pblock elements Grade 12 Nitrogen Family o Electronic

p-block elements Grade 12

Nitrogen Family o Electronic configuration ns 2 np 3 o Atomic and ionic radii Increases as we move down the group. But there is small increase in the size from As to Bi due to completely filled d- and f- orbitals. Q. Atomic radius of elements increases in a group, but there is only a small increase from As to Bi. A. This is due to the presence of completely filled d-orbitals and/or forbitals in them, which have weak screening effect and more effective nuclear charge called as inert- pair effect. o Ionization Enthalpy Decreases down the group. Q. I. E. of Group 15 elements is much greater than that of the Group 14 and Group 16 elements in the corresponding periods. A. Because of the extra stability of a half-filled p-orbital configuration. o Electronegativity Decreases down the group.

Chemical Properties o Oxidation States and Trends in Chemical Reactivity 1. Common oxidation states are -3, +3 and +5. Q. Nitrogen does not exhibit covalency greater than 4. A. Due to the absence of d-orbitals in the valence shell of N. 2. Tendency to exhibit -3 oxidation state decreases down the group due to increase in size and metallic character.

3. The stability of +5 oxidation state decreases down the group but stability of +3 oxidation state increases due to inert pair effect. Q. The stability of +5 state decreases and that of +3 state increases down the group. A. Due to inert pair effect. (explain inert pair effect) Q. Metallic character increases down the group. A. Due to decrease in I. E. and increase in atomic size.

Anomalous properties of Nitrogen o Nitrogen has small size, high electronegativity, high IP and non-availability of d-orbitals. o Form p-p pie bonds with itself. o Nitrogen exists as diatomic with one triple bond. Q. Nitrogen exists as diatomic molecule N 2 gas at room tempertaure. A. Due to the ability of the N atom to form pπ-pπ triple bond with another N atom. o Bond enthalpy is very high. o P-P, As-As, Sb-Sb have single bonds whereas Bi forms metallic bond.

Q. Nitrogen shows anomalous properties. A. Due to its small size, high electronegativity, high I. E. , nonavailability of d-orbitals in its valence shell and its ability to form pπ-pπ multiple bonds with itself, and with other elements like C and O.

o Weak catenation tendency. Q. Tendency for catenation is less in N as compared to the other members of the group. A. N-N single bond is weaker than those of the other elements. This is due to the high inter-electronic repulsion of the non-bonding electrons, owing to the small N-N bond length. o Non-availability of d- orbitals. 1. Reactivity towards hydrogen-All elements form hydrides of EH 3 type. Stability of Hydrides decreases from NH 3 to Bi. H 3, while the reducing character increases. Basicity trend: NH 3>PH 3>As. H 3>Sb. H 3>Bi. H 3 Q. Thermal stability of hydrides decreases from NH 3 to Bi. H 3. A. E-H bond enthalpy (bond strength) decreases down the group. (E Group 15 element) Q. Reducing character of EH 3 increases from NH 3 to Bi. H 3. A. E-H bond strength decreases, so H is easily supplied for reduction, on moving down the group.

Q. Basic character decreases down the group for EH 3. A. Due to the presence of a lone pair of electrons on the central atom, they behave as Lewis bases. N being the smallest in size, electron-density on N is high. Thus, it is the strongest base. Base strength decreases with increase in the size of the central atom. Q. NH 3 is a stronger base than PH 3. (phosphine). A. Due to smaller size of N, electron-density on N is higher. Thus, it is a strong Lewis base. Q. NH 3 has exceptionally high b. p. A. Due to the high electronegativity of N, intermolecular H bonds exist between NH 3 molecules.

Q. Among hydrides of Group 15 elements, Bi. H 3 is the strongest reducing agent. A. Bi. H 3 is the least stable, because Bi-H bond is the weakest (least bond enthalpy) compared to the E-H bond of the other elements of the group. Q. NH 3 molecule is triagonal pyramidal in shape. A. Due to the presence of 3 bp and 1 lp of electrons on the central N atom in the molecule. Structure. Q. NH 3 is used in the detection of metal ions such as Cu 2+, Ag+ etc. A. Due to the presence of a lone pair on the central N atom, NH 3 is a Lewis base. Thus, it donates electron pairs to metal ions and hence forms coloured complexes which help in the detection of metal ions. e. g Cu 2+(aq) + 4 NH 3 → [Cu(NH 3)4]2+(aq) deep blue solution

2. Reactivity towards oxygeno Two types of oxides : E 2 O 3 and E 2 O 5 type. o The oxide in higher oxidation state are more acidic than the lower ones. Acidic character decreases down the group. N 2 O 3 and P 2 O 3 are acidic, As 2 O 3 and Sb 2 O 3 are amphoteric and Bi 2 O 3 are basic.

3. Reactivity towards halogens Two types-EX 3 and EX 5. Nitrogen do not form pentahalides due to the non-availability of d-orbitals. Pentahalides are more covalent than trihalides. Q. PCl 5 exists, but NCl 5 does not. A. Due to the absence of d-orbitals in the valence shell of N, N cannot expand its valence shell. Q. Pentahalides of Group 15 elements are more covalent than trihalides. A. Higher the positive oxidation state of the central atom, the more is its polarizing power and, therefore, more the covalent nature of the bond. 4. Reactivity towards metals Form binary compounds exhibiting -3 oxidation states. e. g. Calcium nitride, Calcium phosphide etc.

Dinitrogen Preparation 1. In the laboratory, nitrogen is prepared by heating a mixture of ammonium chloride and sodium nitrite and a small quantity of water. If ammonium nitrite is heated by itself it decomposes to produce nitrogen gas. However, this reaction is very fast and may prove to be explosive.

2. By thermal decomposition of ammonium dichromate (NH 4)2 Cr 2 O 7 N 2+4 H 2 O+Cr 2 O 3 3. By thermal decomposition of sodium or barium azide Ba(N 3)2 Ba+3 N 2

Properties of Dinitrogen 1. Dinitrogen is inert at room temperature due to the high bond enthalpy of N-N triple bond. Q. N 2 gas is chemically inert at room temperature. A. Due to very high bond enthalpy of N≡N. 2. At high temperature reactivity increases and nitrogen form ionic nitrides with metals and covalent nitrides with nonmetals. 6 Li+N 2 2 Li 3 N

3 Mg+N 2 Mg 3 N 2 3. It reacts with hydrogen at 773 K in the presence of catalyst forms ammonia (Haber`s Process). N 2+3 H 2 2 NH 3 3. It reacts with dioxygen at very high temperature and forms nitric oxide NO. N 2+O 2 2 NO
Ammonia Preparationo By decay of nitrogenous organic matter NH 2 CONH 2+2 H 2 O (NH 4)2 CO 3 2 NH 3+CO 2

o On small scale ammonia can be obtained by decomposition of ammonium salts with caustic soda or lime. NH 4 Cl+Ca(OH)2 NH 3+H 2 O+Ca. Cl 2 o On large scale ammonia can be manufactured by Haber`s process. N 2+3 H 2 2 NH 3

Properties of ammonia 1. Has high melting and boiling point due to H 2 bonding. 2. Trigonal pyramidal in shape. 3. Ammonia is highly soluble in water. Its aqueous solution gives OH- in aqueous solution. NH +H O NH +OH 3 2 4 4. As a weak base, it precipitates the hydroxides of many metals from their salt solutions.

