Patient safety dispensing incidents and learnings Leyla Hannbeck

Patient safety & dispensing incidents and learnings Leyla Hannbeck MSc. MRPharm. S Chief Pharmacist Twitter: @Leyla. Hannbeck

MSO work • Over 200 reports are being sent to the NPA on a monthly basis • We work closely with patient safety team at NHS Improvement – they also bring incidents to our attention • Quarterly reports analysing the reported incidents and learning have been sent regularly to superintendents • A webinar took place summarising the dispensing and prescribing incidents reported to the NPA • All updates and resources are on the NPA website

Two most common types of error • Wrong drug/medicine – 31% of reported errors • Wrong/unclear dose or strength – 28% of reported errors

Look alike/sound alike drugs • The same drugs feature in dispensing errors again and again – including: – Allopurinol/amiloride/ amisulpiride/amitriptyline/ Serious incident resulting in amlodipine/atenolol hospitalisation of patient – The label on – Chlorphenamine/chlorpromazine/clomipramine/ the box of tablets was for ranitidine, but clonazepam/clonidine the actual box of tablets was quetiapine. – Pantoprazole/paroxetine/pravastatin/ prednisolone/prochlorperazine/propranolol – Ropinirole/risperidone • Be aware of these drugs!!

Wrong strength errors – common drugs • • • Allopurinol Amlodipine Amoxicillin Atenolol Atorvastatin Bisoprolol Butrans patch Co-codamol Colecalciferol Furosemide • • • Gabapentin Lansoprazole Levothyroxine Metformin Mirtazapine Pregabalin Ramipril Sertraline Simvastatin Warfarin

Other types of error reported • • • Wrong formulation – 10% Wrong quantity – 7% Mismatching between patient and medicine – 5% Adverse drug reaction – 3% Wrong/omitted/passed expiry date – 2% Wrong frequency – 2% Omitted medicine/ingredient – 1% Wrong/transposed/omitted medicine label – 1% Contraindication to use of medicine – 1% Wrong/omitted verbal patient directions – <1% Wrong method of preparation/supply – <1% Other – 8%

Examples of wrong formulation errors • Aspirin dispersible tablets/enteric coated tablets • Insulin cartridges/prefilled pens • Metformin modified release tablets/ standard release tablets • Ramipril tablets/capsules • Salbutamol metered dose inhalers/breath actuated inhalers • Venlafaxine tablet/modified release tablet

Top 3 important factors in errors • 56% – medicines with a similar looking or sounding name • 14% – poor transfer/ transcription of information between paper and/or electronic forms • 12% – poor labelling and packaging from a commercial manufacturer

Most common contributing factor • For reports that identified contributing factors, 58% selected “work and environment factors” o This category includes poor/excess administration, physical environment, work load, hours of work and time pressures o Examples reported: o “Pharmacist blamed poor decision on excess pressure” o “I believe the environment was busy and chaotic” o “New systems, busy branch”

Errors that have resulted in claims being made against the pharmacist

Methotrexate: wrong frequency • Intended dose of methotrexate 15 mg (6 x 2. 5 mg tablets) once weekly: – Labelled as 15 mg (6 x 2. 5 mg tablets) daily – Patient hospitalised • Key learning points: – Methotrexate is usually taken weekly and fatalities have occurred when accidentally taken daily – Always double check methotrexate dose and frequency and ensure patient understands dosing regimen

Prednisolone: wrong strength • Intended dose 6 x 5 mg tablets daily: – Dose dispensed (and taken) 6 x 25 mg daily – Patient hospitalised with gastritis • Key learning points: – Errors can occur at prescribing or dispensing stage – Always check dose of prednisolone is in appropriate range and that correct strength tablets have been dispensed

Prednisolone: wrong drug • Patient prescribed prednisolone tablets – 6 once daily: – Patient received propranolol in error and took 6 tablets – Patient hospitalised with cardiac symptoms • Key learning points: – All ‘wrong drug’ errors can have serious consequences – risk is increased when prednisolone is the intended drug due to multiple tablet dosing – Always take time to thoroughly check prescriptions for prednisolone

Ranitidine oral solution: children • Ranitidine oral solution 150 mg/10 ml prescribed with a dose “ 5 mg to be taken” – Ranitidine oral solution 150 mg/10 ml supplied and labelled “ 5 ml to be taken” – Dose of 75 mg was administered instead of 5 mg • Key learning points: – Always take extra care with doses for children – Always supply a suitable oral syringe or spoon as appropriate and ensure parent/carer understands how to use it to administer the correct dose

