PBS 803 Class 16 Blymphocyte biology Suggested Reading
PBS 803 - Class #16 B-lymphocyte biology Suggested Reading: Abbas pages 147 -168 Parham pages 9 -12, 9 -14 to 9 -15 The plasma cell, effector B cell that makes antibodies, a potent weapon of adaptive immunity. The marrow cavities of the bones, in which B-cell development occurs

Learning Objectives for Class #16 part I • Compare and contrast features of primary and secondary antibody responses • Discuss the mechanisms associated with activation of B cells through the BCR and innate immune signals • Describe the functional consequences of antigen receptor-mediated B cell activation (e. g. isotype switching, memory or plasma cell formation etc. ) • Describe the mechanism by which B lymphocytes present antigens to helper T cells • Describe the mechanism of helper T-cell mediated activation of B lymphocytes in the germinal center reaction

Learning Objectives for Class #16 part II • Describe the significance of somatic hypermutation and affinity maturation • Compare and contrast the effects of T-independent and T-dependent antigens on B cell activation • Diagram the process by which secretory Ig. A crosses the epithelium and explain the importance of Ig. A for the protection of mucosal surfaces • Describe the mechanisms of passive immunity to both a fetus and a newborn infant • Describe the neutralizing affect of antibodies in preventing diseases caused by viruses, bacteria and toxins
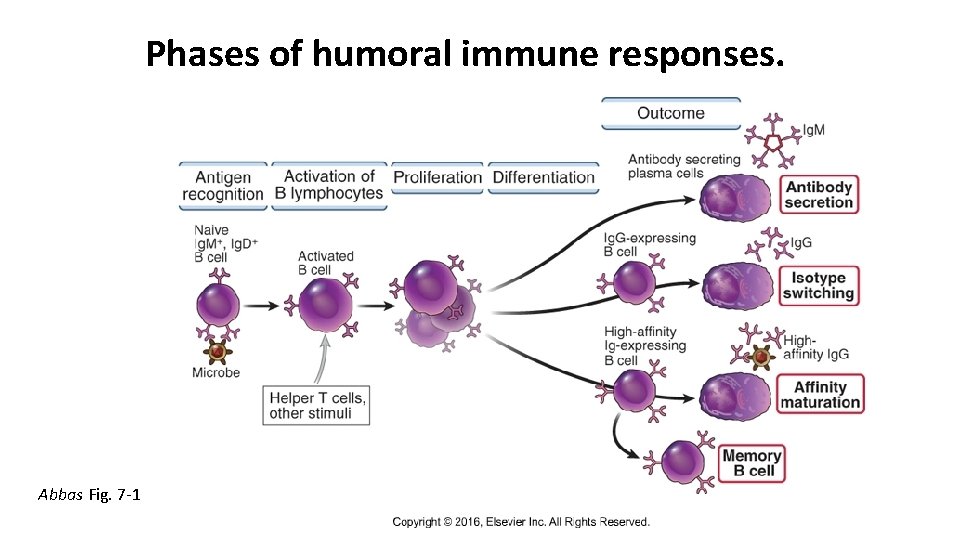
Phases of humoral immune responses. Abbas Fig. 7 -1
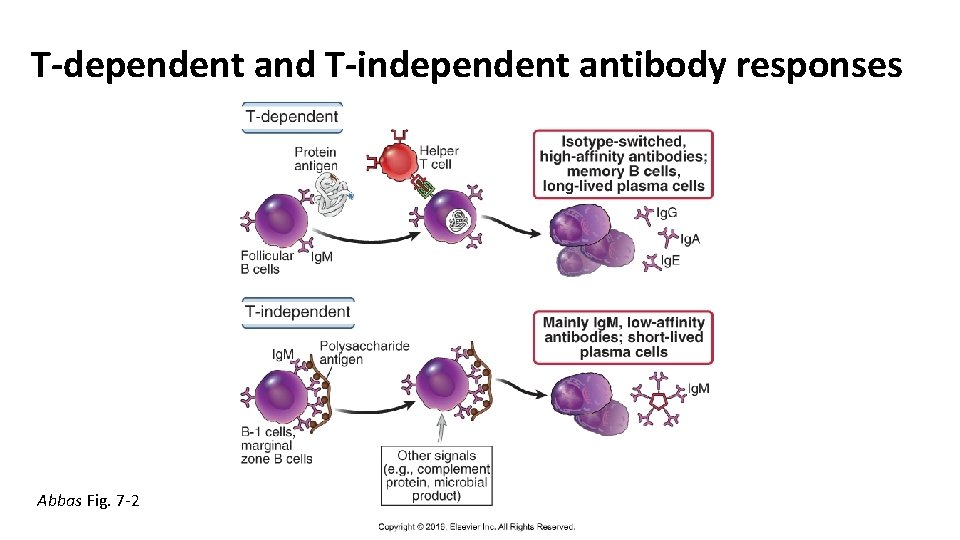
T-dependent and T-independent antibody responses Abbas Fig. 7 -2
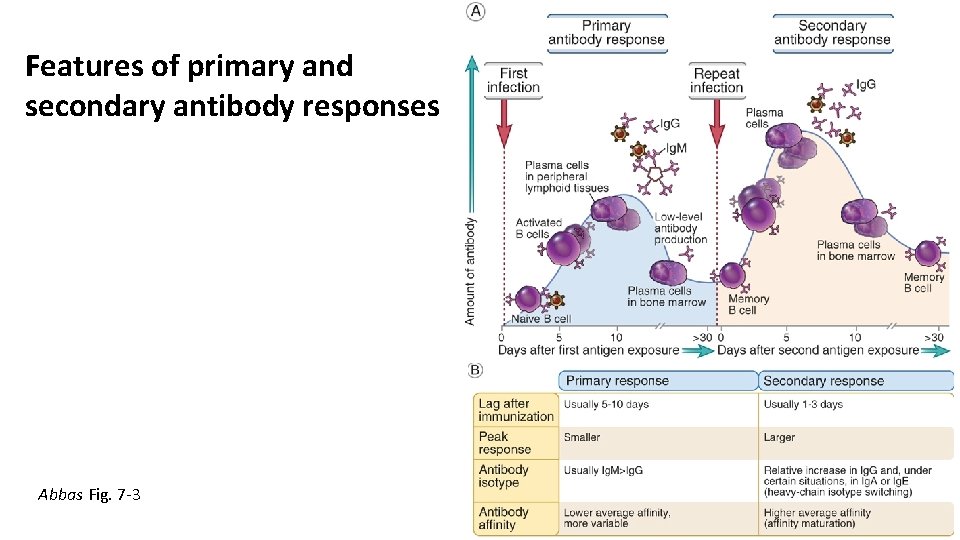
Features of primary and secondary antibody responses Abbas Fig. 7 -3
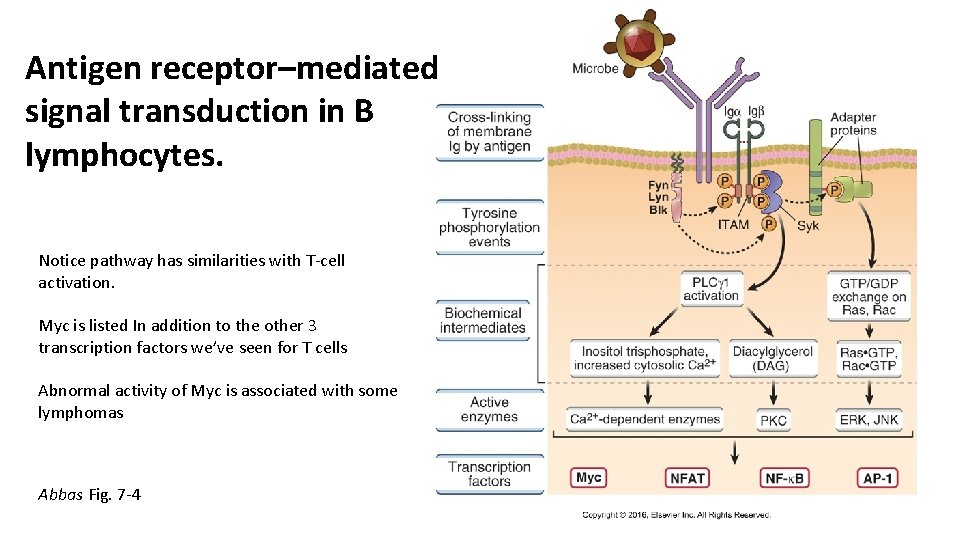
Antigen receptor–mediated signal transduction in B lymphocytes. Notice pathway has similarities with T-cell activation. Myc is listed In addition to the other 3 transcription factors we’ve seen for T cells Abnormal activity of Myc is associated with some lymphomas Abbas Fig. 7 -4
