Pblock Elements V A ELEMENTS GROUP 15 DR

P-block Elements V A ELEMENTS (GROUP 15) DR. NOUF H. ALOTAIBI

P-block Elements- V A elements General trend o Electron configurations ns 2 np 3 (n is the period number) The s orbital is completely filled and p orbital is half filled which makes them stable. o Oxidation states that range from -3 to +5. The chemical properties of these elements are determined by the oxidation states exhibited by them. o Electronegativity decreases down the group. o Ionization energy decreases down the group. o The metallic character of the group increases down the group. o Atomic radii increase in size down the group. o Electron affinity decreases down the group. o Melting point increases down the group. o Boiling point increases down the group. Nitrogen (N), Phosphorus (P), Arsenic (As), Antimony (Sb), and Bismuth (Bi),

P-block Elements- V A elements General trend o As we move down the group, there is a transition from non-metallic to metallic through metalloid character. § Nitrogen and phosphorus, are nonmetals; § The remaining three members, arsenic (As), antimony (Sb), and bismuth (Bi), have some metallic character. § Two of the most dissimilar nonmetallic elements are in the same group: reactive phosphorus and unreactive nitrogen. o There is no clear division of properties between non-metals and metals in this group. The melting and boiling points are a good indication between metals and semimetals.

P-block Elements- V A elements General trend Melting and boiling points of the Group 15 elements o The melting and boiling points increase down the group, except for a decrease in melting point from Sb to Bi For the alkali metals, the melting points of main group metals tend to decrease down a group, whereas those of nonmetals tend to increase down a group. Ø Thus, the increase-decrease pattern indicates that the elements nitrogen and phosphorus are non-metals, arsenic and antimony are metalloids and bismuth is a typical metal. Element Melting point (°C) Boiling point (°C) N 2 -210 196 P 4 44 281 As Sublimes at 615 Sb 631 1387 Bi 271 1564

P-block Elements- V A elements General trend Oxidation State and Chemical Properties o The elements of group 15 generally exhibit -3, +3 and +5 oxidation states. o The tendency to exhibit -3 oxidation state decreases as we move down the group. Ø due to increase in the size of the atom and the metallic character. v Bismuth hardly forms any compound in oxidation state -3. o The stability of +5 state also decreases as we move down the group. v Bi. F 5 is the only well characterized Bi(V) compound.

P-block Elements- V A elements General trend Oxidation State and Chemical Properties o Due to inert pair effect, Ø the stability of +5 state decreases and +3 state increases as we move down the group in the periodic table. o Nitrogen reacts with oxygen and also exhibits +1, +2, +4 oxidation states. o On the other hand phosphorus shows +1 and +4 states in some oxoacids* (H 3 PO 2, H 4 P 2 O 6). o Nitrogen has only 4 electrons in its outermost shell (one in s orbital and 3 in p) which is available for bonding, hence it exhibits a maximum covalence of 4. o The heavier elements have a vacant d orbital in the valence shell which is used for bonding. *oxoacids is the acids that contain oxygen

P-block Elements- V A elements Group 15 hydrogen compounds NH 3 PH 3 As. H 3 Sb. H 3 Bi. H 3 decreasing basicity decreasing stability A-H bond strength 391 322 247 255 v The nitrogen hydrogen compounds are much more stable that all of the other hydrogen compounds formed by the members of Group 15.

P-block Elements- V A elements Group 15 compounds with X = N, C, O o N-X • π bonding important • 3 - and 2 -coordination is common in multiple bonding to X = N, C, O o P-X • pπ-pπ bonding is weak • 3 single bonds + lone-pair or 4 single bonds (cation) both common. Ø Example: Ø N 2 vs. P 4 or other forms of phosphorus, or As

P-block Elements- V A elements Halogen compounds of Group 15 • Halides are formed by direct combination of the elements: Ø 2 A(s) + 3 X 2 2 AX 3 (A = all except N) Ø AX 3 + X 2 AX 5 (A = all except N and Bi) • NF 3 is stable, NCl 3 is reactive (bleach for flour), neither is very basic. (NH 4 Cl + 3 Cl 2 = NCl 3 + 4 HCl) • Halides of P, As, and Sb are important laboratory chemicals: Ø AX 3 and AX 5 forms are both important. • PCl 3 and PBr 3 are reactive and useful as starting materials to other P-containing compounds. • PF 3 more stable. • PF 5, PCl 5, As. F 5, Sb. Cl 5 are strong Lewis acids.