2 Fe. Cl 3+3 NH 4 OH Fe 2 O 3. H 2 O+3 NH 4 Cl (brown ppt. ) 5. Acts as Lewis base due to the presence of lone pair of electron on N 2. e. g. Cu+2+4 NH 3 [Cu(NH 3)4] +2 (deep blue)
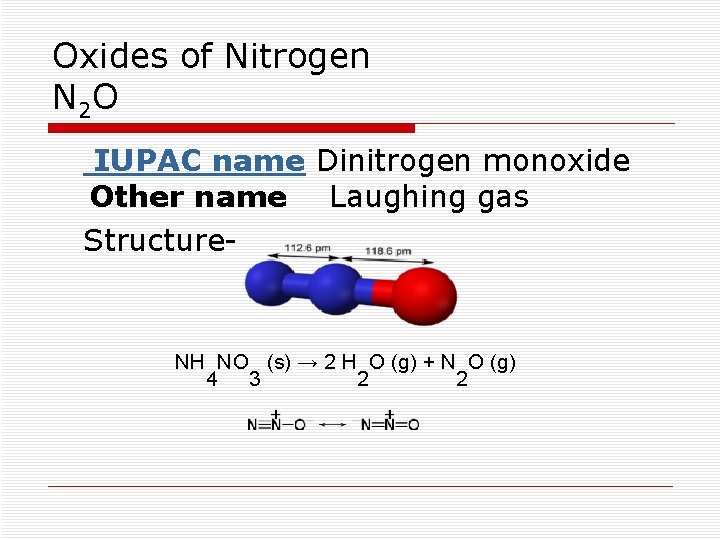
Oxides of Nitrogen N 2 O IUPAC name Dinitrogen monoxide Other name Laughing gas Structure- NH NO (s) → 2 H O (g) + N O (g) 4 3 2 2

NO n Preparation- 2 Na. NO 2+2 Fe. SO 4+3 H 2 SO 4 → Fe 2(SO 4)3+2 Na. HSO 4 + 2 H 2 O + 2 NO Preferred IUPAC name Nitric oxide Systematic name Nitrogen monoxide Other names Nitrogen(II) oxide
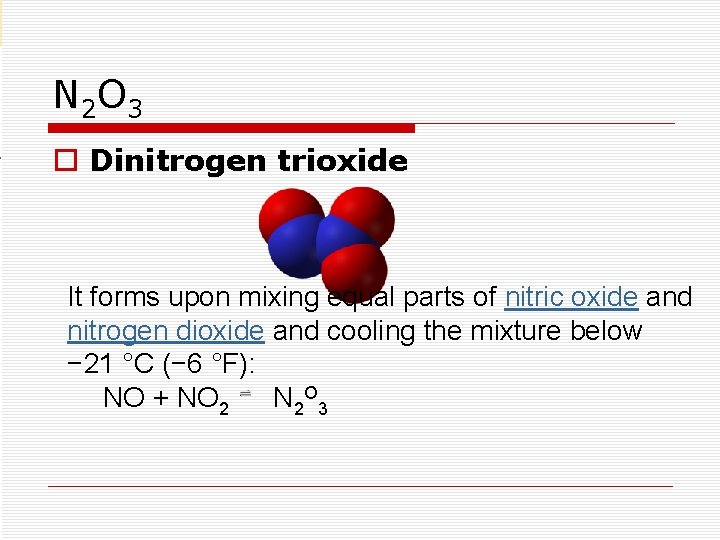
N 2 O 3 o Dinitrogen trioxide It forms upon mixing equal parts of nitric oxide and nitrogen dioxide and cooling the mixture below − 21 °C (− 6 °F): NO + NO 2 N 2 O 3

NO 2 o Nitrogen dioxide The thermal decomposition of some metal nitrates also affords NO : 2 2 Pb(NO ) → 2 Pb. O + 4 NO + O 32 2 2
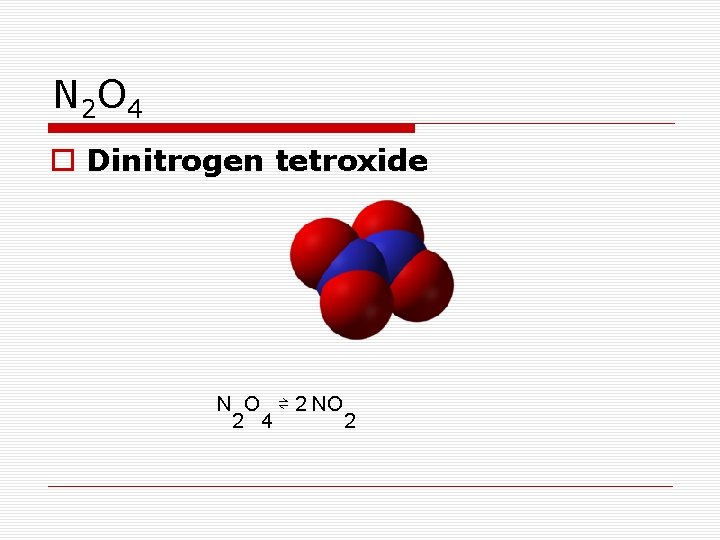
N 2 O 4 o Dinitrogen tetroxide N O ⇌ 2 NO 2 4 2

Q. NO 2 dimerises. A. NO 2 contains an odd no. of valence electrons. Thus, it behaves as a typical odd molecule. On dimerisation, it is converted into a more stable N 2 O 4 molecule with an even no. of electrons.
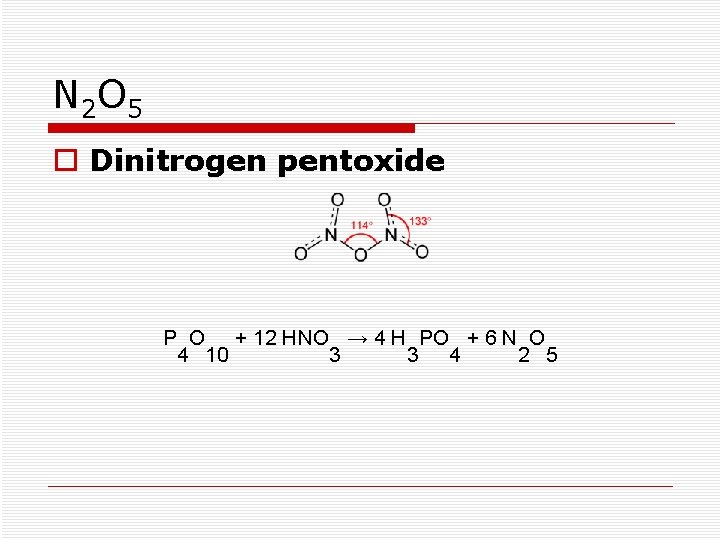
N 2 O 5 o Dinitrogen pentoxide P O + 12 HNO → 4 H PO + 6 N O 4 10 3 3 4 2 5

Nitric Acid 1. Nitric acid is made by reacting nitrogen dioxide (NO 2) with water. 3 NO 2 + H 2 O → 2 HNO 3 +NO 2. Almost pure nitric acid can be made by adding sulfuric acid to a nitrate salt, and heating the mixture with an oil bath. A condenser is used to condense the nitric acid fumes that bubble out of the solution. 2 Na. NO 3 + H 2 SO 4 2 HNO 3+Na 2 SO 4

o On a large scale it can be manufactured by Ostwald`s method. By catalytic (Pt/Rh gauge) oxidation of ammonia with atmospheric oxygen. 4 NH 3+5 O 2 4 NO+6 H 2 O 2 NO+O 2 2 NO 2 3 NO 2+H 2 O 2 HNO 3+NO
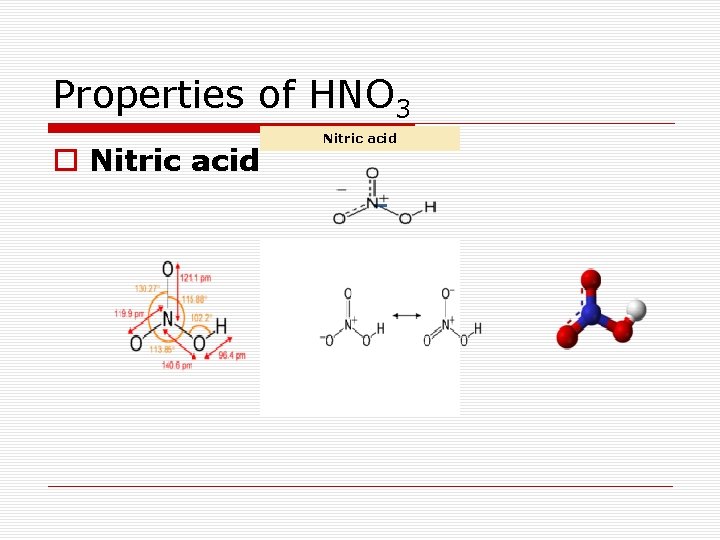
Properties of HNO 3 Nitric acid o Nitric acid

Chemical Properties of HNO 3 o Nitric acid is a strong oxidizing agent. When it undergoes thermal decomposition it yields nascent oxygen as follows: 2 HNO 3 H 2 O+2 NO 2+[O] o Remember: o The nascent oxygen so formed oxidizes non-metals, inorganic as well as organic compounds etc.

