Patricia Deverka MD MS MBE Center for Medical

Patricia Deverka, MD, MS, MBE Center for Medical Technology Policy Emerging and New Technologies May 8, 2012

CAP POLICY MEETING 2012 EMERGING AND NEW TECHNOLOGIES THE IMPACT ON PERSONALIZED HEALTH CARE Patricia Deverka MD, MS May 8, 2012

My role today • Engage in a dialogue about barriers and facilitators of clinical integration of new genomic tests • Provide response to CAP policy initiatives in this area – From perspective of public health, CER, stakeholder engagement • Emphasize the need for better evidence of clinical utility for patient, payer and clinical decision-making • Suggest several ways that pathologists can play a major role in addition to current CAP policy initiatives • Learn from all of you

Impressions • Clearly pathologists and CAP have had and will continue to have a leadership role in genetic testing • Concur that there is uncertainty in regulatory, reimbursement and reporting environments – Lack of evidence re how to make test results clinically actionable and whether use of the test improves net health outcomes for patients is major stumbling block for reimbursement and clinical uptake • Current analysis is weighted toward technology-related factors (costs, speed, complexity) but decision-makers tend to use similar evidentiary frameworks for all MDx tests – Assuming that they are all moderate to high risk • Solutions will require participation by all stakeholders and novel partnerships
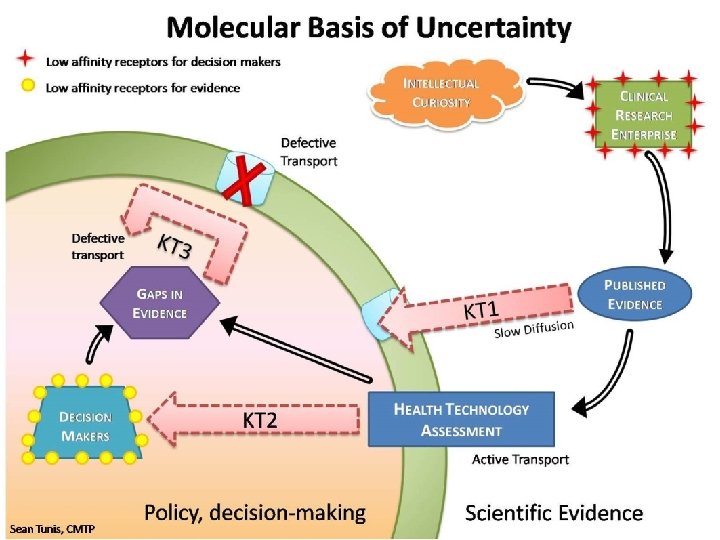
5

Center for Medical Technology Policy (www. cmtpnet. org) • A private, non-profit organization that provides a trusted forum in which patients, clinicians, payers, manufacturers, researchers and other stakeholders can work together to develop strategies that improve the quality of clinical research for healthcare decision making design and implement prospective, real world studies to inform health care decisions. • Major goal is to improve process of generating reliable and credible information about the real-world risks, benefits and costs of promising new medical technologies • Areas of Focus – – – Comparative effectiveness research Stakeholder engagement Methods development Symposia (policy and methods) Public-private partnerships

EGAPP Working Group (Teutsch et al, Genet Med, 2008) • “Of most concern, the number and quality of studies are limited. Test applications are being proposed and marketed based on descriptive evidence and pathophysiologic reasoning, often lacking well-designed clinical trials or observational studies to establish validity and utility, but advocated by industry and patient interest groups”

Clinical Utility of a Genomic Test • Some genomic tests are based only on a statistical association of the test result with the health outcome of interest (clinical validity) • Genomic tests are useful when they have been evaluated to show clinical utility. • Clinical utility is demonstrated when use of a genetic test is shown to improve health outcomes, such as length of life, quality of life or functional ability. • Without information on clinical utility, it is not possible to know which tests are of benefit to patients and which are not

