Pblock Elements VI A Group 16 Dr Nouf

P-block Elements VI A- Group 16 Dr. Nouf H. Alotaibi
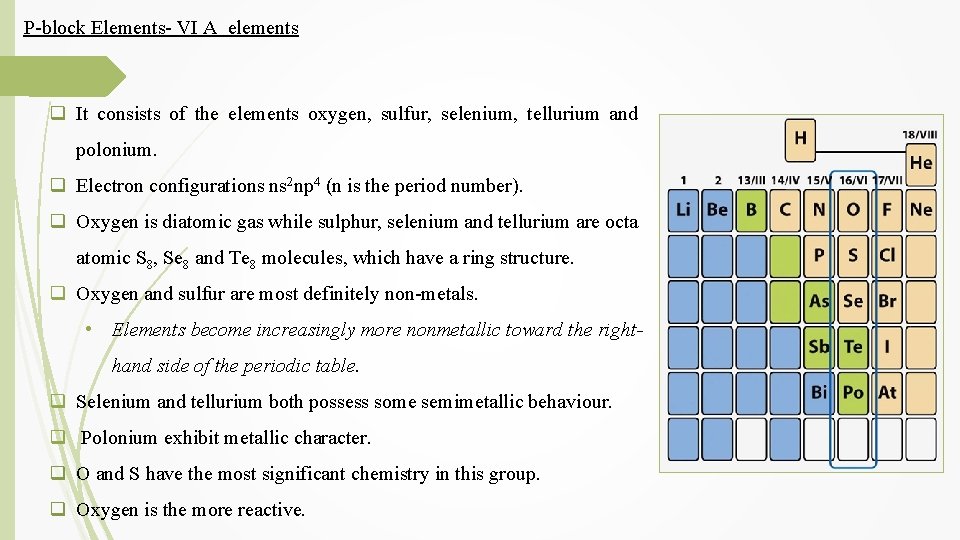
P-block Elements- VI A elements q It consists of the elements oxygen, sulfur, selenium, tellurium and polonium. q Electron configurations ns 2 np 4 (n is the period number). q Oxygen is diatomic gas while sulphur, selenium and tellurium are octa atomic S 8, Se 8 and Te 8 molecules, which have a ring structure. q Oxygen and sulfur are most definitely non-metals. • Elements become increasingly more nonmetallic toward the righthand side of the periodic table. q Selenium and tellurium both possess some semimetallic behaviour. q Polonium exhibit metallic character. q O and S have the most significant chemistry in this group. q Oxygen is the more reactive.

P-block Elements- VI A elements General Trend q The metallic properties increase down the group. q The melting and boiling points show the rising trend characteristic of nonmetals followed by the drop at metallic polonium. q Density, atomic radius, and ionic radius all increase down the group. q Ionization energy decreases down the group. Element M. p (°C) O 2 -219 S 8 119 Se 8 221 Te 452 Po 254 B. p(°C) -183 445 685 987 962

P-block Elements- VI A elements Oxidation State q The most common oxidation state is -2; q Sulfur can exist at a +4 and +6 state, q For Se, Te, and Po, oxidation states of +2, +4, and +6 are possible. Ø The even-numbered oxidation states from +6, through +4 and +2, to -2. q The stability of the -2 and +6 oxidation states decreases down the group. whereas that of the +4 state increases. q The stability of -2 oxidation state decreases down the group due to increase in atomic size and decrease in electronegativity. q The stability of +6 oxidation state decreases and +4 oxidation state increases due to inert pair effect. q Oxygen does not show +6 oxidation state due to absence of d – orbitals.