a) With Non-Metals o With hot concentrated nitric acid, non-metals are oxidized to their oxide while the acid itself gets reduced to nitrogen dioxide. o In all the reactions described below, the nascent oxygen is released during thermal decomposition of the acid, which oxidizes the non-metals. 2 HNO 3 H 2 O+2 NO 2+[O] i) With carbon o Nascent oxygen reacts with carbon to form carbon dioxide. C+2[O] CO 2 o The overall reaction is 4 HNO 3+C 2 H 2 O+4 NO 2+CO 2
![ii) With sulphur o Nascent oxygen reacts with sulphur to form sulphur trioxide. S+3[O] ii) With sulphur o Nascent oxygen reacts with sulphur to form sulphur trioxide. S+3[O]](http://slidetodoc.com/presentation_image/c3aaadfcb85791c8d900d48ea19fb125/image-32.jpg)
ii) With sulphur o Nascent oxygen reacts with sulphur to form sulphur trioxide. S+3[O] SO 3 o Sulphur trioxide in turn reacts with water to form sulphuric acid H 2 O+SO 3 H 2 SO 4 o The overall reactions is: S+6 HNO 3 2 H 2 O+H 2 SO 4+6 NO 2

iii) With phosphorous o Nascent oxygen reacts with phosphorous to form phosphorous (V) oxide. P 4+10[O] P 4 O 10 o Phosphorous (V) oxide in turn reacts with water to form phosphoric acid. 6 H 20+P 4 O 10 4 H 3 PO 4 o The overall reaction is: P 4+20 HNO 3 4 H 2 O+4 H 3 PO 4+20 NO 2

b) With Metals o Nitric acid behaves differently with different metals at different concentrations. i) With sodium, potassium and calcium the reaction is highly explosive. ii) With magnesium and manganese, cold and extremely dilute (1%) nitric acid, reacts to yield hydrogen.

iii) With Copper o With cold dilute nitric acid: Copper reacts with cold and dilute nitric acid to yield copper nitrate, water and nitric oxide. 3 Cu+8 HNO 3 3 Cu(NO 3)2+4 H 2 O+2 NO The formed nitric oxide combines with the oxygen of air to give brown fumes of NO 2. 2 NO+O 2 2 NO 2 o With concentrated nitric acid (cold or hot): Copper reacts with cold or hot concentrated nitric acid to yield copper nitrate, water and nitrogen dioxide. Cu+4 HNO 3 Cu(NO 3)2+2 H 2 O+2 NO 2

iv) With Zinc o With cold dilute nitric acid: Zinc reacts with cold and dilute nitric acid to yield zinc nitrate, water and nitric oxide. 3 Zn+8 HNO 3 3 Zn(NO 3)2+4 H 2 O+2 N 2 O o With concentrated nitric acid (cold or hot): Zinc reacts with cold or hot concentrated nitric acid to yield zinc nitrate, water and nitrogen dioxide. Zn+4 HNO 3 Zn(NO 3)2+2 H 2 O+2 NO 2

o Some metals like Cr, Al do not oxidise in conc. Nitric acid because of the formation of a thin film of oxide on its surface. o Conc. Nitric acid also oxidises non-metals. I 2+10 HNO 3 2 HIO 3+10 NO 2+4 H 2 O C+4 HNO 3 CO 2+2 H 2 O+2 NO 2 S 8+48 HNO 3 8 H 2 SO 4+48 NO 2+16 H 2 O P 4+20 HNO 3 4 H 3 PO 4+20 NO 2+4 H 2 O

Q. Conc. nitric acid is a strong oxidizing agent and oxidises metals like Cu and Zn. However, metals like Cr and Al do not dissolve in conc. HNO 3. A. Due to the formation of passive oxide films on the surface of Cr and Al.

Phosphorus-Allotropic Forms White Phosphorus o Translucent, white, waxy solid. o Insoluble in water but soluble in CS 2. o Dissolves in boiling Na. OH solution. P 4+3 Na. OH+3 H 2 O PH 3+3 Na. H 2 PO 2 o It ignites spontaneously in air at about 50 C and at much lower temp. if finely divided. This combustion gives phosphorus (V) oxide: P 4+5 O 2 P 4 O 10

Red Phosphorus It is prepared by heating white phosphorus to about 540 K in an inert atmosphere of nitrogen for several hours which on heating and under high pressure gives black phosphorus. (i) It is a hard crystalline solid without any smell and is poisonous in nature. (ii) It is insoluble in water as well as in carbon disulphide. (iii) It is more stable and relatively less reactive. (iv) It consists of tetrahedral units of P 4 linked to one another to constitute linear

Black Phosphorus o It is prepared by heating white phosphorus to about 470 K under high pressure of 1200 atmospheres in inert atmosphere. (i) It has metallic lustre. (ii) It is most inactive form of phosphorus. (iii) It has a layer type structure in which each layer consists of phosphorus atoms.

Phosphine o Preparation 1. Reaction of Calcium phosphide with water or dil. acid. Ca 3 P 2+6 H 2 O 3 Ca(OH)2+2 PH 3 Ca 3 P 2+6 HCl 3 Ca. Cl 2+2 PH 3 2. Lab. Method- Heating white phosphorus with conc. Na. OH in inert atmosphere of CO 2.

P 4+3 Na. OH+3 H 2 O PH 3+3 Na. H 2 PO 2 3. From impure phosphine PH 3+HI PH 4 I (+KOH) KI+H 2 O+PH 3 o Properties 1. Slightly soluble in water. 2. Aqueous solution of phosphine decomposes in light and gives red phosphorus and hydrogen.

3. 3 Cu. SO 4+2 PH 3 Cu 3 P 2+3 H 2 SO 4 4. 3 Hg. Cl 2+2 PH 3 Hg 3 P 2+3 HCl 5. PH 3+HBr PH 4 Br Q. PH 3 is a Lewis base. A. Due to the presence of a lone pair of electrons on the P atom. Q. Bond angle in PH 4+ is higher than that of PH 3. A. In PH 4+, there are 4 bond pairs on the central P atom. Thus, it has a tetrahedral shape with a bond angle of 109. 5. However, in PH 3, there is a lone pair on the central P atom. Due to lp-bp repulsion, bond angle in PH 3 is slightly less than 109. 5.

Phosphorus Trichloride Preparation 1. Lab method-Heat white phosphorus in current of dry chlorine. P 4+6 Cl 2 4 PCl 3 2. P 4+8 SOCl 2 4 PCl 3+4 SO 2+2 S 2 Cl 2
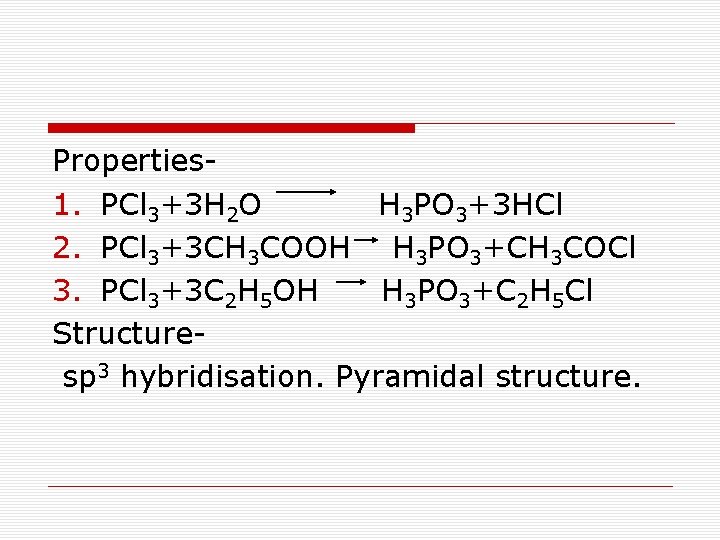
Properties 1. PCl 3+3 H 2 O H 3 PO 3+3 HCl 2. PCl 3+3 CH 3 COOH H 3 PO 3+CH 3 COCl 3. PCl 3+3 C 2 H 5 OH H 3 PO 3+C 2 H 5 Cl Structure sp 3 hybridisation. Pyramidal structure.