Definitions • Analytic Validity: the ability of a genetic test to accurately and reliably measure the genotype (or analyte) of interest in the clinical lab and in specimens that are representative of the population of interest • Clinical Validity: The ability of a genetic test to accurately and reliably predict the clinically defined disorder or phenotype of interest • Clinical Utility: The improved measurable clinical outcomes shown by a genetic test and the usefulness and added value to patient management resulting from a test, compared with current patient management without genetic testing
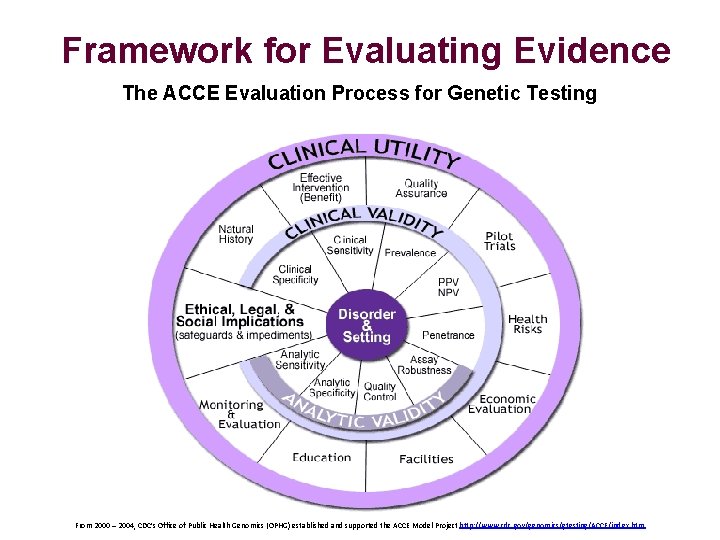
Framework for Evaluating Evidence The ACCE Evaluation Process for Genetic Testing From 2000 – 2004, CDC’s Office of Public Health Genomics (OPHG) established and supported the ACCE Model Project http: //www. cdc. gov/genomics/gtesting/ACCE/index. htm
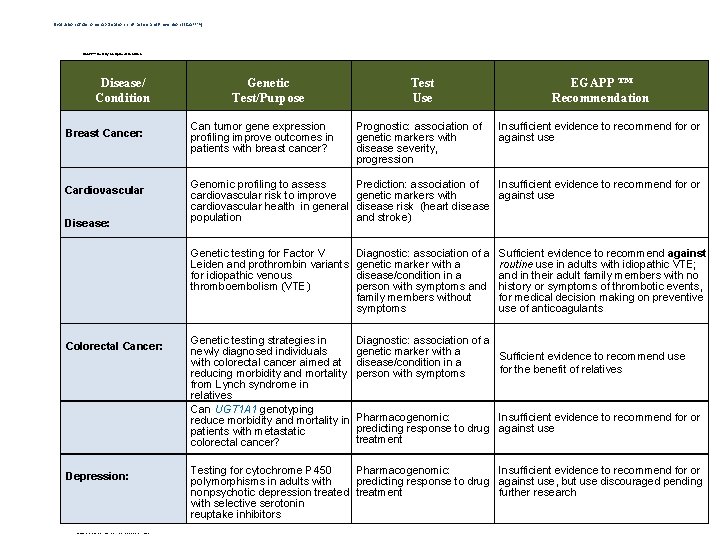
Evaluation of Genomic Applications in Practice and Prevention (EGAPPTM) EGAPP™ Working Group Recommendations Disease/ Condition Breast Cancer: Cardiovascular Disease: Colorectal Cancer: Depression: • © 2009 Evaluation of Genomic Applications in Practice and Prevention (EGAPP) Genetic Test/Purpose Test Use EGAPP ™ Recommendation Can tumor gene expression profiling improve outcomes in patients with breast cancer? Prognostic: association of genetic markers with disease severity, progression Insufficient evidence to recommend for or against use Genomic profiling to assess cardiovascular risk to improve cardiovascular health in general population Prediction: association of Insufficient evidence to recommend for or genetic markers with against use disease risk (heart disease and stroke) Genetic testing for Factor V Leiden and prothrombin variants for idiopathic venous thromboembolism (VTE) Diagnostic: association of a genetic marker with a disease/condition in a person with symptoms and family members without symptoms Genetic testing strategies in newly diagnosed individuals with colorectal cancer aimed at reducing morbidity and mortality from Lynch syndrome in relatives Can UGT 1 A 1 genotyping reduce morbidity and mortality in patients with metastatic colorectal cancer? Diagnostic: association of a genetic marker with a Sufficient evidence to recommend use disease/condition in a for the benefit of relatives person with symptoms Sufficient evidence to recommend against routine use in adults with idiopathic VTE; and in their adult family members with no history or symptoms of thrombotic events, for medical decision making on preventive use of anticoagulants Pharmacogenomic: Insufficient evidence to recommend for or predicting response to drug against use treatment Testing for cytochrome P 450 Pharmacogenomic: Insufficient evidence to recommend for or polymorphisms in adults with predicting response to drug against use, but use discouraged pending nonpsychotic depression treated treatment further research with selective serotonin reuptake inhibitors