P-block Elements- VI A elements Reactivity with hydrogen: q All group 16 elements form hydrides of type MH 2. q Group 16 hydrides have a bent shape. q Hydride bond angles decrease and bond lengths increase down the group. Ø Order of bond angle H 2 O > H 2 Se > H 2 Te > H 2 Po q The ∠HMH bond angle in water is 104°. 311 but in other hydrides it is almost equal to 90°. q In H 2 O oxygen is involved in sp 3 hybridisation but in other hydride pure ‘p’ orbitals are participated in bonding. q Oxygen and sulphur form less stable polyoxides and polysulphides like H 2 O 2, H 2 Sn, (n=2 to 10). q H 2 O and H 2 O 2 can form H bonds, and therefore have higher melting and boiling points than other H 2 M compounds.

P-block Elements- VI A elements Reactivity with hydrogen: q Acidic nature: H 2 O < H 2 Se < H 2 Te § This is because the H-E bond length increases down the group. Ø Therefore, the bond dissociation energy down the group. q H 2 O is a liquid while the hydrides of the other VIA elements are a gas. (poisonous gases) § This is because strong hydrogen bonding is present in water. § This is due to small size and high electronegativity of O.

P-block Elements- VI A elements Reactivity with oxygen: q The other elements in the group are oxidized by O 2: M(s) + O 2 (g) MO 2 (M = S, Se, Te, Po) q VI A group elements form two types of oxides; dioxides of the type MO 2 and trioxides of the type MO 3. q Acidity also decreases down the group. q SO 2 is a gas whereas Se. O 2 is solid. § This is because Se. O 2 has a chain polymeric structure whereas SO 2 forms isolated units. q SO 2 is oxidized further, and the product is used in the final step of H 2 SO 4 manufacture. 2 SO 2 (g) + O 2 (g) 2 SO 3 (g)

P-block Elements- VI A elements Reactivity with oxygen: q Types of oxides: § Acidic oxides: Non- metallic oxides are usually acidic in nature. Ø SO 2 + H 2 O H 2 SO 3 (sulphurous acid) § Basic oxides: Metallic oxides are mostly basic in nature. § Basic oxides dissolve in water forming bases Ø Na 2 O + H 2 O 2 Na. OH § Amphoteric oxides: They show characteristics of both acidic as well as basic oxides. Ø Al 2 O 3 + 6 HCl(aq) 2 Al. Cl 3(aq) + 3 H 2 O Ø Al 2 O 3 + 6 Na. OH(aq) + 3 H 2 O(l) 2 Na 3 [Al(OH)6](aq) § Neutral oxides: These oxides are neither acidic nor basic. Example: CO, NO and N 2 O

P-block Elements- VI A elements Reactivity with halogens: q Halides are formed by direct combination: M(s) + X 2 (g) various halides (M = S, Se, Te ; X = F, Cl) q VI A group elements form; monohalides of the type M 2 X 2 ; dihalides of the type MX 2; tetrahalides of the type MX 4; and hexahalides of the type MX 6 (Where M = S, Se, Te ; X = halogen). q The oxidation states of S, Se and Te in monohalides is +1, in dihalides is +2, in tetrahalides is +4 and in hexahalides is + 6. q Hexafluorides are only stable halides which are gaseous and have sp 3 d 2 hybridisation and octahedral structure.

P-block Elements- VI A elements Reactivity with halogens: q Their structure and reactivity patterns depend on the sizes of the central atom and the surrounding halogens. § As the central atom becomes larger, the halides become more stable. q This pattern is related to the effect of electron repulsions due to crowding of lone pairs and halogen atoms around the central atom. § This is opposite to the previously observed bonding patterns, where bond strength decreases as bond length increases. q The stability of halides decreases in the order F- > Cl-> Br- > I-. § This is because M-X bond length increases with increase in size.

P-block Elements- VI A elements Allotropes in the Oxygen Family q Oxygen has two allotropes: § O 2, which is essential to life, and § O 3 or ozone, which is poisonous. q Sulfur has more than 10 different forms, due to the ability of S to catenate. S–S bond lengths and bond angles may vary greatly. § For example: a. Yellow Rhombic (α - sulphur) and b. Monoclinic (β- sulphur): 369 K Ø α − Sulphur β −Sulphur § At 369 K both forms are stable. It is called transition temperature. § Both of them have S 8 molecules. The ring is puckered and has a crown shape. q Selenium has several allotropes, some consisting of crown-shaped Se 8 molecules.