Phosphorus Pentachloride Preparation 1. Reaction of white phosphorus with excess of dry chlorine. P 4+10 Cl 2 4 PCl 5 2. P 4+10 SO 2 Cl 2 4 PCl 5+10 SO 2 Properties 1. PCl 5+H 2 O POCl 3+2 HCl POCl 3+3 H 2 O H 3 PO 4+3 HCl

2. On heating PCl 5 PCl 3+Cl 2 3. C 2 H 5 OH+PCl 5 C 2 H 5 Cl+POCl 3+HCl 4. 2 Ag+PCl 5 2 Ag. Cl+PCl 3 5. Sn+2 PCl 5 Sn. Cl 4+2 PCl 3 Structure In gaseous and liquid phases, it has a trigonal bipyramidal structure. 3 equatorial P-Cl bonds are equivalent while 2 axial bonds are longer than equatorial bonds.

Q. PCl 3 fumes in the presence of moisture. A. This is due to the formation of HCl. PCl 3 + 3 H 2 O H 3 PO 4 + 3 HCl Q. All the 5 P-Cl bonds in PCl 5 are not equivalent. A. PCl 5 molecule is trigonal bipyramidal in shape. The 3 equatorial bonds are of the same length but the 2 axial bonds are longer due to repulsion between bond pairs of the axial and equatorial bonds. Q. PCl 5 in the gaseous and solid states, respectively, do not have the same geometry. A. PCl 5 (g) is trigonal bipyramidal in shape. In the solid state, PCl 5 is ionic with tetrahedral [PCl 4]+ cation and octahedral [PCl 6]- anion.

Oxoacids of Phosphorus Oxidation State/ Formula /Name/ Acidic Protons/Compounds +1/H 3 PO 2 /hypophosphorous acid /1/ acid, salts +3/H 3 PO 3/(ortho)phosphorous acid/ 2/ acid, salts +5/(HPO 3)n/metaphosphoric acids/ n /salts (n=3, 4) +5/H 5 P 3 O 10/triphosphoric acid /3/ salts +5/H 4 P 2 O 7/pyrophosphoric acid /4/ acid, salts +5/H 3 PO 4/(ortho)phosphoric acid/ 3/ acid, salts
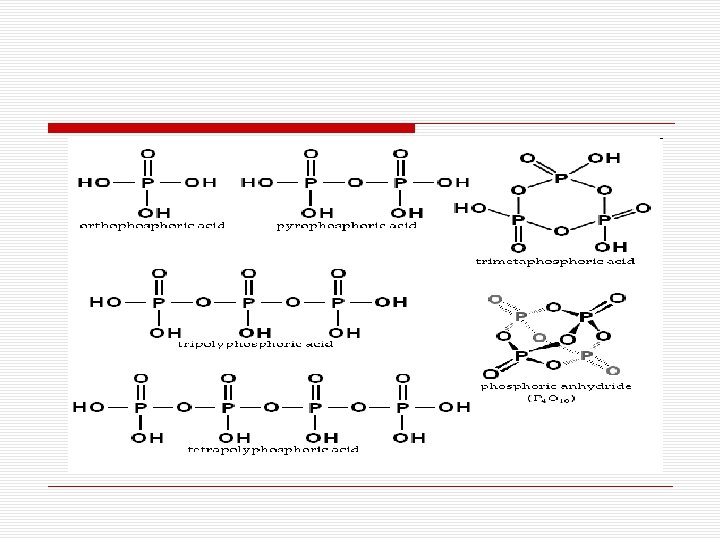

Q. Orthophosphorous acid H 3 PO 3 undergoes disproportionation. A. In H 3 PO 3, P is in the +3 state. ─3 and +5 states of P are more stable. So, it undergoes disproportionation. 4 H 3 PO 3 3 H 3 PO 4 + PH 3 Q. Ortho phosphorous acid H 3 PO 3 (phosphonic acid) is diprotic (or dibasic) while ortho phosphoric acid H 3 PO 4 is triprotic (or tribasic). A. Only those H atoms which are attached to oxygen in P-OH form are ionisable and cause basicity. Thus, H 3 PO 3 with only 2 P-OH bonds is dibasic and H 3 PO 4 with 3 P-OH bonds is tribasic. Q. Hypophosphoric acid H 3 PO 2 (phosphinic acid) is monobasic or monoprotic. A. There is only one P-OH bond. Thus, there is only 1 ionisable H atom and, hence only one H+ ion can be liberated. Q. H 3 PO 2 is a strong reducing agent. A. Oxoacids with P-H bonds have reducing properties and H 3 PO 2 (above fig. ) has 2 P-H bonds in it.

Q. NH 3 forms hydrogen bonds but PH 3 does not. A. Due to the small size and high electronegativity of the central N atom, NH 3 is able to form intermolecular H-bonds. Q. R 3 P=O exists, but R 3 N=O does not. (R alkyl group) A. P can form dπ-pπ bond with O. However, N, due to the absence of d-orbitals, cannot form dπ-pπ bond with O. Q. P(C 2 H 5)3 and As(C 6 H 5)3 act as ligands and bond with transition metals. A. Due to the ability of P and As to form dπ-dπ bond with transition metals. Q. Nitrogen exists as N 2 gas while phosphorous exists as P 4 solid. A. N can form pπ-pπ triple bond with another N atom while P, due to its larger size, cannot form pπ-pπ bond with another P atom. Q. The HNH bond angle in NH 3 is higher than HPH, HAs. H and HSb. H bond angles in their respective hydrides. A. Due to the small size of the central N atom and the small length of the N-H bond, bp-bp repulsion in NH 3 is greater than that of the other halides of the group. Moreover, in NH 3, hybridisation is sp 3 while in heavier elements the bond is between s and p orbitals of hydrogen and the element respectively.

Oxygen Family Electronic Configuration-ns 2 np 4. Atomic and Ionic Radii- Increases as we move down the group. Ionisation Enthalpy- Decreases down the group. Melting and Boiling Point- Increases down the group.

Electron Gain Enthalpy- Value becomes less negative as we move down the group. Q. Electron gain enthalpy of O is less negative than that of S. A. Due to the very small size of the O atom there is greater inter-electronic repulsion in the relatively smaller 2 p sub-shell hence the incoming electron experiences less attraction in oxygen atom. Electronegativity- Decreases down the group. Q. O 2 exists as a gas while other members are solids. A. Due to the ability of O to form pπ-pπ double bond with another O atom.

Oxidation States and Trends in Chemical Reactivity Shows -2, -1 and +2 oxidation states. Tendency to show -2 oxidation state decreases down the group. o Unlike oxygen, other elements show +2, +4 and +6 oxidation states. o Stability of +6 oxidation state decreases down the group but +4 increases due to inert – pair effect. Q. O shows only negative oxidation state, except in OF 2. A. Since O is a highly electronegative element, it shows only ─2 oxidation state. However, in OF 2, F is more electronegative due to which O has +2 oxidation state. o o

Q. Oxygen does not exhibit higher oxidation states (+4, +6) like the heavier members of its group. A. Due to the absence of d-orbitals in the valence shell of oxygen. Q. Stability of +6 state decreases and that of +4 state increases down Group 16. A. Due to inert pair effect.

Anomalous Behaviour of Oxygen o Small size , high electronegativity. o Absence of d- orbital. o Presence of strong hydrogen bonding in H 2 O but absent in H 2 S.

Reactivity with Hydrogen RTA o Form hydrides of type H 2 E. o Acidic character increases from H 2 O to H 2 Te. A o Thermal stability decreases down the group. T o All hydrides except H 2 O possess reducing property and it increases from H 2 S to H 2 Te. R

Q. Acidic character of the hydrides of Group 16 increases down the group. A. Due to decrease in H-E bond enthalpy. (E Group 16 element) Q. Thermal stability of H 2 E decreases down the group. A. H-E bond strength decreases. Q. Reducing character increases down the group. A. H-E bond strength decreases. Q. Water has exceptionally high m. p. and b. p. A. Due to intermolecular H-bonds, owing to the high electronegativity and small size of the O atom.

Reactivity with Oxygen o Form EO 2 and EO 3 types of oxides. o Reducing property of dioxide decreases from SO 2 to Te. O 2. o Both EO 2 and EO 3 types are acidic in nature. Q. Reducing property of dioxides reduces from SO 2 to Te. O 2. A. Due to inert pair effect the stability of +6 oxidation state decreases from S to Te.

Reactivity towards Halogens o Form EX 6, EX 4 and EX 2 types. o Stability of halides decreases from Fluoride to iodide. o Among EX 6 type, hexafluorides are only stable. o SF 6 is exceptionally stable for steric hindrance.