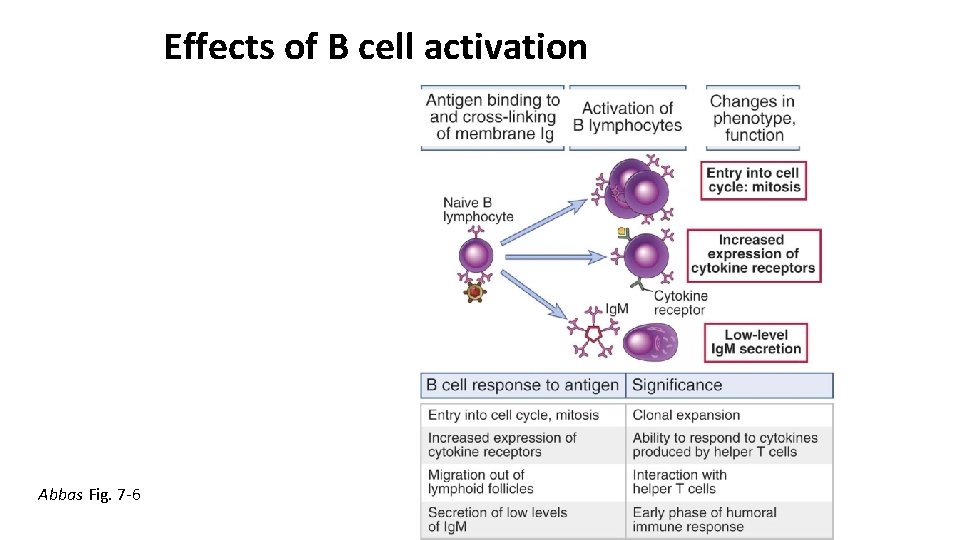
Effects of B cell activation Abbas Fig. 7 -6
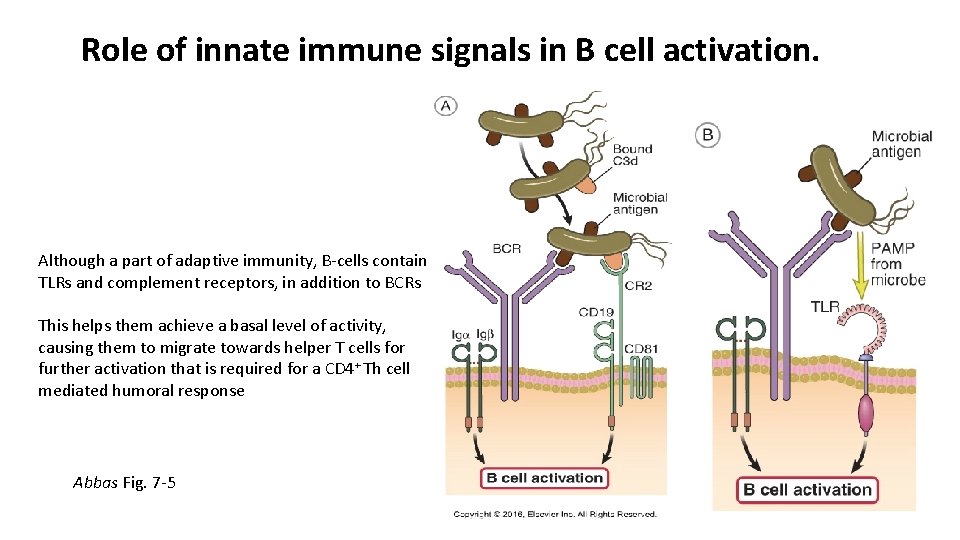
Role of innate immune signals in B cell activation. Although a part of adaptive immunity, B-cells contain TLRs and complement receptors, in addition to BCRs This helps them achieve a basal level of activity, causing them to migrate towards helper T cells for further activation that is required for a CD 4+ Th cell mediated humoral response Abbas Fig. 7 -5
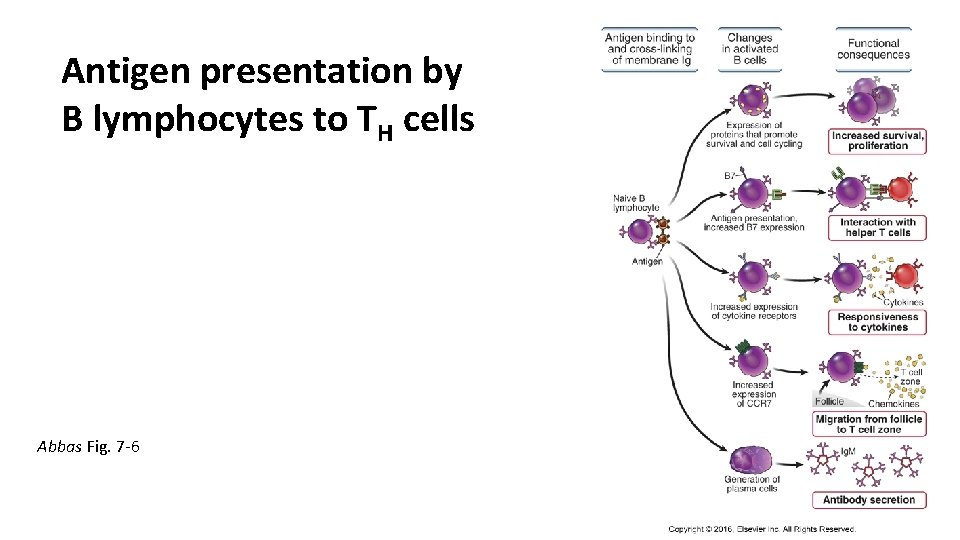
Antigen presentation by B lymphocytes to TH cells Abbas Fig. 7 -6
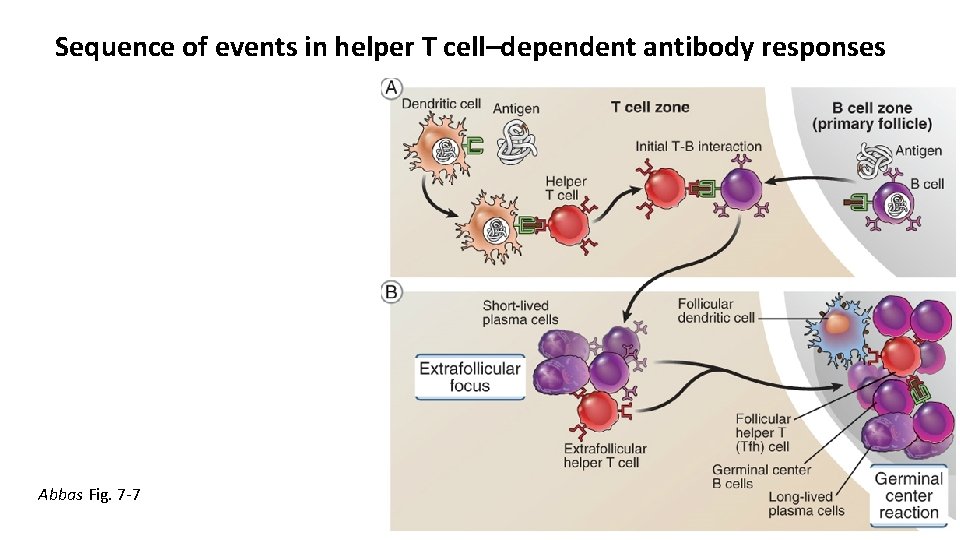
Sequence of events in helper T cell–dependent antibody responses Abbas Fig. 7 -7
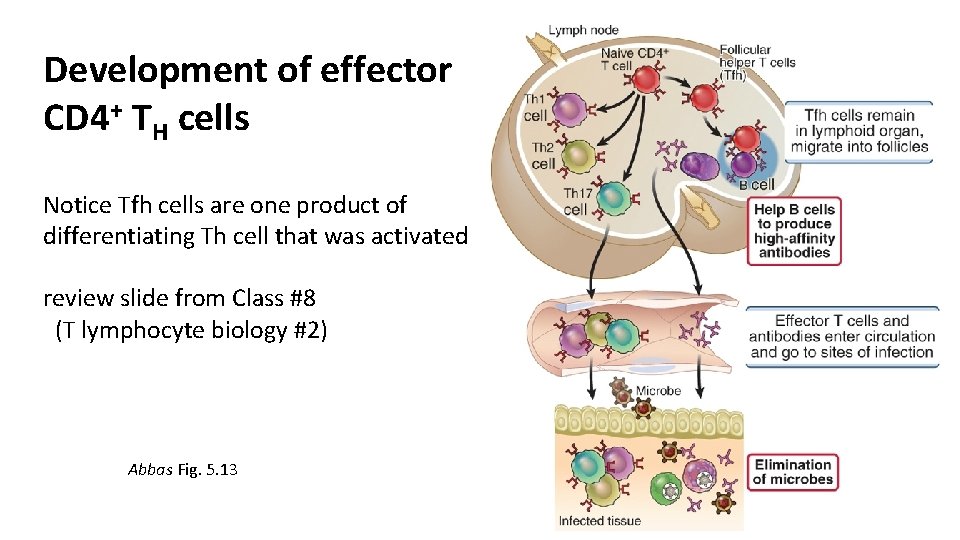
Development of effector CD 4+ TH cells Notice Tfh cells are one product of differentiating Th cell that was activated review slide from Class #8 (T lymphocyte biology #2) Abbas Fig. 5. 13