BCBSA Criteria • The scientific evidence must permit conclusions concerning the effect of the technology on health outcomes. – The evidence should consist of well-designed and well-conducted investigations published in peer-reviewed journals. – The evidence should demonstrate that the technology can measure or alter the physiological changes related to a disease, injury, illness, or condition. In addition, there should be evidence or a convincing argument based on established medical facts that such measurement or alteration affects health outcomes.
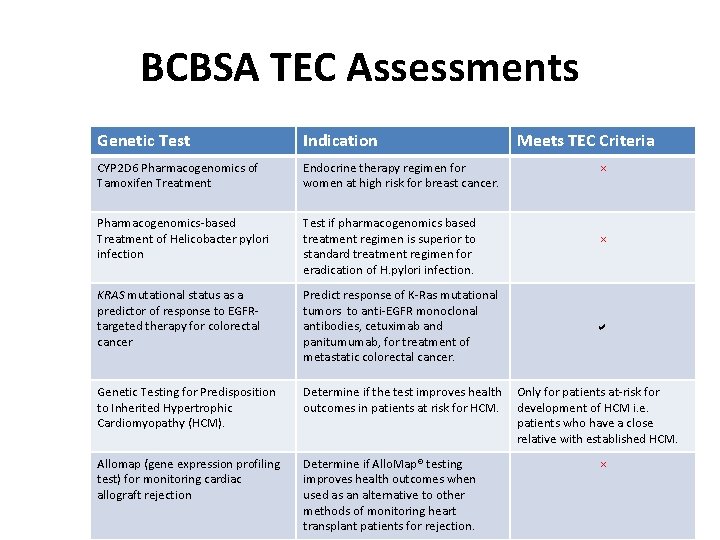
BCBSA TEC Assessments Genetic Test Indication Meets TEC Criteria CYP 2 D 6 Pharmacogenomics of Tamoxifen Treatment Endocrine therapy regimen for women at high risk for breast cancer. Pharmacogenomics-based Treatment of Helicobacter pylori infection Test if pharmacogenomics based treatment regimen is superior to standard treatment regimen for eradication of H. pylori infection. KRAS mutational status as a predictor of response to EGFRtargeted therapy for colorectal cancer Predict response of K-Ras mutational tumors to anti-EGFR monoclonal antibodies, cetuximab and panitumumab, for treatment of metastatic colorectal cancer. Genetic Testing for Predisposition to Inherited Hypertrophic Cardiomyopathy (HCM). Determine if the test improves health Only for patients at-risk for outcomes in patients at risk for HCM. development of HCM i. e. patients who have a close relative with established HCM. Allomap (gene expression profiling test) for monitoring cardiac allograft rejection Determine if Allo. Map® testing improves health outcomes when used as an alternative to other methods of monitoring heart transplant patients for rejection. × × a ×
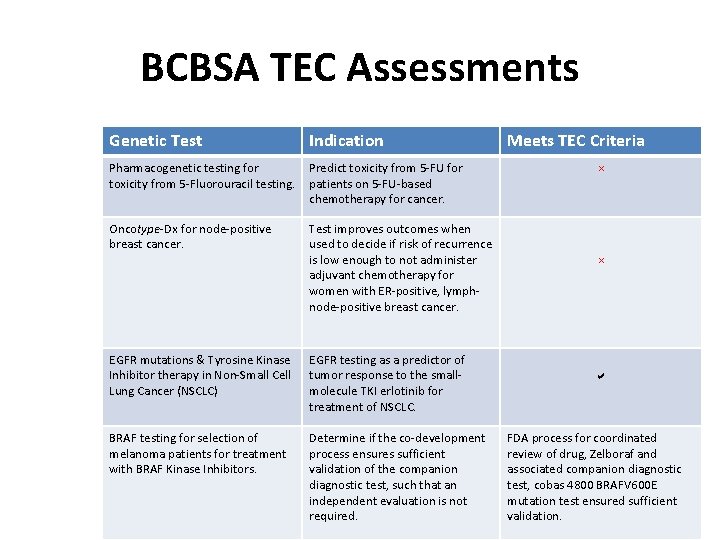
BCBSA TEC Assessments Genetic Test Indication Pharmacogenetic testing for Predict toxicity from 5 -FU for toxicity from 5 -Fluorouracil testing. patients on 5 -FU-based chemotherapy for cancer. Oncotype-Dx for node-positive breast cancer. Test improves outcomes when used to decide if risk of recurrence is low enough to not administer adjuvant chemotherapy for women with ER-positive, lymphnode-positive breast cancer. EGFR mutations & Tyrosine Kinase Inhibitor therapy in Non-Small Cell Lung Cancer (NSCLC) EGFR testing as a predictor of tumor response to the smallmolecule TKI erlotinib for treatment of NSCLC. BRAF testing for selection of melanoma patients for treatment with BRAF Kinase Inhibitors. Determine if the co-development process ensures sufficient validation of the companion diagnostic test, such that an independent evaluation is not required. Meets TEC Criteria × × a FDA process for coordinated review of drug, Zelboraf and associated companion diagnostic test, cobas 4800 BRAFV 600 E mutation test ensured sufficient validation.