Q. Stability of halides of Group 16 decreases in the order F > Cl > Br > I. A. Stability of halides decrease with a decrease in electronegativity of the halogen. Q. SF 6 is very stable, chemically inert and resistant to hydrolysis. A. The S atom in SF 6 is sterically protected by 6 F atoms. Q. SF 4 and Se. F 6 are more easily hydrolysed. A. They are less sterically protected. In SF 4 steric protection is less due to lesser no. of F atoms and in Se. F 6 steric protection is less due to larger size of Se.

Dioxygen Preparationo Lab method- 2 KCl. O 3 2 KCl + 3 O 2(heat, Mn. O 2) 2. By thermal decomposition 2 Ag 2 O 4 Ag + O 2 2 Hg. O 2 Hg+O 2 2 Pb 3 O 4 6 Pb. O +O 2
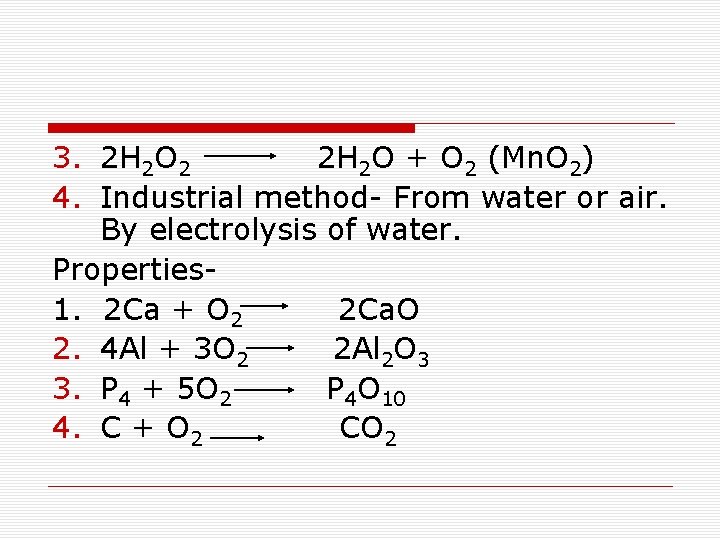
3. 2 H 2 O 2 2 H 2 O + O 2 (Mn. O 2) 4. Industrial method- From water or air. By electrolysis of water. Properties 1. 2 Ca + O 2 2 Ca. O 2. 4 Al + 3 O 2 2 Al 2 O 3 3. P 4 + 5 O 2 P 4 O 10 4. C + O 2 CO 2
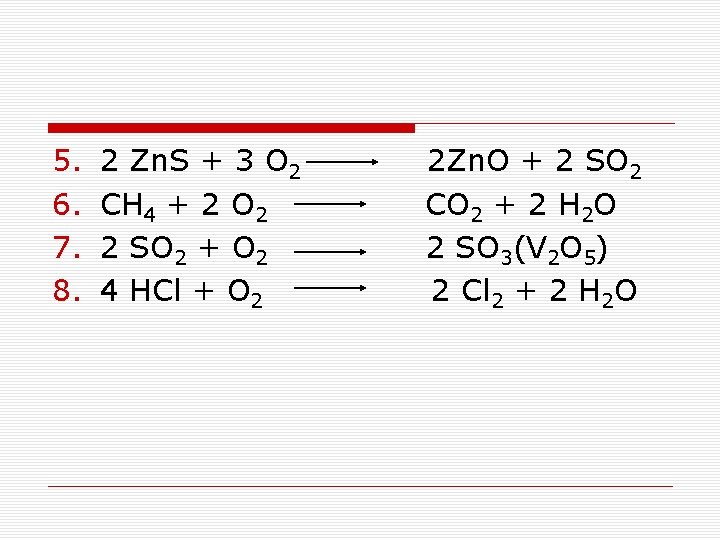
5. 6. 7. 8. 2 Zn. S + 3 O 2 2 Zn. O + 2 SO 2 CH 4 + 2 O 2 CO 2 + 2 H 2 O 2 SO 2 + O 2 2 SO 3(V 2 O 5) 4 HCl + O 2 2 Cl 2 + 2 H 2 O

Simple Oxides o A binary compound of oxygen with another element is called oxide. o Oxides may be simple or mixed. o Simple oxides may be acidic, basic or amphoteric. o Oxide + H 2 O Acid (Acidic Oxide) Example- SO 2, CO 2

o SO 2 + H 2 O H 2 SO 3 o Non- metal oxides are acidic but oxides of metals in high oxidation states also have acidic character. (e. g. Mn 2 O 7, Cr. O 3) o Oxide + H 2 O Base (Basic Oxide) Examples- Na 2 O, Ca. O, Ba. O. o Ca. O + H 2 O Ca(OH)2 o Metallic oxides are basic.

o Some metallic oxides shows both acidic as well as basic character called as Amphoteric oxides. o Some oxides are neither acidic nor basic called as Neutral oxides.

Ozone Preparation- When a slow dry stream of oxygen is passed through a silent electrical discharge conversion of oxygen to ozone occurs. The product is OZONISED OXYGEN. 3 O 2 2 O 3 – Heat This is Endothermic Reaction.

Properties 1. Powerful oxidizing agent. O 3 O 2 + O 2. Oxidizes Pb. S to Pb. SO 4 and I- to I 2. Pb. S + 4 O 3 Pb. SO 4 +4 O 2 2 I- +H 2 O +O 3 2 OH- + I 2 + O 2 3. NO + O 3 NO 2 + O 2

Q. Though the formation of oxides with elements is exothermic, some external heating is required to initiate the reaction. A. Bond dissociation enthalpy of O=O is very high. Q. The 2 O-O bond lengths in the ozone molecule are identical. A. Ozone is a resonance hybrid of 2 canonical structures. Q. O 3 is a powerful oxidizing agent. A. O 3 O 2 + O The above reaction is highly exothermic ( H negative and S positive, thus, G negative). The nascent O liberated makes O 3 a powerful oxidizing agent.

Sulphur-Allotropic Forms o Main allotropic forms are 1. Yellow rhombic sulphur 2. Monoclinic sulphur o Sulphur forms yellow, rhombic crystals out of 8 membered rings of sulphur atoms (S 8). o Sulphur forms yellow, monoclinic, needle-like crystals out of 8 -membered rings of sulphur atoms (S 8). o Plastic sulphur is yellow and made up of long chains of sulphur atoms. It reverts to S 8 rings in time.

Sulphur Dioxide Preparation 1. Sulphur is burnt in air. S + O 2 SO 2 2. Lab method- Treating SO 3 - with H 2 SO 4. SO 3 - + 2 H+ SO 2 + H 2 O 3. Industrial- As a by- product of roasting of sulphide ores. 4 Fe. S 2 + 11 O 2 2 Fe 2 O 3 + 8 SO 2

Properties 1. Colourless gas with pungent smell. 2. Soluble in water. 3. SO 2 + H 2 O H 2 SO 3(sulphurous acid) 4. 2 Na. OH + SO 2 Na 2 SO 3 + H 2 O + SO 2 2 Na. HSO 3 5. SO 2 + Cl 2 SO 2 Cl 2(sulphuryl chloride) ( +nce of charcoal ) 2 SO 2 + O 2 2 SO 3( V 2 O 5, Oxidation)

6. Moist SO 2 behaves as reducing agent. • Fe (iii) Fe (ii) • Decolorizes acidified KMn. O 4 solution. 2 Fe 3+ + SO 2 + 2 H 2 O 2 Fe 2+ + SO 42 - + 4 H+ 5 SO + 2 Mn. O - + 2 H O 5 SO 2 - 2 4 + 4 H+ + 2 Mn 2+
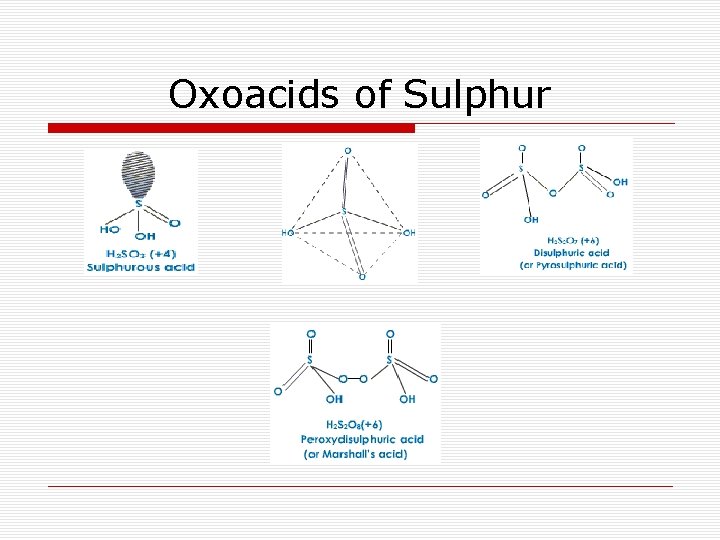
Oxoacids of Sulphur

