Patterns In Organic Alcohol Press space bar to
Patterns In Organic Alcohol Press <space bar> to start

Warning: Several of the chemicals in this lab are poisonous. Wash hands when finished. Press <space> to continue

The physical & chemical properties of molecules are based on their structure. These properties behave in predictable ways. For organic molecules, the properties (in part) depend on: - the length of the carbon chain - what functional groups are attached Press <space> to continue
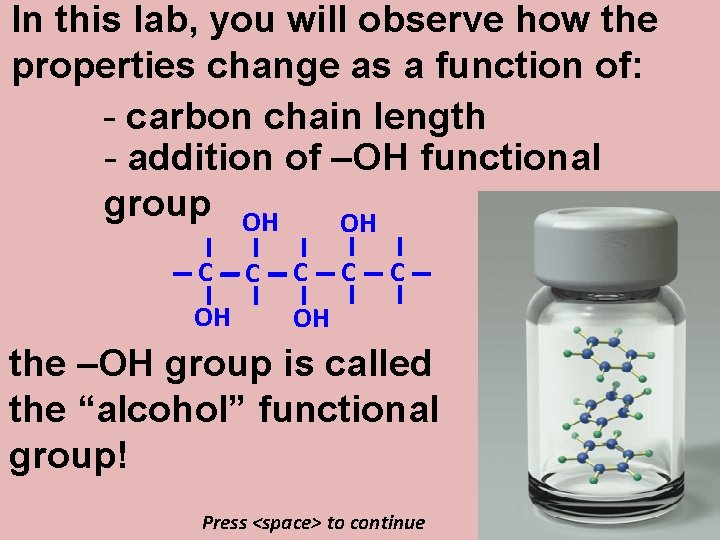
In this lab, you will observe how the properties change as a function of: - carbon chain length - addition of –OH functional group OH OH C C C C OH the –OH group is called the “alcohol” functional group! Press <space> to continue

You will be testing for volatility, viscosity, and combustion. Volatility is how quickly a substance evaporates. Gasoline & nail polish remover are examples of very volatile substances. Viscosity is resistance to flow. Viscous substances tend to be thick & syrupy. Honey has a high viscosity. Viscosity has units of c. P (centipoise). Press <space> to continue