Antigen presentation by B lymphocytes to helper T cells. Notice that B cells activated by TH cells recognize the same antigen, though not necessarily same epitope Abbas Fig. 7 -8

Mechanisms of helper T cell–mediated activation of B lymphocytes Strict requirement for MHC-TCR interaction prevents neighboring B cells from being activated Abbas Fig. 7 -9
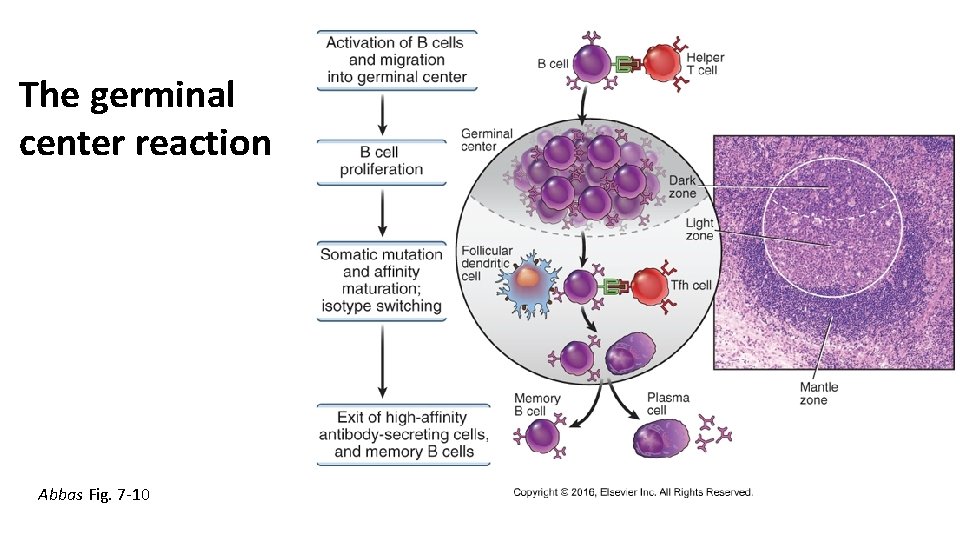
The germinal center reaction Abbas Fig. 7 -10
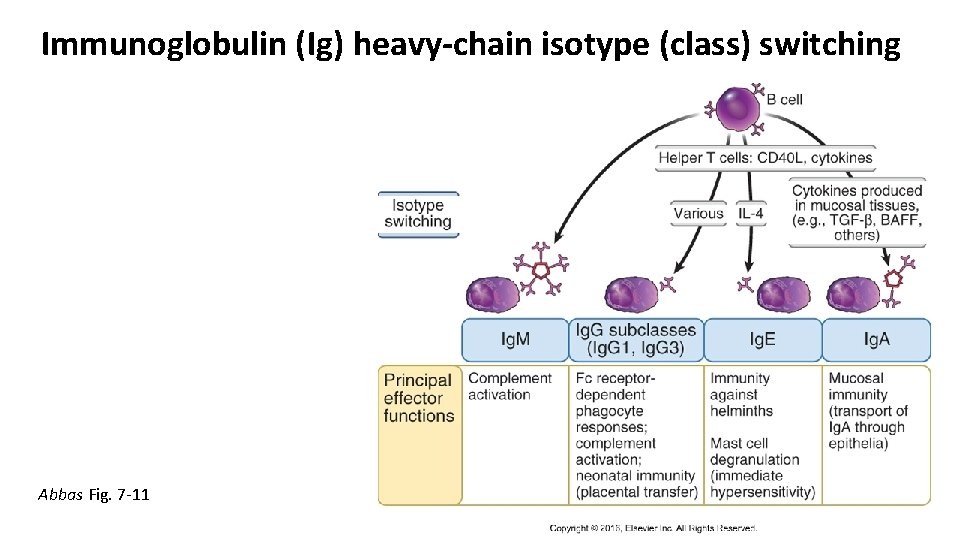
Immunoglobulin (Ig) heavy-chain isotype (class) switching Abbas Fig. 7 -11
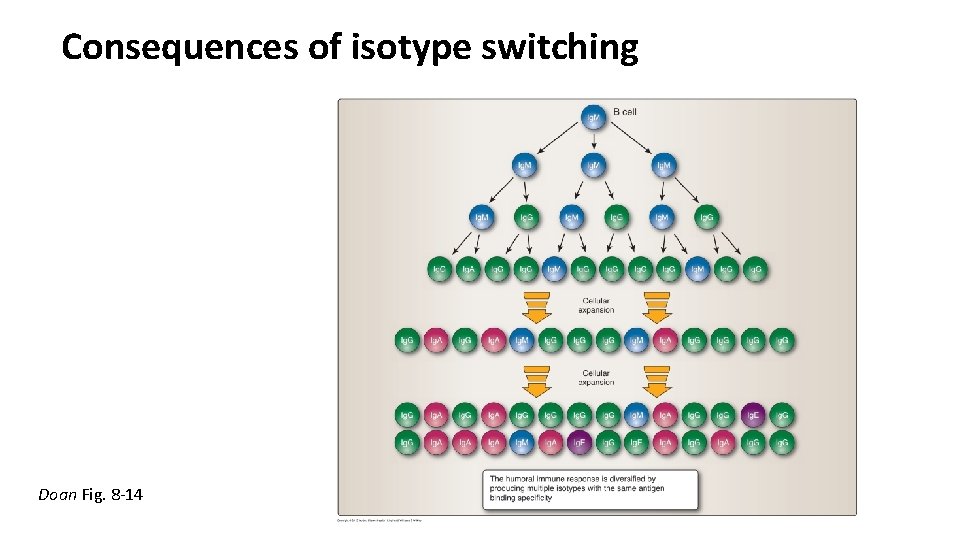
Consequences of isotype switching Doan Fig. 8 -14
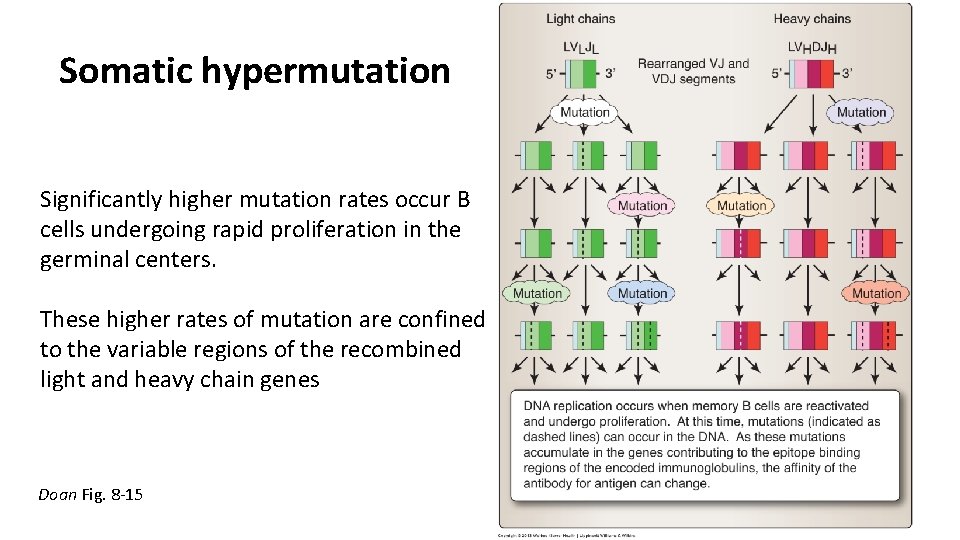
Somatic hypermutation Significantly higher mutation rates occur B cells undergoing rapid proliferation in the germinal centers. These higher rates of mutation are confined to the variable regions of the recombined light and heavy chain genes Doan Fig. 8 -15