Sulphuric Acid 1. Manufacture- By Contact Process i) S 8+8 O 2 8 SO 2(burning sulphur) 4 Fe. S 2+11 O 2 2 Fe 2 O 3 + 8 SO 2 (roasting of sulphur rich ore) ii) 2 SO 2 + O 2 2 SO 3+ HEAT(oxidn. ) (V 2 O 5 or Platinised asbestos) iii) SO 3 + H 2 SO 4(conc. ) H 2 S 2 O 7 +H 2 O 2 H 2 SO 4

Properties 1. Dissociation H 2 SO 4 H 2 O + SO 3 2. Acidic Nature H 2 SO 4 H+ + HSO 4 - H+ + SO 42 -

3. As dehydrating agent i) Drying of gases-Gases which do not react with H 2 SO 4 like CO 2, SO 2, Cl 2. ii) Charring- C 12 H 22 O 11 H 2 O + 12 C (conc. H 2 SO 4) 4. Oxidizing Action-Strong oxidising agent. Cu + 2 H 2 SO 4 Cu. SO 4 + SO 2 +2 H 20(conc. )

Halogen Family o Electronic Configuration- ns 2 np 5 o Atomic and Ionic Radii- Increases as we move down the group. o Ionisation Enthalpy- Decreases down the group. o Electron Gain Enthalpy- Halogens have max. –ve electron gain enthalpy and it decreases down the group.

o Electronegativity- Decreases down the group. o Colour- Halogens are coloured. Colour deepens down the group.

Oxidation States and Trends in Chemical Reactivity o All halogens exhibit -1 oxidation state. o Cl, Br and I exhibit +1, +3, +5 and +7 oxidation states. o F has absence of d- orbitals. o All halogens are highly reactive. o Reactivity decreases down the group.

Anomalous Behaviour of Fluorine o Small size, highest electronegativity, low F-F bond dissociation enthalpy and non- availability of d- orbital. o Reactions of fluorine are Exothermic. o HF is liquid while others are gases.

Reactivity towards Hydrogen o Form HX type. o The affinity for hydrogen decreases from fluorine to iodine. o Acidic strength increases from HF to HI. o Reducing character increases down the group. o Thermal stability decreases down the group.

Reactivity towards Oxygen o Halogens forms oxides with oxygen but most of them are unstable. o Fluorine forms two types of oxides- OF 2(+1) and O 2 F 2(+2). o Only O 2 F 2 is thermally stable

Reactivity towards metals o Form Metal Halides. o Ionic character decreases from MF to MI. o The Halides in higher oxidation state will be more covalent than the one in lower oxidation state.

Reactivity of Halogens with other Halogens. o Form inter- Halogen Compounds like XX`, XX 3`, XX 5`, XX 7` [ where X= larger size Halogen and X` is smaller size Halogen]
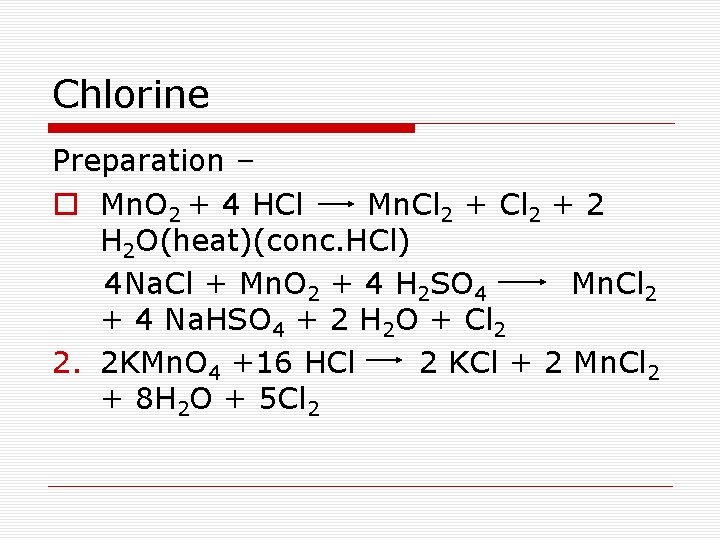
Chlorine Preparation – o Mn. O 2 + 4 HCl Mn. Cl 2 + 2 H 2 O(heat)(conc. HCl) 4 Na. Cl + Mn. O 2 + 4 H 2 SO 4 Mn. Cl 2 + 4 Na. HSO 4 + 2 H 2 O + Cl 2 2. 2 KMn. O 4 +16 HCl 2 KCl + 2 Mn. Cl 2 + 8 H 2 O + 5 Cl 2

Manufacture of Cl 21. Deacon`s Process- By Oxidation 4 HCl + O 2 2 Cl 2+2 H 2 O (Cu. Cl 2, 723 K) 2. Electrolytic Process- By electrolysis of brine (conc. Na. Cl solution). Cl 2 is liberated at anode.

Properties of Chlorine o React with metals and non- metals to form chlorides. 1. 2 Al + 3 Cl 2 2 Al. Cl 3 2. P 4 + 6 Cl 2 4 PCl 3 o It has great affinity for hydrogen. H 2 + Cl 2 2 HCl H 2 S + Cl 2 2 HCl + S

o Excess ammonia 8 NH 3 + 3 Cl 2 6 NH 4 Cl +N 2 Excess chlorine NH 3 + 3 Cl 2 NCl 3 + 3 HCl o Cold and dilute Na. OH 2 Na. OH + Cl 2 Na. Cl + Na. OCl(hypochlorite) + H 2 O Hot and conc. Na. OH 6 Na. OH + 3 Cl 2 5 Na. Cl + Na. Cl. O 3(chlorate) + 3 H 2 O
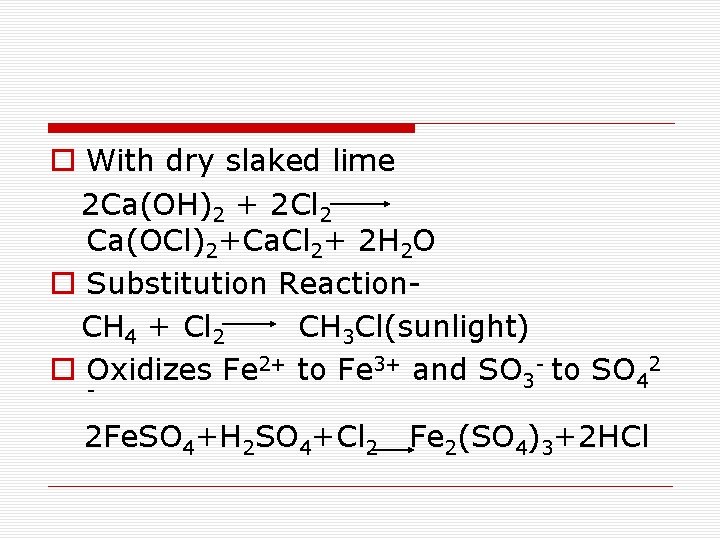
o With dry slaked lime 2 Ca(OH)2 + 2 Cl 2 Ca(OCl)2+Ca. Cl 2+ 2 H 2 O o Substitution Reaction CH 4 + Cl 2 CH 3 Cl(sunlight) o Oxidizes Fe 2+ to Fe 3+ and SO 3 - to SO 42 - 2 Fe. SO 4+H 2 SO 4+Cl 2 Fe 2(SO 4)3+2 HCl
![o Powerful bleaching agent. Cl 2 + H 2 O 2 HCl + [O] o Powerful bleaching agent. Cl 2 + H 2 O 2 HCl + [O]](http://slidetodoc.com/presentation_image/c3aaadfcb85791c8d900d48ea19fb125/image-94.jpg)
o Powerful bleaching agent. Cl 2 + H 2 O 2 HCl + [O] Coloured subs. + [O] Colourless subs.