P-block Elements- VI A elements Oxygen q Oxygen is a gas at room temperature and 1 atm. q It is colorless and tasteless. q It is the most abundant element in the Earth's crust and the most abundant element in sea water. q It is second to nitrogen as the most abundant in the atmosphere. q There are many commercial uses for oxygen gas. q It is used in the manufacture of iron, steel, and other chemical manufacturing. q It is also used in water treatment, as an oxidizer in rocket fuel, for medicinal purposes, and in petroleum refining. q It exists as two allotropes: O 2 and O 3. q Its primary oxidation states are -2, -1, 0 and -1/2 in O 2 -.

P-block Elements- VI A elements Oxygen q When oxygen reacts with metals, it forms oxides that are mostly ionic in nature. q It is rarely featured as the central atom in a structure and can never have more than 4 elements bonded to it due to its small size and its inability to create an expanded valence shell. q When it reacts with hydrogen, it forms water, which is extensively hydrogen-bonded, has a large dipole moment and is considered an universal solvent.

P-block Elements- VI A elements Sulfur q Sulfur is a solid at room temperature and 1 atm pressure. q It is the sixteenth most abundant element in Earth's crust. q It exists in a variety of forms naturally, including elemental sulfur, sulfides, sulfates, and organosulfur compounds. q Sulfur is very unique in its ability to form a wide range of allotropes, more than any other element in the periodic table. q At room temperature, the sulfur molecule is a crown-shaped ring of eight atoms. q The most stable S allotrope is orthorhombic α-S 8, which consists of cyclo-S 8.

P-block Elements- VI A elements Sulfur q The most common state for sulfur to be in is the solid S 8 ring. q Sulfur exists in the gaseous form in five different forms (S, S 2, S 4, S 6, and S 8). q In order for sulfur to get to these states one must apply a sufficient amount of heat. q Two very common oxides of sulfur are sulfur dioxide (SO 2) and sulfur trioxide (SO 3). q These two compounds are used in the production of sulfuric acid. q Sulfur also exhibits a wide range of oxidation states, with values ranging from -2 to +6. q It is often the central ion in a compound and can easily hold up to 6 atoms around itself. q When in the presence of hydrogen it forms the compound H 2 S which is a poisonous gas, without hydrogen bonds and a very small dipole moment. q This reaction with hydrogen epitomizes how different oxygen and sulfur act despite their common valence electron configuration and common nonmetallic properties.

P-block Elements- VI A elements Sulfur q Common Compounds of Sulfur with Various Oxidation States

P-block Elements- VI A elements Contrasts in the Chemistry of Oxygen and Sulfur q Oxygen is limited to a maximum of four covalent bonds, whereas sulfur forms up to six covalent bonds. q For example, oxygen forms only one normal oxide with fluorine, OF 2, whereas sulfur forms several compounds with fluorine, including SF 6. q the oxygen-oxygen double bond is much stronger than the oxygen-oxygen single bond. q For sulfur and the other members of the group the energy of two single bonds is greater than that of one double bond. q For this reason, multiple bonding is only common for oxygen. TAB Bond energy Oxygen bonds O O (k. J/mol) 142 494 Bond Sulfur bonds S S (k. J/mol) 268 425

Contrasts in the Chemistry of Oxygen and Sulfur q Oxygen prefer to bond to other elements rather than to itself. q For example, the covalent bond to hydrogen is very strong q Due to the weakness of the oxygen-oxygen single bond compared to its bonds to other elements q Conversely, the sulfur tends to stabilize its compounds by catenation. q the sulfur-sulfur single-bond energy is only slightly lower than the energy of sulfur’s bonds to other elements Bond energy Oxygen bonds O O O Cl O H (k. J/mol) 142 218 459 Bond Sulfur bonds S S S Cl S H (k. J/mol) 268 271 363