P-block Elements- V A elements Nitrogen o Rare in the Earth’s crust but it is the principal component of our atmosphere (76% by mass) o N≡N triple bond strength is 944 k. J·mol-1 making it almost as inert as the noble gases. o N is used in medicines, fertilizers, explosives, and plastics. o The biggest commercial use for elemental nitrogen gas is for the formation of ammonia in the Haber process. o N has a wide range of oxidation numbers. o Nitrogen compounds are known to have every whole number oxidation number from -3 to +5. In addition, some fractional oxidation numbers are known to exists. o N can only form up to four bonds

P-block Elements- V A elements o The triple nitrogen-nitrogen bond energy is greater than that for the triple carbon-carbon bond. o Conversely, the single bond between two nitrogen atoms is much weaker than the carbon-carbon single bond. It is this large difference between N N and N N bond strengths (742 KJ/mol) that contributes to the preference in N chemistry for the formation of the dinitrogen molecule in a reaction rather than chains of nitrogen-nitrogen single bonds, as occurs in carbon chemistry. o Furthermore, the fact that dinitrogen is a gas means that an entropy factor also favors the formation of the dinitrogen molecule in chemical reactions. Nitrogen bonds N N Bond energy (k. J/mol) 247 942 Carbon bonds C C Bond energy (KJ/mol) 346 835

P-block Elements- V A elements Phosphorus o The radius of phosphorus is nearly 50% bigger than that of nitrogen. Thus P is too big to approach each other close enough for their 3 p orbitals to overlap and form π bonds o The availability of the 3 d orbitals means that phosphorus can form as many as six bonds. o It has two allotropes; white and red. o P 4 molecule found in white phosphorous while red phosphorous appears as a chain structure. White Red

P-block Elements- V A elements Phosphorus o Condensed phosphorus vapor is called white phosphorus and is a soft, white, poisonous, molecular solid consisting of tetrahedral P 4 molecules. o White phosphorus is highly reactive. o White phosphorus changes to red phosphorus (amorphous network) when heated in the absence of air. o Red phosphorus is much less reactive. o Red phosphorus is used in the striking surfaces of matchbook because he phosphorus ignites with friction. o White phosphorous reacts vigorously with the oxygen in air and inflames in air at only 35° C so must be stored under water. Red phosphorus is stable in air. https: //youtu. be/ch 3 jw. HZw 5 n. E o Phosphates (PO 43 -) and phosphoric acid (H 3 PO 4) are the most common compounds of phosphorus.
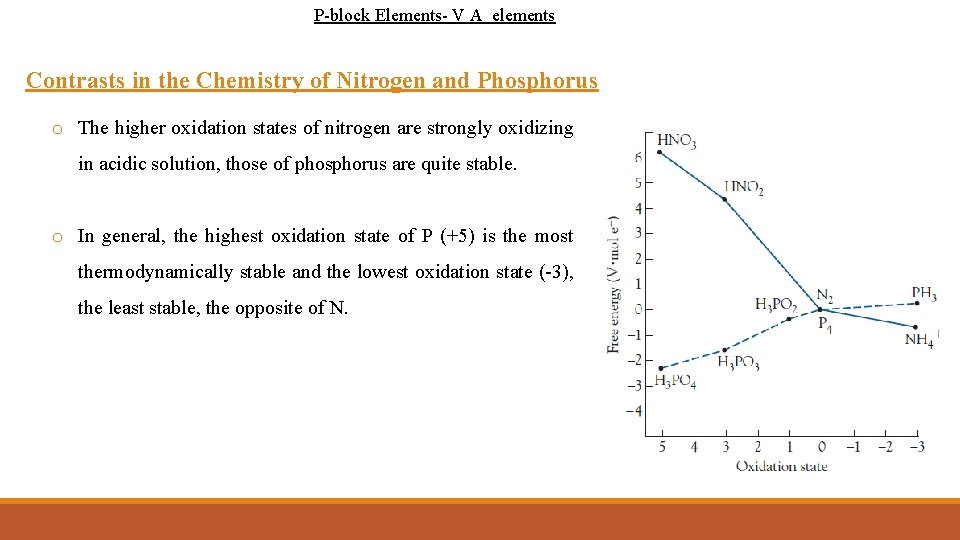
P-block Elements- V A elements Contrasts in the Chemistry of Nitrogen and Phosphorus o The higher oxidation states of nitrogen are strongly oxidizing in acidic solution, those of phosphorus are quite stable. o In general, the highest oxidation state of P (+5) is the most thermodynamically stable and the lowest oxidation state (-3), the least stable, the opposite of N.