Warfarin incident • Can have serious consequences • Examples of warfarin incidents that can occur: – Correct strength prescribed but incorrect strength dispensed – Incorrect strength prescribed and dispensed – Interaction with other prescribed medicine or OTC product • Key learning points: – Double check strength! – Always ask to see warfarin book or INR print-out to confirm recent INR test result and correct dose – Check all drug interactions with warfarin, including products sold OTC – Follow SOP for dispensing warfarin

Prescribing errors • 100 mg MST for a opiate naive patient should have been 10 mg – Patient was hospitalised • Key learning points: For all opioids: - Familiarise yourself with the dosing information for commonly dispensed opioid formulations and be aware of symptoms of overdose and common side effects - Always check recent history of opioid use - Always check dose is appropriate - Remember to take special care when patients are: o starting a strong opioid for the first time o being switched from one formulation to another o being switched from one opioid to another o using more than one opioid concurrently o having their dose increased

How to prevent errors: dispensing best practice • New NPA resource “Dispensing process: best practice” launched in July 2016 – Intended to help you review your standard operating procedures (SOPs) covering the dispensing process – Sets out best practice to minimise the risk of dispensing errors – Covers 8 stages of the dispensing process, with special guidance for monitored dosage systems and controlled drugs
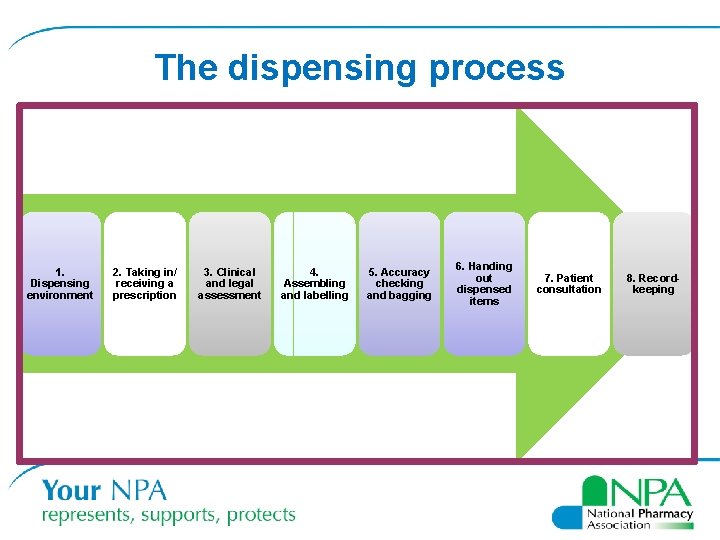
The dispensing process 1. Dispensing environment 2. Taking in/ receiving a prescription 3. Clinical and legal assessment 4. Assembling and labelling 5. Accuracy checking and bagging 6. Handing out dispensed items 7. Patient consultation 8. Recordkeeping

1. Dispensing environment • Have clear signage • Minimise distractions such as background noise • Promote a dispensary atmosphere that encourages good concentration • Have designated areas for different stages of dispensing process • Baskets/trays for individual patients • Ensure daily/weekly/monthly checks are carried out as per SOPs

2. Taking in/receiving prescriptions • Check whether customer presenting prescription is patient or representative • Confirm patient details – name, address, age or date of birth as appropriate – and clarify unclear details where necessary – Clarifying details can prevent errors caused by mismatching patient and medicine • Advise customer of approximate waiting time – This can help reduce time pressure on dispensing staff by managing the patient’s expectations

3. Clinical and legal assessment • Take into account the following when assessing the prescription for clinical appropriateness: – – Age, sex, weight of patient Pregnant or breastfeeding Relevant clinical condition(s) Information on the Patient Medication Record (PMR) / Summary Care Record (SCR), if appropriate – Interactions with other drugs (including those bought over the counter), foods, supplements or disease states – Dose and dose changes, strength and strength changes, formulation, route of administration, contra-indications – New medicines

3. 1 - Clinical and legal assessment • Where, in your professional judgement, the prescription may not be clinically appropriate take steps to obtain additional information in order to come to a final decision. Consider: – Discussing issues with the prescriber and/or patient – Checking appropriate reference materials (e. g. British National Formulary (BNF), Martindale, Stockley’s Drug Interactions etc) – Contacting the NPA Pharmacy Services team on 01727 891800

