PCR Polymerase Chain Reaction Troubleshooting PCR Troubleshooting Is

PCR Polymerase Chain Reaction Troubleshooting

PCR Troubleshooting: Is a collection of techniques that alter PCR reaction in order to achieve optimum PCR results.

There is no single set of conditions that is optimal for all PCR. Therefore, each PCR is likely to require specific optimization for the template/primer pairs chosen. Lack of optimization often results in problems, such as no detectable PCR product or low efficiency amplification of the chosen template; the presence of nonspecific bands or smeary background; the formation of “primer-dimers”

Optimization of Polymerase Chain Reactions Optimization of a particular PCR can be time consuming and complicated because of the various parameters that are involved. These parameters include the following: (1) quality and concentration of DNA template. (2) design and concentration of primers. (3) concentration of magnesium ions. (4) concentration of the four deoxynucleotides (d. NTPs). (5) PCR buffer systems. (6) selection and concentration of DNA polymerase. (7) PCR thermal cycling conditions. (8) addition and concentrations of PCR additives/cosolvents.

For each PCR, the following components are mixed together: 1. Template DNA (1– 500 ng). 2. Primers (0. 05– 1. 0 M). 3. Mg 2+ (0. 5– 5 m. M). 4. d. NTP (20– 200 M each). 5. 1× PCR buffer: 1 m. M Tris-HCl and 5 m. M KCL. 6. DNA polymerase (0. 5– 2. 5 U for each 50 L of PCR).
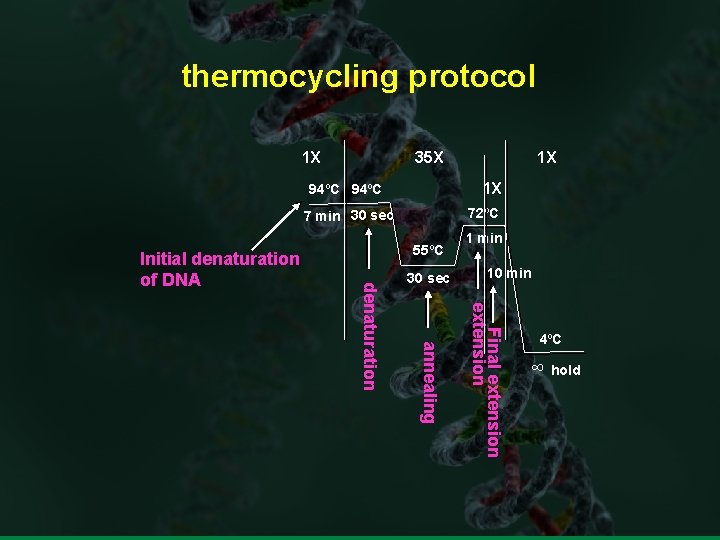
thermocycling protocol 1 X 35 X 1 X 94ºC 72ºC 7 min 30 sec 55ºC 30 sec 1 min 10 min Final extension annealing denaturation Initial denaturation of DNA 1 X 4ºC ∞ hold

Troubleshooting PCR Poor or no amplification of bands Problem with thermo cycler, set-up, reagents Run positive control Too little DNA template is present in the reaction Increase Amount of DNA The template DNA is damaged or degraded Assure the purity and integrity of the DNA Enzyme concentration low Concentration in increments of 0. 5 units per 100 L Annealing temp too high Lower annealing temperature in increments of 2°C. Extension time too short Increase Time for longer products Cycle number too low Increase cycle number by 5 to 10 cycles. .