P-block Elements- VI A elements Highlights of Sulfur Chemistry q Ozone (O 3): § ozone, is a very good oxidizing agent. § It can be used as a substitute for chlorine in purifying drinking water without giving the water an odd taste. § However, because of its unstable nature it disappears and leaves the water unprotected from bacteria. § Ozone at very high altitudes in the atmosphere is responsible for protecting the Earth's surface from ultraviolet radiation; however, at lower altitudes it becomes a major component of smog.
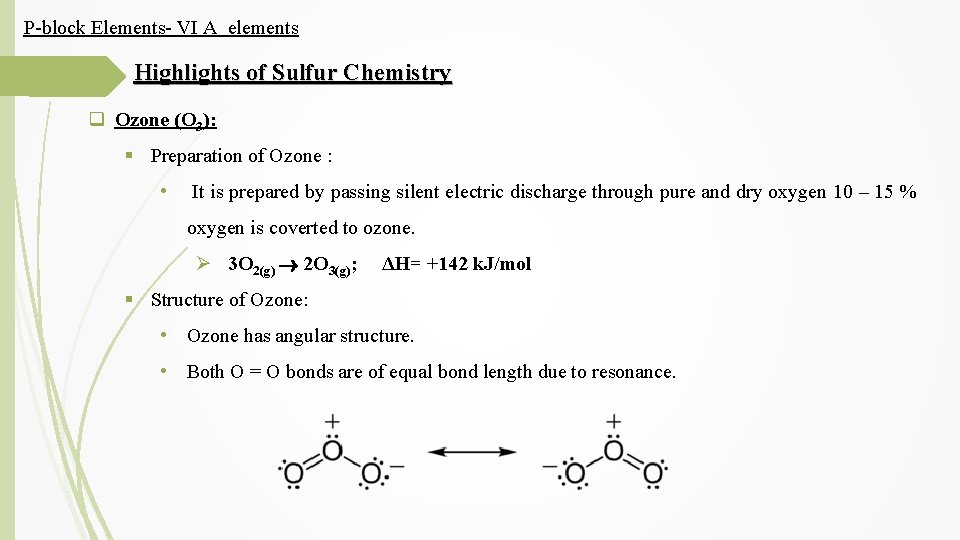
P-block Elements- VI A elements Highlights of Sulfur Chemistry q Ozone (O 3): § Preparation of Ozone : • It is prepared by passing silent electric discharge through pure and dry oxygen 10 – 15 % oxygen is coverted to ozone. Ø 3 O 2(g) 2 O 3(g); ΔH= +142 k. J/mol § Structure of Ozone: • Ozone has angular structure. • Both O = O bonds are of equal bond length due to resonance.
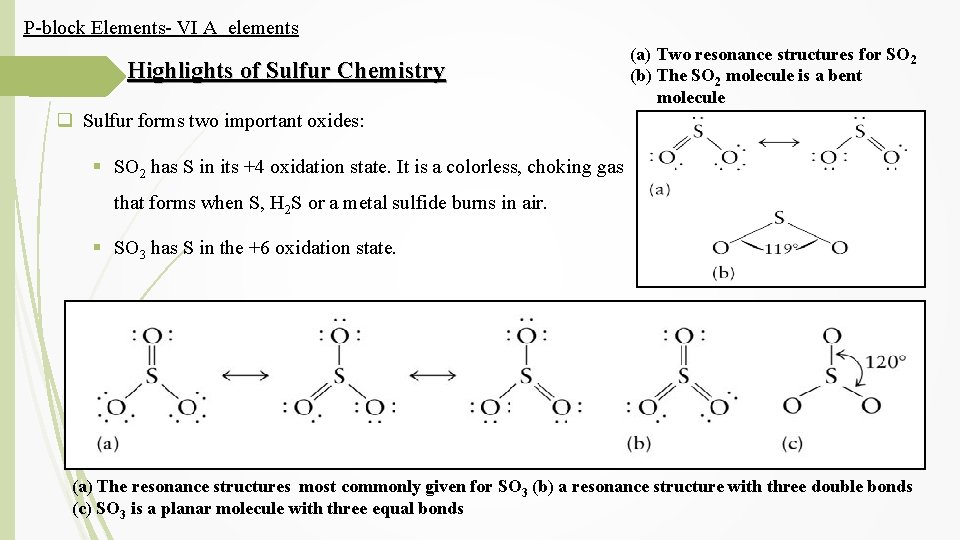
P-block Elements- VI A elements Highlights of Sulfur Chemistry (a) Two resonance structures for SO 2 (b) The SO 2 molecule is a bent molecule q Sulfur forms two important oxides: § SO 2 has S in its +4 oxidation state. It is a colorless, choking gas that forms when S, H 2 S or a metal sulfide burns in air. § SO 3 has S in the +6 oxidation state. (a) The resonance structures most commonly given for SO 3 (b) a resonance structure with three double bonds (c) SO 3 is a planar molecule with three equal bonds

