 NO 3

Concentration review Calculate the concentration of sodium chloride in water if 6 grams are dissolved in 50 m. L a. in g/L b. in mol/dm 3 State, without calculating, the concentrations if 3 grams are dissolved in 25 m. L

powers and logs Find a. 103 b. 106 c. 10 -1 d. 10 -4 Find a. log (0. 1) b. log (0. 001) c. log (1000) d. log (1 000)

Acids

What is an acid?

Arrhenius definition: acids give H+ ions in aqueous solution H 2 SO 4(l) 2 H+(aq) + SO 42 -(aq)

Indicators

Indicators Image: http: //commons. wikimedia. org/wiki/File: Indicateurs_color%C 3%A 9 s_de_p. H. jpg
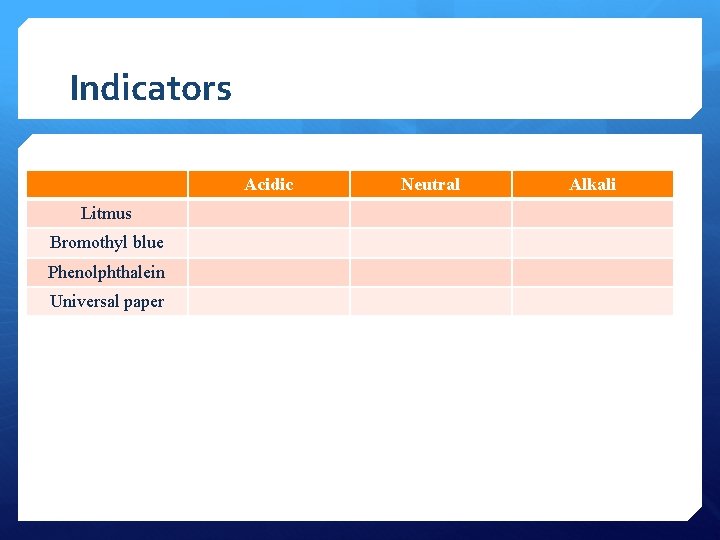
Indicators Acidic Litmus Bromothyl blue Phenolphthalein Universal paper Neutral Alkali

p. H scale
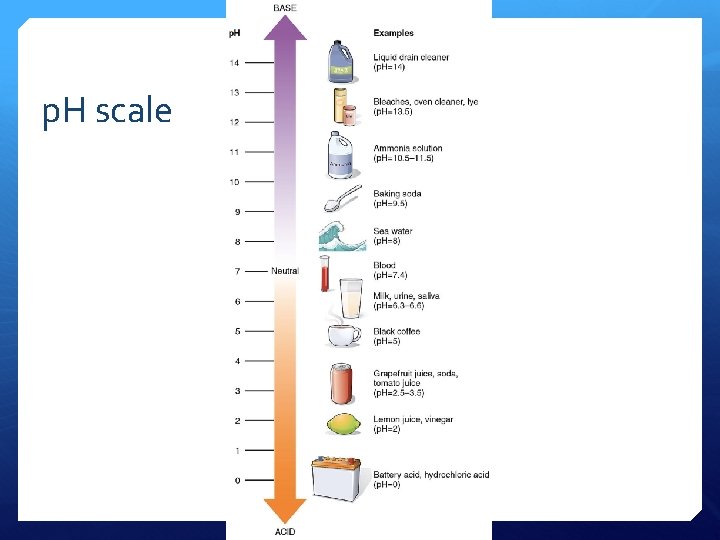
p. H scale

p. H scale measures H+ ion concentration p. H = -log 10 + [H ] p. H measured using: • indicators • p. H meter
![Which of the following solutions are acidic? [H+] = 10 -7 M [H+] = Which of the following solutions are acidic? [H+] = 10 -7 M [H+] =](http://slidetodoc.com/presentation_image/18125f66b221b9204972e60908a1afcd/image-15.jpg)
Which of the following solutions are acidic? [H+] = 10 -7 M [H+] = 10 -3 M [H+] = 10 -14 M

p. H calculations 1. Calculate the p. H of HCl with a concentration of: a) 0. 1 M b) 0. 01 M 2. Calculate the concentration of a strong acid of p. H 2. 3. Calculate the concentration of a diluted acid of p. H 5.