Troubleshooting PCR Poor or no amplification of bands Primer design and composition. Design new primers Mg. Cl 2 concentration too low Increase Concentration in increments 0. 1 m. M. The denaturation time is too long or too short Adjust denaturation time in increments of 5 s. The denaturation temperature is too high or too low. Change denaturation temperature in increments of 1°C. Check for inhibitor(s) during template DNA preparation Re purification of the DNA template may remove some inhibitors of PCR.
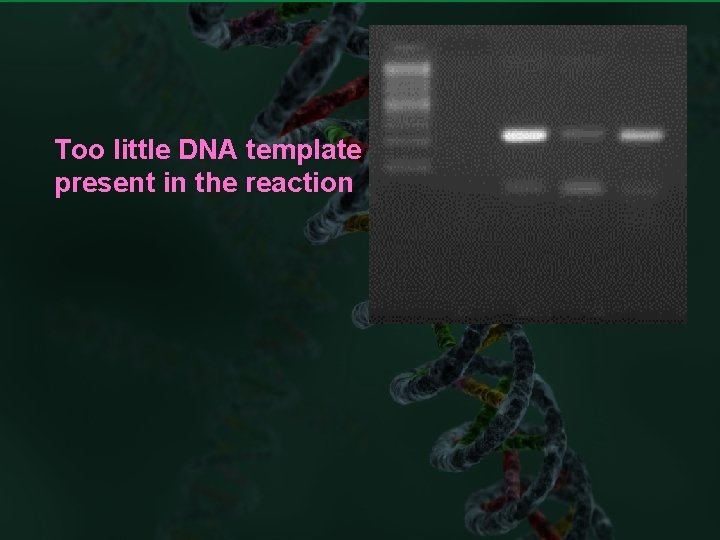
Too little DNA template is present in the reaction

The template DNA is damaged or degraded

Troubleshooting PCR multiple product bands(Non-specific bands on your gel) or smear is detected: Reagents, set-up Run negative control Too much DNA template is present in the reactions Decrease the amount of DNA template in the reaction mix. Annealing temperature is too low Increase annealing temperature in increments of 2°C. DNA polymerase concentration is too high Decrease enzyme concentration in increments of 0. 5 units per 100 - L reaction. Magnesium concentration is too high. Decrease the magnesium concentration in increments of 0. 1 m. M.

Troubleshooting PCR multiple product bands(Non-specific bands on your gel) or smear is detected: Denaturation time is too short Increase the denaturation time in increments of 5 s. Denaturation temperature is too low Increase the denaturation time in increments of 1°C. Cycle number is too high Reduce the cycle number by 5 to 10 cycle. Extension time is too long Reduce the extension time in increments of 1 min. Review primer design and composition Design new primers and try PCR again. Check for carry-over contamination.
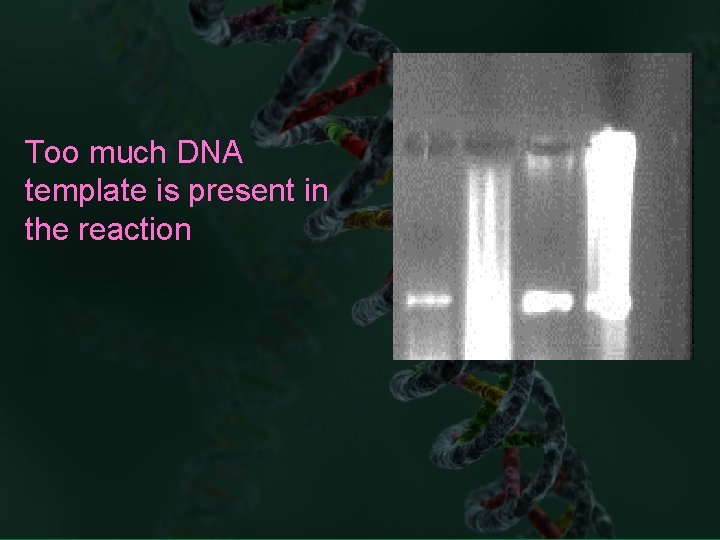
Too much DNA template is present in the reaction
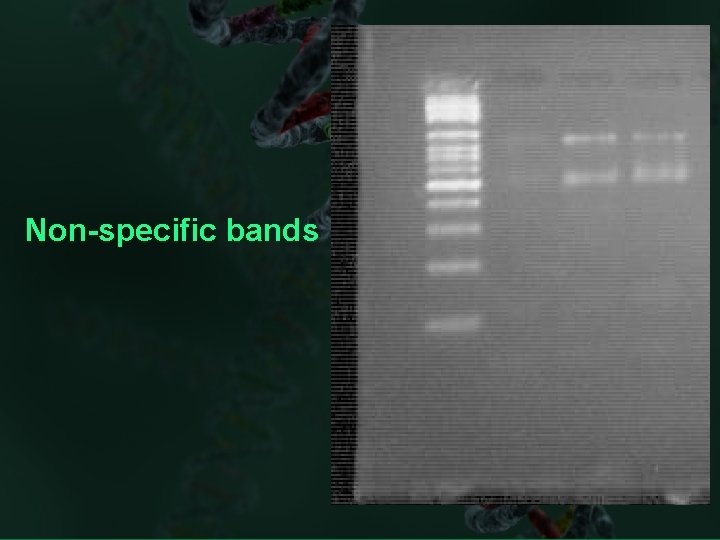
Non-specific bands