Hydrogen Chloride Preparation 1. Lab method Na. OH+H 2 SO 4(conc. ) Na. HSO 4+HCl (420 K) Na. HSO 4 +Na. Cl Na 2 SO 4+HCl (823 K) HCl can be dried by passing through conc. H 2 SO 4.

Properties 1. Ionizes as HCl + H 2 O H 3 O+ + Cl 2. NH 3 + HCl NH 4 Cl 3. 3 conc. HCl +1 conc. HNO 3 =Aqua Regia It is used for dissolving platinum and gold.
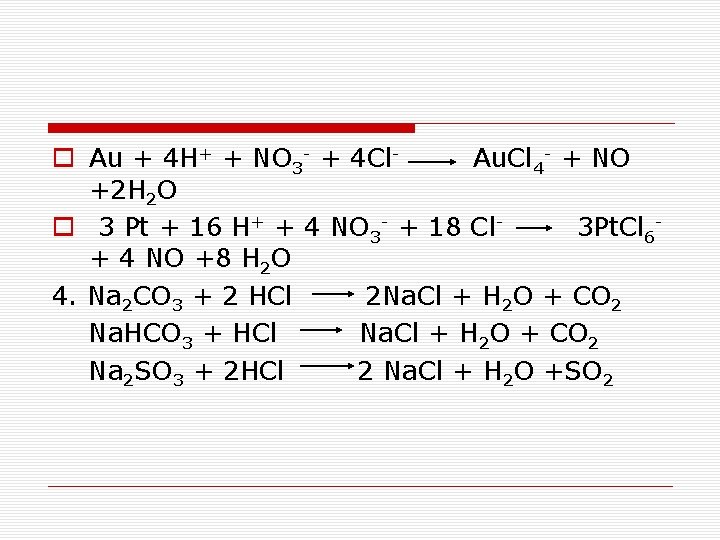
o Au + 4 H+ + NO 3 - + 4 Cl- Au. Cl 4 - + NO +2 H 2 O o 3 Pt + 16 H+ + 4 NO 3 - + 18 Cl- 3 Pt. Cl 6 - + 4 NO +8 H 2 O 4. Na 2 CO 3 + 2 HCl 2 Na. Cl + H 2 O + CO 2 Na. HCO 3 + HCl Na. Cl + H 2 O + CO 2 Na 2 SO 3 + 2 HCl 2 Na. Cl + H 2 O +SO 2

Oxoacids of Halogens Oxidation. State/Chlorine/Bromine/Iodine +1/HCl. O Hypochlorous acid/HBr. O Hypobromous acid/HIO Hypoiodous acid +3/HCl. O 2 Chlorous acid / - / +5/HCl. O 3 Chloric acid / HBr. O 3 Bromic acid/HIO 3 Iodic acid +7/HCl. O 4 Perchloric acid/HBr. O 4 Perbromic acid/HIO 4 Periodic acid

o Due to high electronegativity and small size , Fluorine forms only one oxoacid , HOF known as Fluoric (I) acid or Hypofluorous acid.
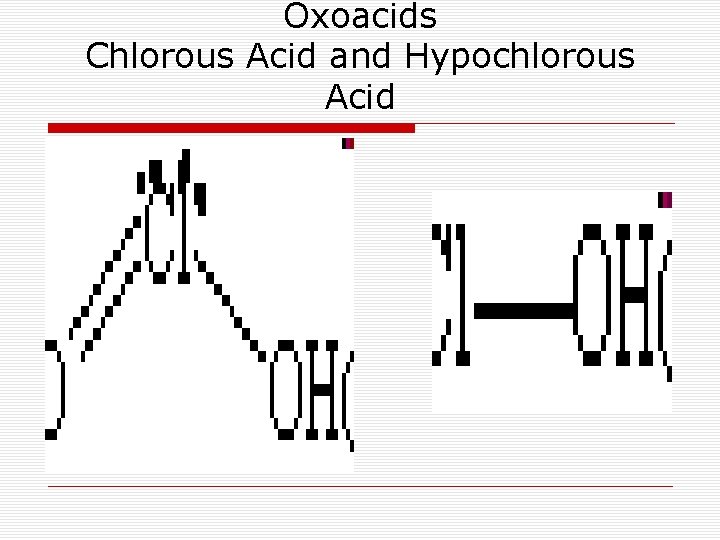
Oxoacids Chlorous Acid and Hypochlorous Acid
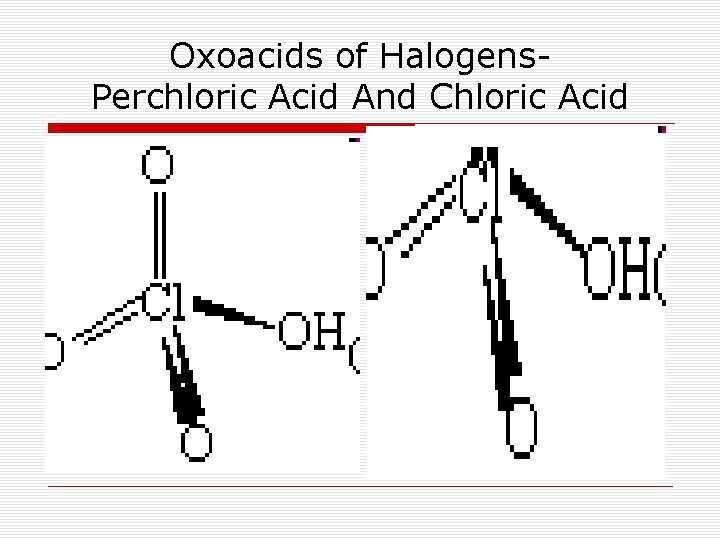
Oxoacids of Halogens. Perchloric Acid And Chloric Acid

Inter - Halogen Compounds Preparation 1. Equal volume Cl 2 + F 2 2 Cl. F (437 K) 2. Cl 2 in excess I 2 + 3 Cl 2 2 ICl 3 3. Excess F 2 Cl 2 + 3 F 2 2 Cl. F 3 ( 573 K )

4. Equimolar I 2 + Cl 2 2 ICl 5. Diluted with water Br 2 + 3 F 2 2 Br. F 3 6. Excess Fluorine Br 2 + 5 F 2 2 Br. F 5

Properties 1. Covalent compounds. 2. More reactive than the constituent halogen ( A-X bond is relatively weaker than X-X bond). 3. Diamagnetic in nature. 4. Good oxidising agent. 5. Melting and Boiling point increases with increase in difference in electronegativity.