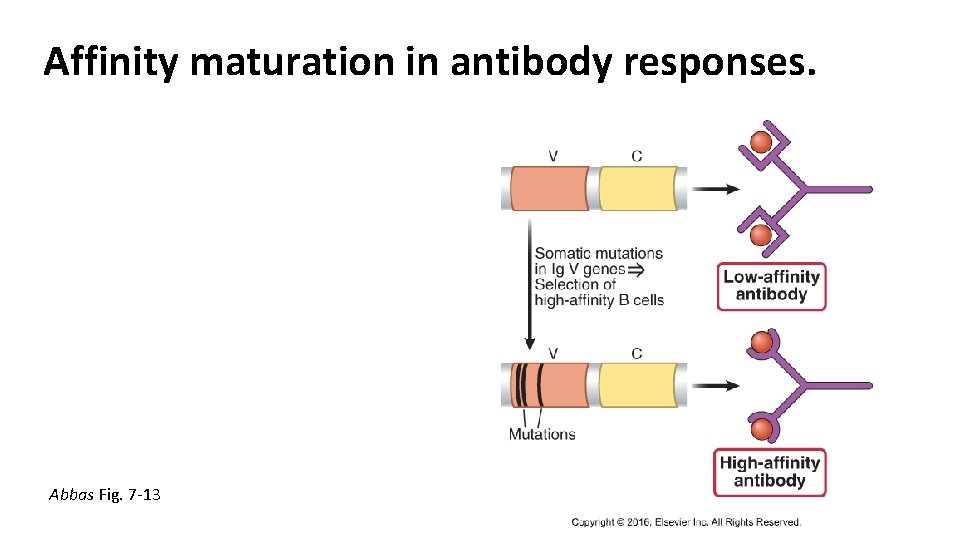
Affinity maturation in antibody responses. Abbas Fig. 7 -13

Selection of B cells with highest -affinity antibodies in the germinal centers Abbas Fig. 7 -14
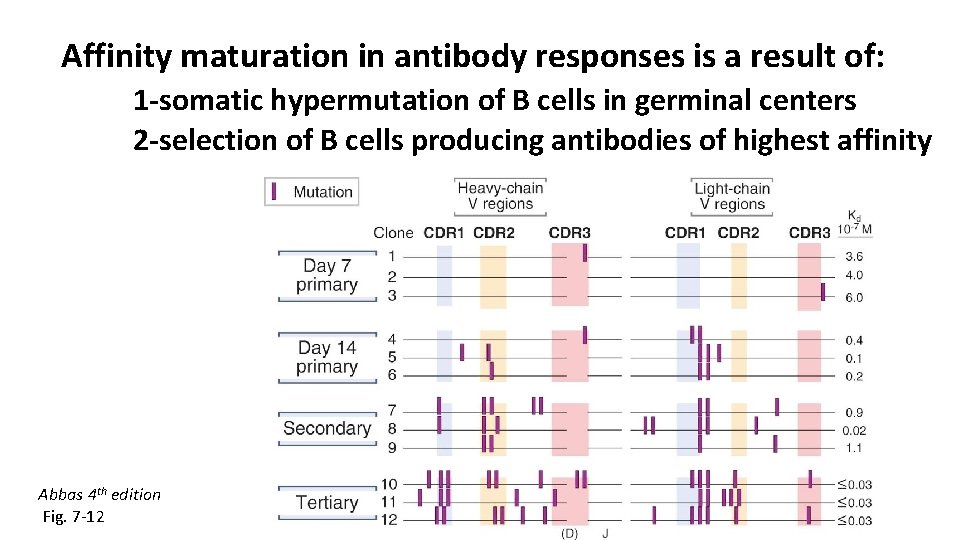
Affinity maturation in antibody responses is a result of: 1 -somatic hypermutation of B cells in germinal centers 2 -selection of B cells producing antibodies of highest affinity Abbas 4 th edition Fig. 7 -12
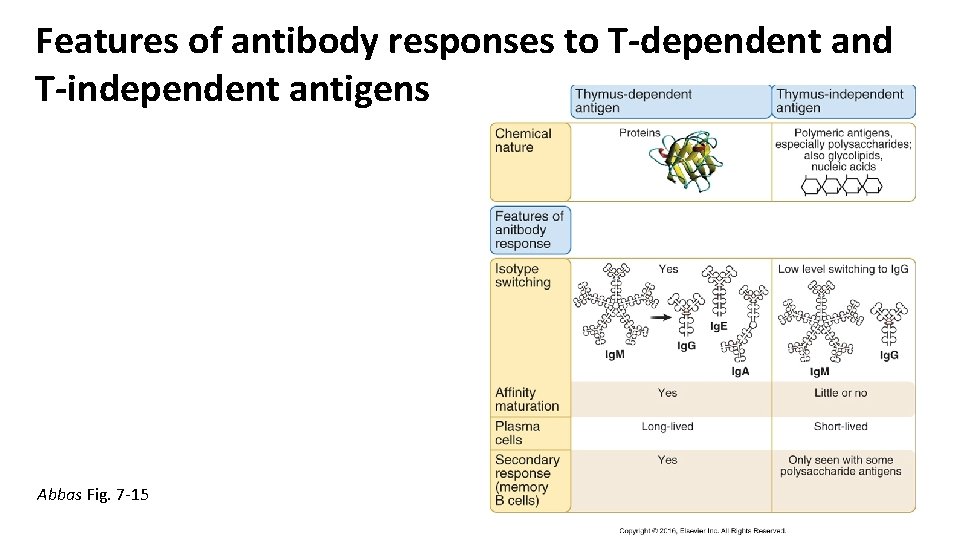
Features of antibody responses to T-dependent and T-independent antigens Abbas Fig. 7 -15

Transcytosis of a dimeric Ig. A antibody across epithelia J (joining) chain –holds dimeric Ig. A together and provides stability Poly-Ig receptor utilized to endocytose s. Ig. A from the basolateral side of epithelial cell Monomeric Ig. A also secreted into serum Parham Fig. 9 -18
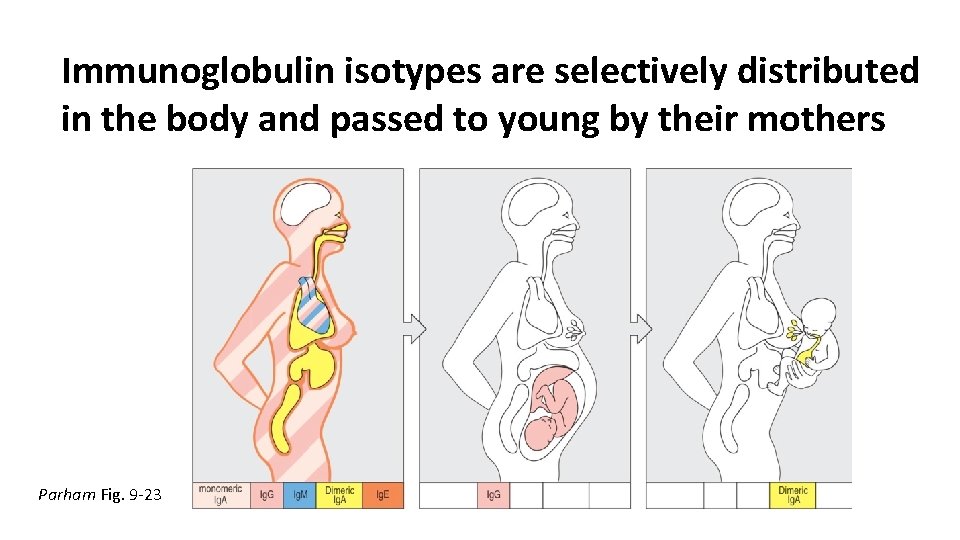