Mg. Cl 2 (m. M) 1. 5 2 3 4 Magnesium Chloride (Mg. Cl 2 - usually 0. 5 -5. 0 m. M) Magnesium ions have a variety of effects Mg 2+ acts as cofactor for Taq polymerase Required for Taq to function Excess magnesium results in accumulation of nonspecific amplification products seen as multiple bands on an agarose gel, whereas insufficient magnesium results in reduced yield of the desired PCR product. 5
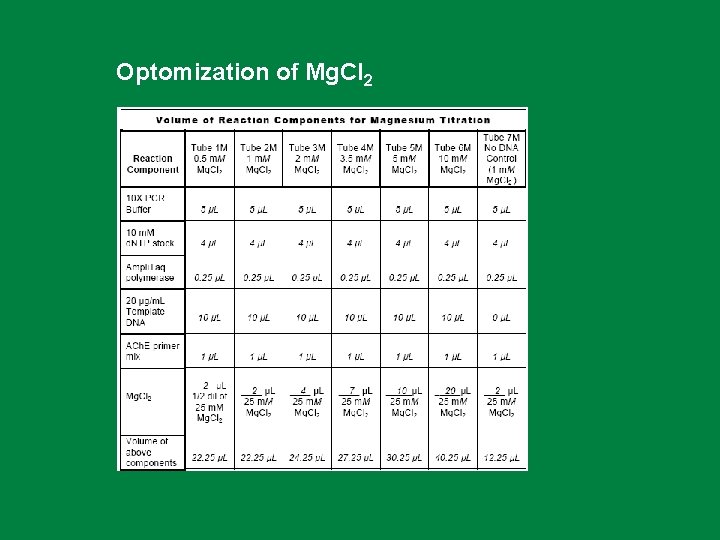
Optomization of Mg. Cl 2

Preparing a Master Mix. Remember that the volumes you have entered in Table 4 were all for each tube. That means you have to add 5 μL of PCR buffer to each of 7 tubes. Then add 4 μL of 10 m. M d. NTP stock to 7 tubes and so on. That is a lot of pipetting and each time you pipet you can introduce some error in the measurement. So we will use a way to minimize the pipetting. We will make a Master Mix. The Master Mix for this experiment should contain all the reagents needed for the PCR except for the one component being titrated (in this case, magnesium) and the template DNA (which should always be added last to a PCR). The Master Mix will also contain the smallest amount of water any of the 7 tubes will need. Once prepared, an aliquot of the Master Mix will be delivered to each reaction tube. Then, different amounts of Mg. Cl will be added to each tube, and additional water will be added to each reaction such that the final volume will be 50 μL. Template DNA will be added as the final step prior to thermal cycling 2