Structure of Interhalogen Compounds o AX type: In AX type, A is the central atom and X is the bonded atom. In these type of compounds, both atoms share their electrons and form single covalent bond. A----X
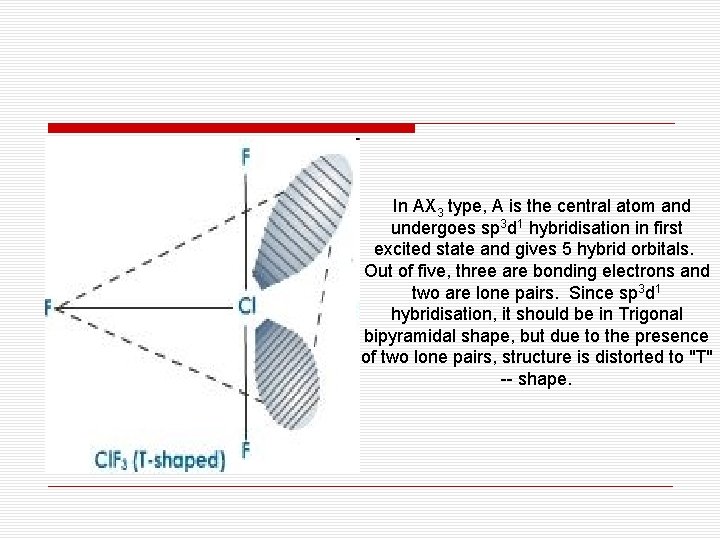
In AX 3 type, A is the central atom and undergoes sp 3 d 1 hybridisation in first excited state and gives 5 hybrid orbitals. Out of five, three are bonding electrons and two are lone pairs. Since sp 3 d 1 hybridisation, it should be in Trigonal bipyramidal shape, but due to the presence of two lone pairs, structure is distorted to "T" -- shape.
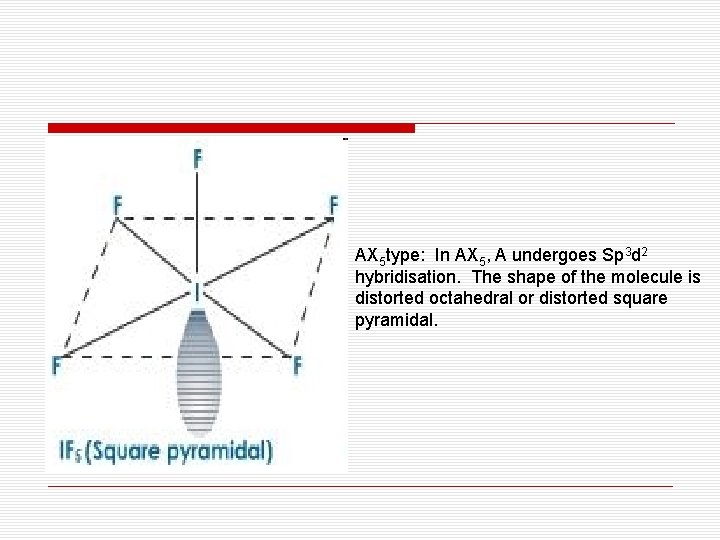
AX 5 type: In AX 5, A undergoes Sp 3 d 2 hybridisation. The shape of the molecule is distorted octahedral or distorted square pyramidal.
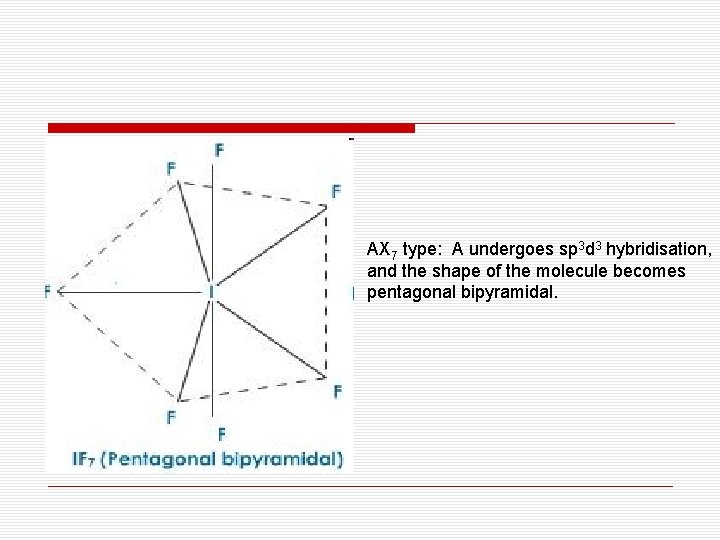
AX 7 type: A undergoes sp 3 d 3 hybridisation, and the shape of the molecule becomes pentagonal bipyramidal.

Group – 18 Elements o Electronic configuration- ns 2 np 6 o Atomic Radii- Increases down the group. o Ionisation Enthalpy- Very high. Decreases down the group. o Electron gain enthalpy- Do not accept electrons and have large +ve values of electron gain enthalpy. o M. P. and B. P. - Low melting and boiling points.

Chemical Properties 1. Least reactive. The inertness is due to: o Stable closed shell electronic configuration. o Exceptionally high ionisation enthalpy. o Very low electron affinities.

Xenon – Fluorine Compounds o Form 3 binary fluorides- Xe. F 2 , Xe. F 4 and Xe. F 6. o Xe (excess) + F 2 Xe. F 2(673 K, 1 bar, Ni tube) o 1: 5 ratio Xe +2 F 2 Xe. F 4(873 K, 7 bar, Ni tube) o 1: 20 ratio Xe +3 F 2 Xe. F 6( 573 K, 60 -70 bar, Ni )

o o o Xe. F 4 + O 2 F 2 Xe. F 6 + O 2 2 Xe. F 2 +2 H 2 O 2 Xe + 4 HF + O 2 Xe. F 2 +PF 5 [Xe. F]+[PF 6]Xe. F 4 +Sb. F 5 [Xe. F 3]+[Sb. F 6]Xe. F 6 + MF M+[Xe. F 7](M=Na, K, Rb, Cs)
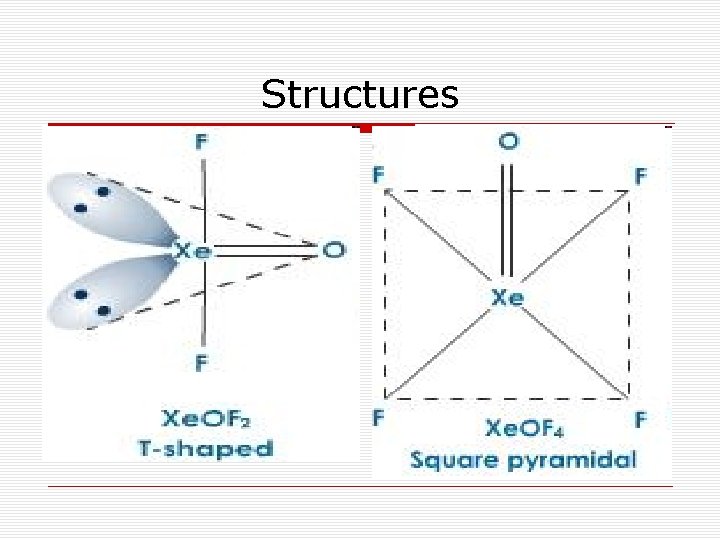
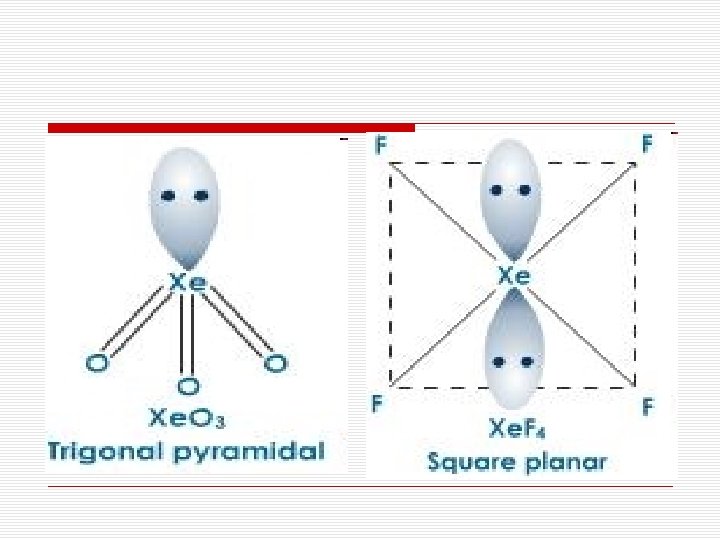
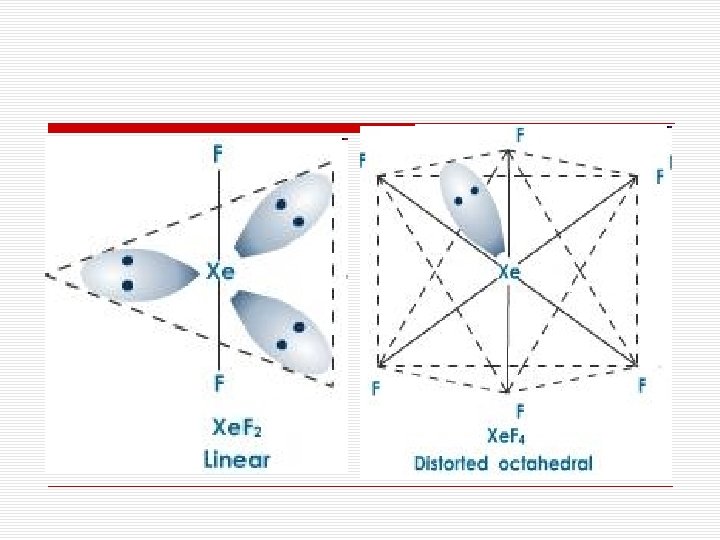
Structures
- Slides: 115