The most simple alcohol is methanol. Its structure is based on methane: Methane is a flammable gas. However methanol is not a gas, but a liquid at room temp. ! Press <space> to continue
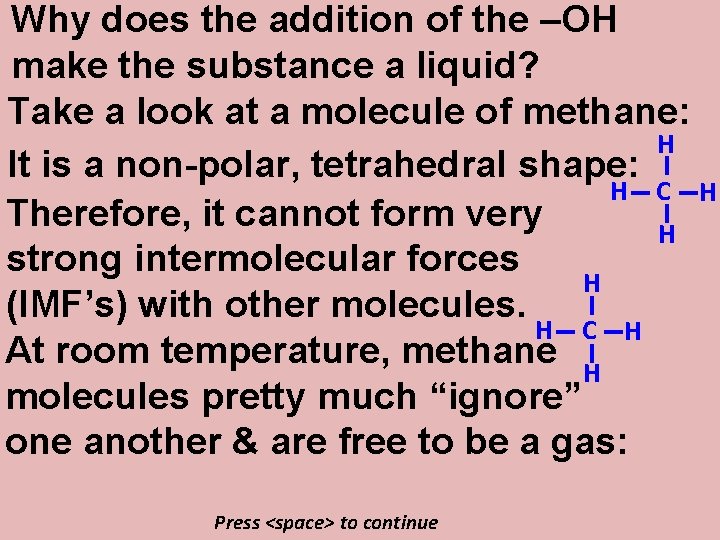
Why does the addition of the –OH make the substance a liquid? Take a look at a molecule of methane: H It is a non-polar, tetrahedral shape: H C H Therefore, it cannot form very H strong intermolecular forces H (IMF’s) with other molecules. H C H At room temperature, methane H molecules pretty much “ignore” one another & are free to be a gas: Press <space> to continue
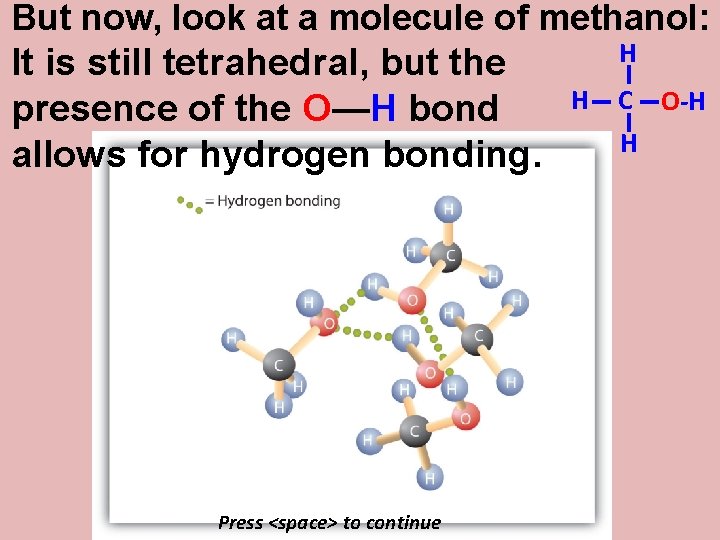
But now, look at a molecule of methanol: It is still tetrahedral, but the presence of the O—H bond allows for hydrogen bonding. Press <space> to continue H H C H O-H
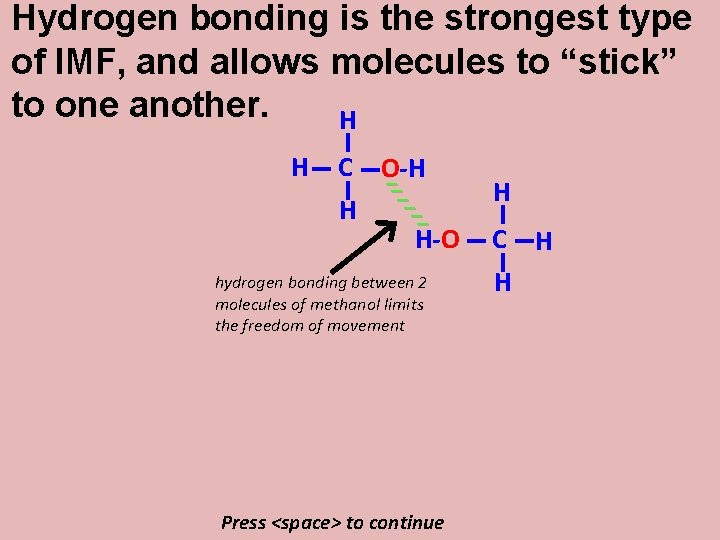
Hydrogen bonding is the strongest type of IMF, and allows molecules to “stick” to one another. H H C H O-H __ __ __ H-O hydrogen bonding between 2 molecules of methanol limits the freedom of movement Press <space> to continue H C H H

Watch how to perform the volatility test: Read all these instructions BEFORE you begin! 1) Wrap strip of filter paper around tip of thermometer & secure with plastic ring: 2) When stable, record initial temp on data sheet. 3) Add 5 drops of the alcohol to tip of filter paper. 4) Wait 20 seconds, & record temp on data sheet. 5) Replace the filter paper for each new test. 6) NOW, perform the test for each substance! Press <space> to continue

Watch how to perform the viscosity test: Read all these instructions BEFORE you begin! 1) Place 1 drop of substance on fingertip: 2) Rub liquid between fingers & feel the relative “thickness”, using chart below: 1 - very thin, like nail polish remover 2 - thin, like gasoline 3 - slightly thinner than water 4 - about the same as water 5 - slightly thicker than water (oily) 6 - thick, like syrup 7 - very thick, like honey 3) Record this value on your data sheet. NOW, perform the test for each substance! Press <space> to continue