P-block Elements- V A elements Contrasts in the Chemistry of Nitrogen and Phosphorus o N 2 is the stable form for the element. * Ø due to the very high strength of the N-N triple bond compared to the single (or double) bonds. o Elemental phosphorus contains groups of singly bonded phosphorus atoms (P 4). Ø there is a much smaller difference between the single and triple bond energies, v compare to the large difference between N‚N and N N bond strengths (742 KJ/mol). o N is very stable to oxidation, P reacts vigorously with oxygen to give oxides. Ø due to the strength of phosphorus-oxygen single bond Bond energy Nitrogen bonds (k. J/mol) Phosphorus bonds (k. J/mol) N N 247 P P 200 N N 942 P P 481 201 P O 335 * N 2 is a common. N O product from nitrogen-containing compounds in chemical reactions
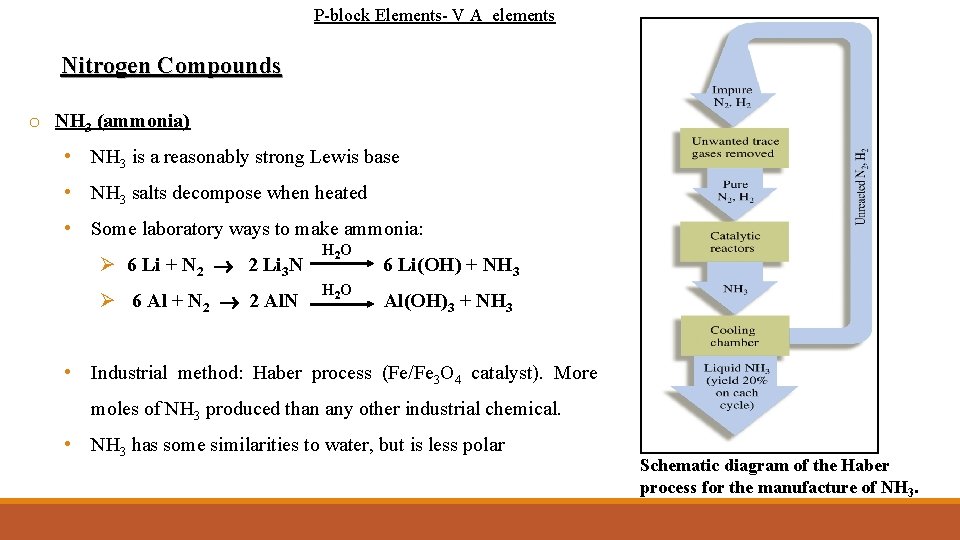
P-block Elements- V A elements Nitrogen Compounds o NH 3 (ammonia) • NH 3 is a reasonably strong Lewis base • NH 3 salts decompose when heated • Some laboratory ways to make ammonia: Ø 6 Li + N 2 2 Li 3 N Ø 6 Al + N 2 2 Al. N H 2 O 6 Li(OH) + NH 3 Al(OH)3 + NH 3 • Industrial method: Haber process (Fe/Fe 3 O 4 catalyst). More moles of NH 3 produced than any other industrial chemical. • NH 3 has some similarities to water, but is less polar Schematic diagram of the Haber process for the manufacture of NH 3.

P-block Elements- V A elements Nitrogen Compounds Nitrogen Ions o Azide ion (N 3 - ) • Highly reactive polyatomic anion • Its most common salt is sodium azide (Na. N 3) • Like most of the azide salts, Na. N 3 is shock sensitive • Na. N 3 is used in airbags where it decomposes to elemental sodium and nitrogen when detonated Ø 2 Na. N 3 (s) 2 Na(s) + 3 N 2 (g) • The azide ion is a weak base and accepts a proton to from its conjugate acid, hydrazoic acid (HN 3) which is a weak acid. • N 3 - Lewis Structure Ø https: //www. youtube. com/watch? v=KLY 7 b 8 Fglv 0 Ø https: //www. youtube. com/watch? v=e. T 0 x. WRFGB 78

P-block Elements- V A elements Nitrogen Compounds • Hydrogen Azide • It is a colorless liquid, • is quite different from the other nitrogen hydrides. • It is acidic, with a p. Ka similar to that of acetic acid: Ø HN 3(aq) + H 2 O(l) H 3 O-(aq) + N 3+(aq) • It is highly explosive, producing hydrogen gas and nitrogen gas: Ø 2 HN 3(l) H 2(g) + 3 N 2(g)

P-block Elements- V A elements Nitrides (solids that contain the nitride ion (N 3 -) • The combination of dinitrogen on heating with the Group 2 metals and lithium and Aluminium to form ionic nitrides, containing the N 3 - ion. • The reaction with lithium is Ø 6 Li(s) + N 2(g) → 2 Li 3 N(s) • Nitrides are only stable for small cations such as lithium or magnesium • Most nitrides dissolve in water to produce ammonia and the corresponding hydroxide Example: Ø Mg 3 N 2(s) + 6 H 2 O(l) 3 Mg(OH)2(s) + 2 NH 3(g)