Reactions of acids

Reactions of acids with carbonates and hydrogen carbonates Reactions of acids with metals above hydrogen in the reactivity series Displacement of ammonia from ammonia salts by bases Reaction of acids and bases with indicators

Acid + metal

Acid + base
Acid + alkali

Acid + metal carbonate

Why use burettes and pipettes? Calculate the percentage uncertainty in each of the following 1. 10 m. L a measuring cylinder uncertainty ± 0. 2 m. L 2. 10 m. L burette uncertainty ± 0. 1 m. L 3. 10 m. L a pipette uncertainty ± 0. 02 m. L 4. 10 m. Lmeasured in a 50 m. L beaker ± 5 m. L 5. 10 m. L measured using a balance 10. 00 g ± 0. 01 g

Strong and weak acids

How strong can an acid get? concentrated ≠ dilute strong ≠ weak “Alien blood is an extremely potent acid and is capable of corroding on contact almost any substance with alarming speed. ” Alien (creature in Alien franchise). (2013, April 28). In Wikipedia, The Free Encyclopedia. Retrieved 19: 56, May 12, 2013, from http: //en. wikipedia. org/w/index. php? title=Alien_( creature_in_Alien_franchise)&oldid=552537824 corrosive

Strong acids: Totally dissociated Hydrochloric acid: HCl Cl- + H+ chloride Nitric acid: HNO 3 NO 3 - + H+ nitrate Sulphuric acid: H 2 SO 4 SO 42 - + H+ sulphate

HCl H+ +Cl. Start 100% End 0% 0% 100%

Weak acids Partially dissociated Ethanoic acid, (Acetic acid) CH 3 COOH CH 3 COO- + H+ ethanoate ion (acetate ion) Carbonic acid, H 2 CO 3 CO 32 - + 2 H+ carbonate ion

CH 3 COOH H+ + CH 3 COOstart 100% 0% end 99% 1%

Bases

Strong bases Contain the OH- ion All group I hydroxides: Na. OH(s) Na+ + OH- Group II hydroxides Ba(OH)2 Ba 2+ + 2 OH-

Weak bases Partially dissociated Ammonia NH 3 + H 2 O NH 4+ + OH-

Neutralization

Neutralization H+(aq) + OH-(aq) H 2 O(l) ΔH°=-57 k. Jmol-1

Neutralisation Acid + base salt + water HCl + Na. OH Na. Cl + H 2 O
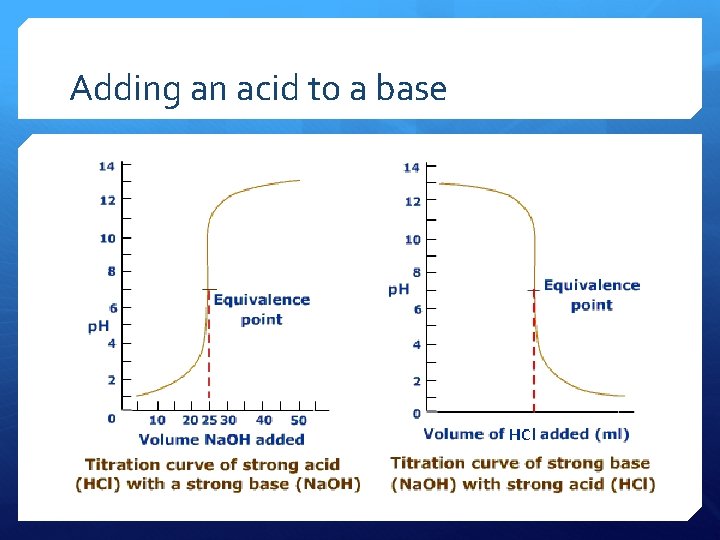
Adding an acid to a base

Titration

Acid-base titrations use a known concentration of an base to find the concentration of an acid Measure the volume of acid that neutralizes the base. Calculate concentration from the equivalence point as the number of moles of acid and base is equal

Apparatus burette pipette retort stand pipette filler conical flask
Image: http: //en. wikipedia. org/wiki/File: Periodic_table. svg

Links http: //phet. colorado. edu/en/simulation/acid-base-solutions Khan academy videos about acids and bases https: //www. khanacademy. org/science/chemistry/acids-andbases
- Slides: 41