Now, watch the teacher demo for the combustion test! Press <space> to continue

Methanol is a volatile alcohol used as race car fuel. It is also highly poisonous. 10 m. L can cause permanent blindness. 30 m. L can be fatal. Liver enzymes convert methanol into formic acid, which renders the body unable to use oxygen. Methanol is also called “wood alcohol”, because it was once obtained through distillation of wood. Press <space> to continue
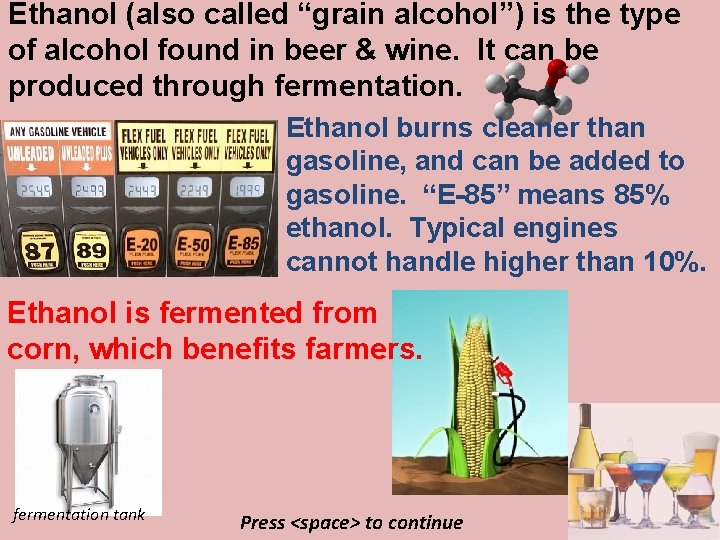
Ethanol (also called “grain alcohol”) is the type of alcohol found in beer & wine. It can be produced through fermentation. Ethanol burns cleaner than gasoline, and can be added to gasoline. “E-85” means 85% ethanol. Typical engines cannot handle higher than 10%. Ethanol is fermented from corn, which benefits farmers. fermentation tank Press <space> to continue

Ethanol is also the antidote for methanol poisoning. Ethanol reduces the formation of formic acid from methanol by “competitive inhibition”. Basically, the ethanol ties-up (or “inhibits”) the enzyme, so the methanol can be excreted from the body. In the US, a liquor license is required to sell consumable alcohol. Hardware stores & chemical suppliers sell “denatured” alcohol without needing a license. Denatured means it has been made non-drinkable by adding methanol. Press <space> to continue

1, 2 -ethandiol is commonly known as ethylene glycol. Ethylene glycol is automotive anti-freeze. The additional –OH group adds an additional site for hydrogen bonding, and therefore increases the viscosity. Ethylene glycol is very toxic. Liver enzymes convert it into oxalic acid, which causes kidney failure. The antidote is the same as for methanol poisoning, and it works for the same reason: competitive inhibition Press <space> to continue

2 - propanol is also known as isoproply alcohol. It is relatively inexpensive, and has several common household uses: • cleaning electronic components • removing gluey residue from stickers • removes “permanent” ink from many surfaces • surface disinfectant on cuts & wounds • cleans smudges from DVD’s Press <space> to continue

1, 2, 3 -propantriol is better known as glycerin (or glycerol). Its three –OH groups makes it very viscous, but also water soluble. Its many uses include: • a sweetener in food • makes cough syrup smoother • certain types of soaps • laxative glycerin soap Press <space> to continue

1 -butanol, or n-butanol (n for “normal”) Used in perfumes, paint solvent, and degreasers. Also found naturally in small quantities in certain foods. Butanol has potential as a biofuel & a gasoline alternative. Butanol fueled car in St. Louis. Press <space> to continue

Viscosity is measured in c. P, or centipoises. For reference, water at room temperature is 1. 0 c. P. has one –OH group: 1 carbon: methanol = 0. 59 c. P 2 carbons: ethanol = 1. 0 c. P 3 carbons: 2 -propanol = 2. 0 c. P 4 carbons: 1 -butanol = 3. 0 c. P has several –OH groups: 1, 2 -ethandiol = 16. 1 c. P 1, 2, 3 -propantriol = 1400 c. P Press <space> to re-start
- Slides: 20