P-block Elements- VI A elements Highlights of Sulfur Chemistry q SO 2 (Sulfur dioxide) § Sulfur burns in air to form SO 2 § Properties: § Gas, Colorless and Poisonous § Where the SO 2 in our air comes from Uses: Refrigerant, preserve dried fruit, bleach for textiles and flour, producing sulfuric acid § ~7× 1010 kg decomposition of vegetation and volcanic emissions § ~1× 1011 kg naturally occurring H 2 S which is oxidized to SO 2 by O § ~1. 5× 1011 kg industry and transportation (Electricity Plants) § Sulfur can act as either oxidizing agents or an reducing agent. § SO 2 is the starting material for making sulfur trioxide § The SO 3 is then used to make sulfuric acid Ø 2 SO 2(g) + O 2(g) 2 SO 3(g)

P-block Elements- VI A elements Highlights of Sulfur Chemistry q Sulfur forms two important oxoacids. § Sulfurous acid (H 2 SO 3) is a weak acid with two acidic protons. § Sulfuric acid (H 2 SO 4) is a strong acid, it is an important industrial chemical. It is an excellent dehydrating agent. H 2 SO 4 Two resonance structures for H 2 SO 3

P-block Elements- VI A elements Highlights of Sulfur Chemistry q H 2 SO 4 (Sulfuric acid) § H 2 SO 4 is the most heavily produced inorganic chemical worldwide. § About 2/3 of the sulfuric acid produced goes into manufacturing § Uses: Ø It is widely used in industry for the production of fertilizers, petrochemicals, and detergents. § Three important chemical properties of H 2 SO 4 are that it is; Ø a strong Bronstead acid (proton donor), q Properties: § Colorless Ø a dehydrating agent, and § Corrosive Ø an oxidizing agent. § Oily liquid

P-block Elements- VI A elements Highlights of Sulfur Chemistry q Sulfur Halides q Properties: § Sulfur reacts directly with all the halogens except iodine. § Colorless q SF 6 (Sulfur hexafloride) § Thermally stable § Sulfur reacts spontaneously in fluorine and burns brightly to give SF 6 § Nontoxic gas § Despite its high oxidation number (+6), It is not a good oxidizing agent § It is a good insulator in air and is used in switches on high-voltage power lines) SF 4 SF 6 S 2 F 10 S 2 Cl 2

P-block Elements- VI A elements Group 6 A(16) Elements Oxygen, like nitrogen, occurs as a low-boiling diatomic gas, O 2. Sulfur, like phosphorus, occurs as a polyatomic molecular solid. Selenium, like arsenic, commonly occurs as a gray metalloid. Tellurium, like antimony, displays network covalent bonding. Polonium, like bismuth, has a metallic crystal structure.
- Slides: 26