Primer Design 1. Typically 18 to 28 bases in length 2. Annealing temperature dependent upon primer sequence (~ 50% GC content) 3. Avoid secondary structure, particularly 3’ 4. Avoid primer complementarity (primer dimer) 5. The last 3 nucleotides at the 3` end is the substrate for DNA polymerase - G or C 6. Many good freeware programs available
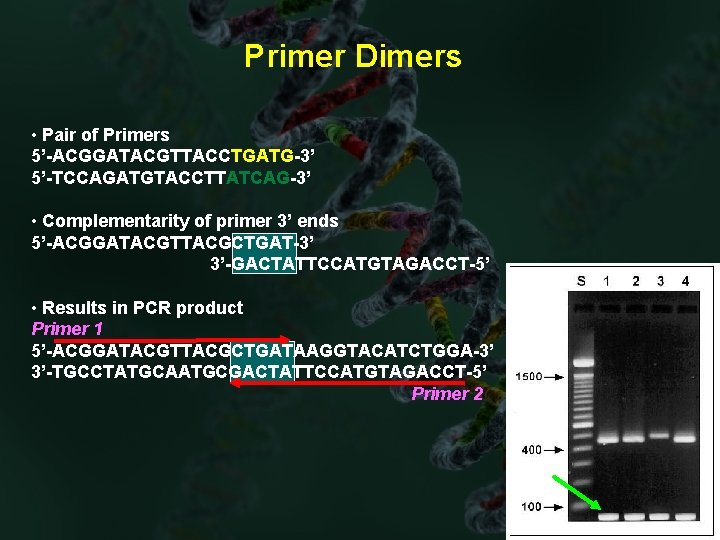
Primer Dimers • Pair of Primers 5’-ACGGATACGTTACCTGATG-3’ 5’-TCCAGATGTACCTTATCAG-3’ • Complementarity of primer 3’ ends 5’-ACGGATACGTTACGCTGAT-3’ 3’-GACTATTCCATGTAGACCT-5’ • Results in PCR product Primer 1 5’-ACGGATACGTTACGCTGATAAGGTACATCTGGA-3’ 3’-TGCCTATGCAATGCGACTATTCCATGTAGACCT-5’ Primer 2
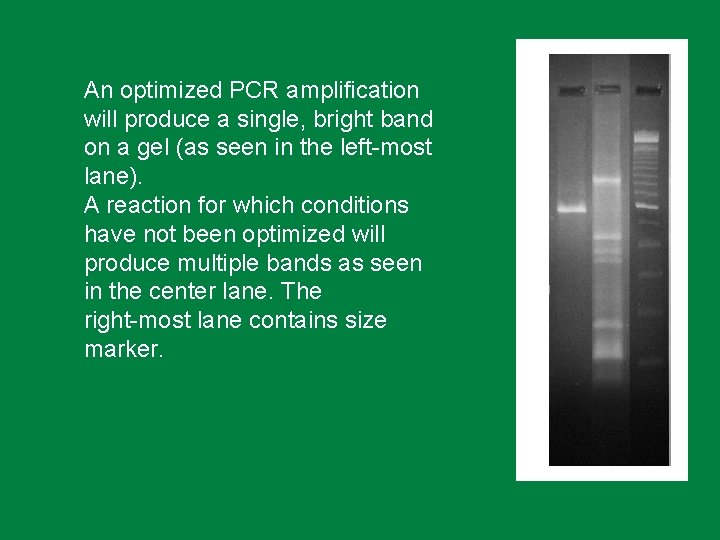
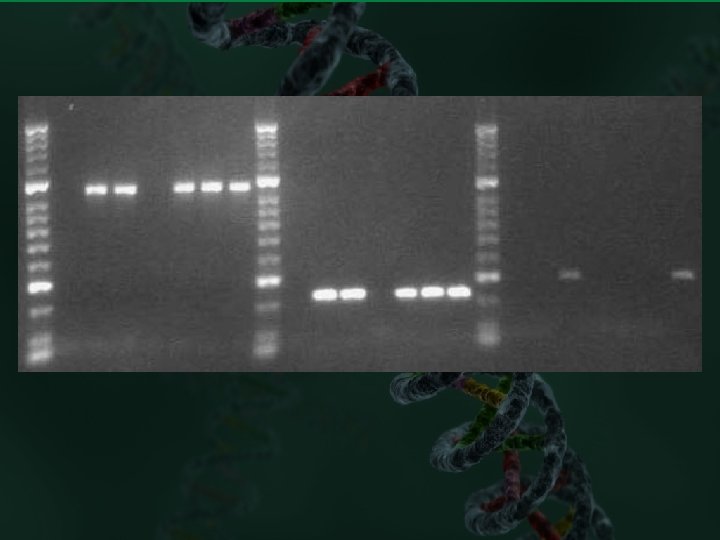
An optimized PCR amplification will produce a single, bright band on a gel (as seen in the left-most lane). A reaction for which conditions have not been optimized will produce multiple bands as seen in the center lane. The right-most lane contains size marker.

Questions?
- Slides: 23