Reasons for lack of CU data • Lack of formal regulatory requirements for test manufacturers • Field is changing so rapidly, evidence becomes quickly outdated • Academic research community has not prioritized CU studies • Lack of incentives for private sector investment in expensive studies because of lack of value-based reimbursement • Existing paradigms for generating and evaluating evidence are too slow, costly, unrepresentative to be useful to decisionmakers in a timely manner

The CER/PCOR Hypothesis • Gaps in evidence will be reduced with greater engagement of end users (e. g. , patients, clinicians, policymakers, payers) in – Selecting research questions – Developing study protocols / methods – Implementing research • Successful engagement will lead to more useful research because it deliberately attempts to answer the questions of most relevance to end-users, rather than primarily investigators or funders

PCORI Methods Committee View JAMA. 2012; 307(15): 1636 -1640 • “Engagement of patients at every step of the research process is viewed as essential, including in the selection of research questions, study design, conduct, analysis, and implementation of findings. ” • “As such, the Methodology Committee is engaged in developing standards to support the validity and generalizability of research, as well as patient-centeredness. ”

How will genetic testing be involved in CER? • Top 25 CER priorities recommended by IOM – Compare the effectiveness of genetic and biomarker testing and usual care in preventing and treating breast, colorectal, prostate, lung, and ovarian cancer, and possibly other clinical conditions for which promising biomarkers exist • As the subject of analysis for head-to-head comparisons of alternative laboratory tests or comparisons of a lab test to another test intervention for a particular health care condition

Methods for CER (including CU) • Systematic reviews (not timely and often limited by missing data) • Explanatory RCTs • Pragmatic clinical trials • Bayesian or adaptive trials • Observational studies • Decision modeling and health economics • CED

CED Definition and Purpose • Coverage contingent on participation in clinical study – Reconciles tension between desire for CER evidence and rapid access to innovation • When evidence limited, payers in relatively poor position to restrict access to technology • Evidence is almost always “limited” for new tech – Allows for CMS views on relevant patients, comparators, outcomes, design