Immunoglobulin isotypes are selectively distributed in the body and passed to young by their mothers Parham Fig. 9 -23
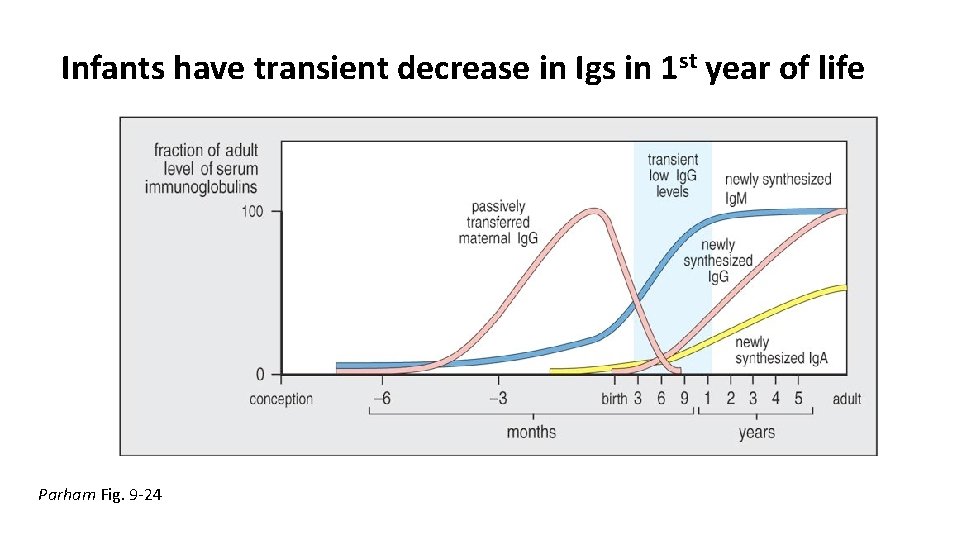
Infants have transient decrease in Igs in 1 st year of life Parham Fig. 9 -24

9 -14: Mothers provide protective antibodies to their young, both before and after birth During pregnancy, Ig. G from the maternal circulation is transported across the placenta and is delivered directly into the fetal bloodstream. This mechanism is so efficient that, at birth, human babies have as high a level of Ig. G in their plasma as their mothers, and as wide a range of antigen specificities. Ig. G is transported across the placenta by Fc. Rn (see Figure 9. 23, center panel). As a result of this mechanism, at birth the baby will have protection against pathogens that provoke an Ig. G response but not against pathogens that infect mucosal surfaces and provoke a secreted, dimeric Ig. A response. To rectify this deficit, infants obtain dimeric