P-block Elements- V A elements Nitrogen Oxide and Oxoacids o Nitrogen forms an excess of common oxides: o dinitrogen oxide, N 2 O; nitrogen monoxide, NO; dinitrogen trioxide, N 2 O 3; nitrogen dioxide, NO 2; dinitrogen tetroxide, N 2 O 4; and dinitrogen pentoxide, N 2 O 5. o In addition, there is nitrogen trioxide, NO 3, commonly called the nitrate radical, which is present in tiny but essential proportions in the atmosphere. o Each of the oxides is actually thermodynamically unstable with respect to decomposition to its elements, but all are kinetically stabilized. o Almost all nitrogen oxides are acidic o Some are acid anhydrides (a compound that forms an oxoacid when it reacts with water)

P-block Elements- V A elements Nitrogen Oxide and Oxoacids
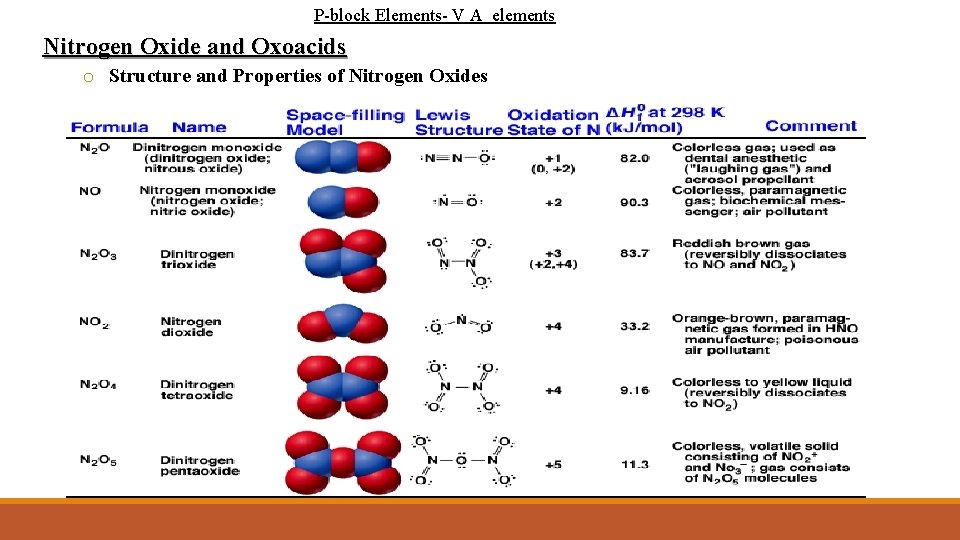
P-block Elements- V A elements Nitrogen Oxide and Oxoacids o Structure and Properties of Nitrogen Oxides

P-block Elements- V A elements Nitrogen Oxide and Oxoacids o HNO 3 (Nitric acid) • HNO 3 is used in the production of fertilizers and explosives • It is both an acid an oxidizing agent • It is made in the three-step Ostwald process STEP 1: Oxidation of ammonia 4 NH 3 (g) + 5 O 2 (g) 850 o. C, 5 atm, Pt/Rh→ 4 NO(g) + 6 H 2 O(g) STEP 2: Oxidation of nitrogen oxide 2 NO(g) + O 2(g) 2 NO 2 (g) STEP 3: Disproportionation (single atom is both oxidized and reduced) in water; 3 NO 2 (g) + H 2 O(l) 2 HNO 3(aq) + NO(g) (a) molecular structure of HNO 3 (b) resonance structure

P-block Elements- V A elements Phosphorus Oxides and Oxoacids o Oxoacids and oxoanions of phosphorous are among the most heavily manufactured chemicals. o Phosphate fertilizer production consumes two-thirds of all the sulfuric acid produced in the US. o The structures of the phosphorus oxides are based on the tetrahedral PO 4 unit. o Under acidic conditions, the highest oxidation state, +5, is the most stable species, as phosphoric acid, H 3 PO 4, and under basic conditions, the phosphate ion, PO 43 -, is the most stable o H 3 PO 4 (phosphoric acid) • Used primarily for the production of fertilizer, food additives, and detergent. • Many soft drinks have a small amount of phosphoric acid. • Although the phosphorus in H 3 PO 4 has an oxidation number of +5, the acid shows a oxidizing power at temperatures above 350ºC

P-block Elements- V A elements Phosphorus Oxides and Oxoacids o Phosphates (compounds contain PO 43 - ) • Phosphate rock is mined in huge quantities in Florida-USA and Morocco • The rock is crushed and treated with sulfuric acid to give a mixture of sulfates and phosphates called superphosphates, a major fertilizer Ø Ca 3(PO 4)2(s) + 2 H 2 SO 4 (l) 2 Ca. SO 4 (s) + Ca(H 2 PO 4)2(s)
- Slides: 25