CED and Genomic Diagnostics • CED provides coverage contingent on participation in a clinical study (clinical trial, registry, etc) – For diagnostic tests, evidence of impact on health outcomes may not be feasible for initial coverage – However, unconditional coverage significantly reduces incentives to confirm health impacts – Optimal public health benefits from genomic diagnostics may be achieved through initial coverage at clinical validity, studies of clinical utility under CED

SACGHS recommendation • “Information on clinical utility is critical for managing patients, developing professional guidelines, and making coverage decisions. ” • “HHS should create a public private entity of stakeholders to…. establish evidentiary standards and levels of certainty required for different situations”

Guidance for CER: “Effectiveness Guidance Documents” • • • Provide specific recommendations on the design of studies intended to inform decisions by patients, clinicians and payers Aim to achieve balance between internal validity, relevance and feasibility in order to provide decisionmakers with a reasonable level of confidence that the intervention improves net health outcomes Target clinical researchers – Working in industry or academic settings Stakeholders involved throughout the process Intended to be analogous and complementary to FDA guidance documents, but focused on design elements of particular relevance to clinical and health policy decision -making

Potential next steps • Support efforts to generate evidence of clinical utility – RCTs will never address all of the questions – LDTs will still exist • Gather real-world data – Document gaps in knowledge about actual clinical practice – Eventually new codes should facilitate data collection • Standardize testing procedures and interpretation – Use of decision support algorithms – Patient-centered approaches to ensure informed decision-making • Develop collaborations between academia, industry and government to expand the evidence base – Pathology should help inform the priority-setting, data collection, study design and results interpretation

Contact Info • • Pat. deverka@cmtpnet. org www. cmtpnet. org 410 547 2687 x 136 (W) 919 491 0225 (M)
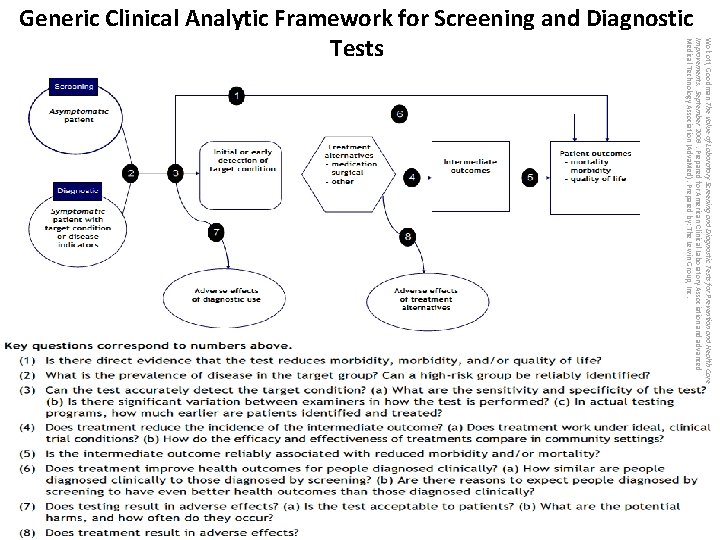
Wolcott, Goodman The Value of Laboratory Screening and Diagnostic Tests for Prevention and Health Care Improvements. September 2009. Prepared for American Clinical Laboratory Association and advanced Medical Technology Association (Adva. Med). Prepared by: The Lewin Group, Inc. Generic Clinical Analytic Framework for Screening and Diagnostic Tests

FDA Regulation of IVD • reasonable assurance that the probable benefits outweigh any probable risks – 21 CFR 860. 7(d)(1) • reasonable assurance that the use of the device will provide clinically significant results – 21 CFR 860. 7(e)(1)

Medicare guidelines for evaluation of dx tests (clinical utility) • Question 1: Is the evidence adequate to determine whether the test provides more accurate diagnostic information? • Question 2: If the test changes accuracy, is the evidence adequate to determine how the changed accuracy affects health outcomes?

- Slides: 29