Ig. A in their mother’s milk, which contains antibodies against the microorganisms to which the mother has mounted an Ig. A response. On breast-feeding, the Ig. A is transferred to the baby’s gut, where it binds to microorganisms, preventing their attachment to the gut epithelium and facilitating their expulsion in feces. The transfer of preformed Ig. A from mother to child in breast milk is an example of the passive transfer of immunity (Figure 9. 23, right panel); another is the intravenous immunoglobulin given to patients with genetic defects in B-cell function During the first year of life there is a window of time when all infants are relatively deficient in antibodies and especially vulnerable to infection. As the maternally derived Ig. G is catabolized and the consumption of breast milk diminishes, the antibody level gradually decreases until about 6 months of age, when the infant’s own immune system starts to produce substantial antibody (Figure 9. 24). Consequently, Ig. G levels are lowest in infants aged 3– 12 months, and this is when they are most susceptible to infection. This problem is particularly acute in babies born prematurely, who begin life with lower levels of maternal Ig. G and take longer to attain immune competence after birth than babies born at term. (Parham 268)
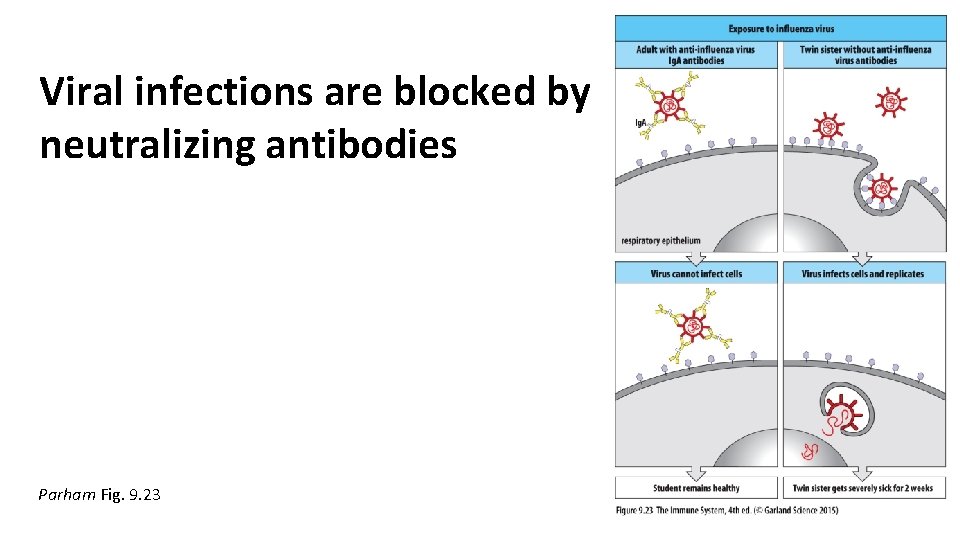
Viral infections are blocked by neutralizing antibodies Parham Fig. 9. 23

Disease-causing infections at mucosal surfaces can be prevented by neutralizing Ig. A antibodies Ig. G is the main source of neutralizing antibodies for the tissues of the human body Parham Fig. 9. 24
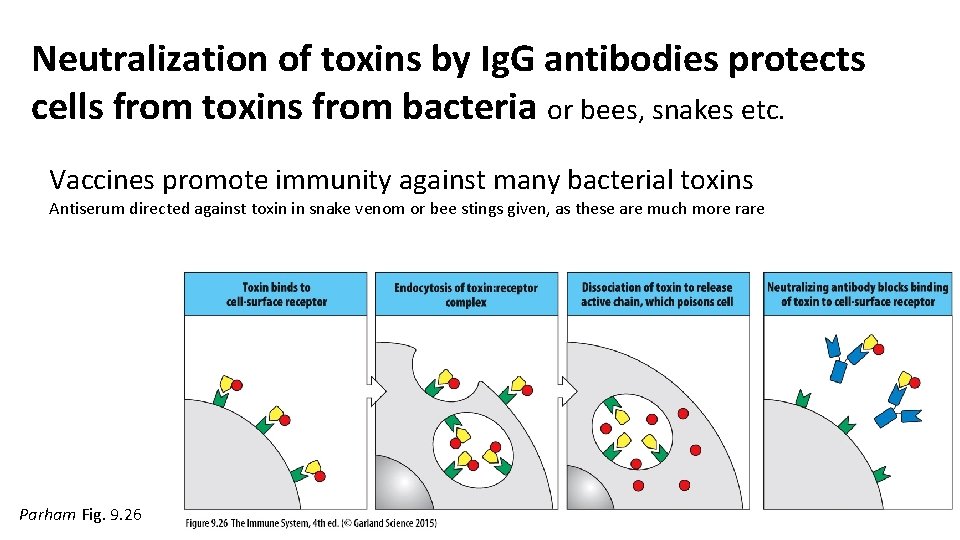
Neutralization of toxins by Ig. G antibodies protects cells from toxins from bacteria or bees, snakes etc. Vaccines promote immunity against many bacterial toxins Antiserum directed against toxin in snake venom or bee stings given, as these are much more rare Parham Fig. 9. 26
- Slides: 29