3. 2 - Clinical and legal assessment • Where, in your professional judgement, the prescription is not legally valid and/or clinically appropriate then inform the patient or representative of the decision, and the rationale, and refer them back to the prescriber or liaise with the prescriber directly • Remember – this is your opportunity to spot prescribing errors, inappropriate doses, strength changes etc which could all result in a patient safety incident

4. Assembling and labelling • Using the actual prescription (or EPS 2 dispensing/prescription token), carefully select the medicine(s), checking the name, strength, formulation and quantity for each item and place into a dispensing basket/tray • Check the medicine(s) are within the expiry date • Check if patient has a PMR record, carefully crosschecking address and date of birth to ensure that the correct patient is selected

4. 1 - Assembling and labelling • If generating labels using the existing PMR, check that the following have not changed and, where they have, amend as applicable: – Dose, form, strength, quantity • Carefully label each item one at a time, checking the name, strength, form and quantity • Apply the label to the space designed by the manufacturer for this purpose if applicable – otherwise be careful not to cover up the name or strength or other important information

4. 2 - Assembling and labelling • The GPh. C guidance “Responding to complaints and concerns, September 2010” advises to “produce dispensing labels before any product is selected from the shelf” • However, it is the responsibility of the pharmacy owner /superintendent pharmacist, to ensure that all pharmacy SOPs reflect best practice, taking into account the workflow in the pharmacy premises, and ensuring safe and effective dispensing while minimising risk of errors

5. Accuracy checking and bagging • Read the whole prescription and check each item individually in the order that it appears on the prescription • For multiple packs, check each pack • Cross check the labels(s) and medicines against the prescription, and each other for the following, where applicable:

5. 1 - Accuracy checking &bagging – Name of the medicine – be careful with drug names which look or sound similar – Strength of the product – be careful with the units, for example, mg and mcg – Form of the product – Quantity of the product – a physical inspection of the actual quantity of the product should be conducted wherever reasonably possible for unsealed solid formulations – for CDs the quantity must be checked

5. 2 - Accuracy checking &bagging • For medicines dispensed from bulk packs, visually check that the contents of the stock pack match the contents of the dispensed product • Check the expiry date on each of the patient packs or the stock pack • For liquid formulations, add an appropriate plastic spoon or oral syringe • Ensure that stock packs are not placed into the patient’s bag

5. 3 - Accuracy checking & bagging 4 • Where the check has revealed an error, alert the colleague who dispensed the medicines to the error and make an entry into the near miss log; correct the error • The accuracy check is your opportunity to identify and correct any dispensing errors – do not rush it!

6. Handing out dispensed items • Request the prescription receipt stub; where this is not available: – Locate the correct bag (if not with you already) and check the customer’s first name and surname against the details on the sealed bag • Check the sealed bag for any prescription alert stickers or notes which might indicate that: – The pharmacist wishes to speak to the patient and hand over the medicines personally – An additional CD accompanies the sealed bag – A fridge item accompanies the sealed bag

6. 1 - Handing out dispensed items • Cross-check the patient’s first name, surname and address against the details on the docket stub (if used), the prescription and the label on the bag containing the dispensed medicines • Confirm the patient’s first name, and surname and address before handing over to the customer • Check if the customer has any questions about their medicines; if so, follow the procedures outlined in the relevant pharmacy SOP for pharmacy staff, referring to the pharmacist where appropriate

Monitored dosage systems (MDS) • MDS trays should be dispensed in a designated area • Specific MDS SOPs must be in place, particularly where “at risk” medicines are included, such as warfarin (these should only be included in MDS in exceptional circumstances) • Ensure that each MDS is assembled and labelled for one patient at a time – assembling for multiple patients simultaneously can lead to errors

Dispensing process: best practice • The previous slides show a selection of key points relating to patient safety • The full resource can be accessed from the NPA website: https: //www. npa. co. uk/wpcontent/uploads/2016/07/Dispensingprocess-best-practice-July-2016. pdf

Final points. . • Remember to supply patient information leaflet • Check the dose of the prescription is suitable for patient • Be empathetic towards the patient and apologies if you have made an error – patient is less likely to put in a complaint or claim against you • Remember to supply relevant syringe or spoons for kids doses • Follow the protocol when supplying EHC – respect patient confidentiality • If you get a letter or complaint notify the NPA so we can support you and advise you on how to respond
- Slides: 35