patient s A Outcome assessment B Outcome assessment












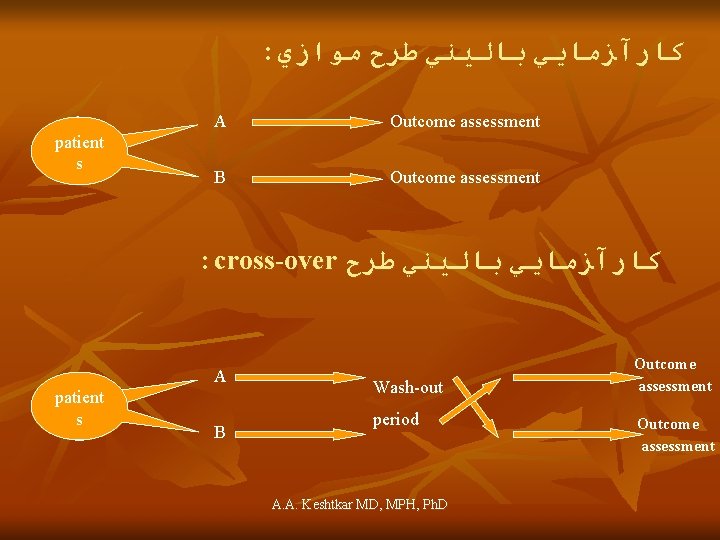
: ﻛﺎﺭآﺰﻣﺎﻳﻲ ﺑﺎﻟﻴﻨﻲ ﻃﺮﺡ ﻣﻮﺍﺯﻱ patient s A Outcome assessment B Outcome assessment : cross-over ﻛﺎﺭآﺰﻣﺎﻳﻲ ﺑﺎﻟﻴﻨﻲ ﻃﺮﺡ A patient s B Wash-out period A. A. Keshtkar MD, MPH, Ph. D Outcome assessment


Goals and objectives of Clinical Trials A. A. Keshtkar MD, MPH, Ph. D

The ultimate goal of Clinical Trials n Obtaining an unbiased inference with possibly best precision in order to scientifically address the clinical questions regarding the study drug / intervention under investigation with respect to a target patient population A. A. Keshtkar MD, MPH, Ph. D

Most common mistake in the goal setting: The investigator(s) often attempts to answer all possible questions with respect to a certain therapeutic area in a single trial regardless the size of the trial. A. A. Keshtkar MD, MPH, Ph. D

Example: effect of test drug on postmenopausal osteoporosis Primary Objectives: 1. This trial is a randomized, double-blind, placebocontrolled trial conducted in x centers to evaluate the efficacy based on bone mineral density (BMD) of the test drug under investigation at dose y, frequency z, compared to a placebo, in the treatment of postmenopausal women with osteoporosis. 2. This trial is a randomized, double-blind, placebocontrolled trial conducted in x centers to evaluate the safety of the test drug under investigation at dose y, frequency z, compared to a placebo, in the treatment of postmenopausal women with osteoporosis. A. A. Keshtkar MD, MPH, Ph. D

Example: effect of test drug on postmenopausal osteoporosis Secondary Objectives: 1. To evaluate the effectiveness of the test drug under investigation on the incidence of vertebral fractures. 2. To evaluate the effectiveness of the test drug under investigation on biochemical markers of bone turnover. A. A. Keshtkar MD, MPH, Ph. D

Example: 1 year survival following non small cell lung cancer treatment Primary Objectives: This is a randomized, parallel-group trial to demonstrate that the one-year survival of the patients with pretreated advanced (Stage IIIB/IV) non-small-cell lung cancer (NSCLC) receiving the oral investigational drug is not inferior to those receiving intravenous (IV) docetaxel. A. A. Keshtkar MD, MPH, Ph. D

Example: 1 year survival following non small cell lung cancer treatment Secondary Objectives: evaluate overall survival, time to progression, response rate, time to response, improvement in quality of life, and qualitative and quantitative toxicities. A. A. Keshtkar MD, MPH, Ph. D

Target population and Patients selection A. A. Keshtkar MD, MPH, Ph. D

Eligibility criteria: Inclusion & Exclusion criteria n n n patients must meet all the inclusion criteria and patients meeting any of the exclusion criteria will be excluded from the study. The strict criteria increases relaibility and decreases generalizability of study results. The inclusive criteria increases generalizability and confounds study results. A. A. Keshtkar MD, MPH, Ph. D

Example: RCT of clinical and microbiologic efficacy and safety of an antibiotic agent in the treatment of febrile episodes in neutropenic cancer patients A. A. Keshtkar MD, MPH, Ph. D

Single study site vs Multicenter study sites n n To recruit enough number of patients and to complete the study within the time frame, as an alternative, a multicenter trial is usually considered How many study sites should be used? n As a rule of thumb, the number of sites should not be greater than the number of patients within each selected study site A. A. Keshtkar MD, MPH, Ph. D

Selection of Control Group in Clinical Trials A. A. Keshtkar MD, MPH, Ph. D

Two main options for selecting control group: n n concurrent control group as one chosen from the same population as the test group external or historical control is chosen separated from the target patient population treated in the same trial A. A. Keshtkar MD, MPH, Ph. D

4 types of treatment that a patient can receive in the control group: (1) placebo (2) no treatment (3) different dose or regimen of the test drug, (4) different active treatments A. A. Keshtkar MD, MPH, Ph. D

Different Designs of Clinical Trials A. A. Keshtkar MD, MPH, Ph. D

Parallel Group Design Definition: a parallel group design is a complete randomized design in which each patient receives one and only one treatment in a random fashion two types of parallel group design: • group comparison (or parallel-group) designs • matched pairs parallel designs A. A. Keshtkar MD, MPH, Ph. D
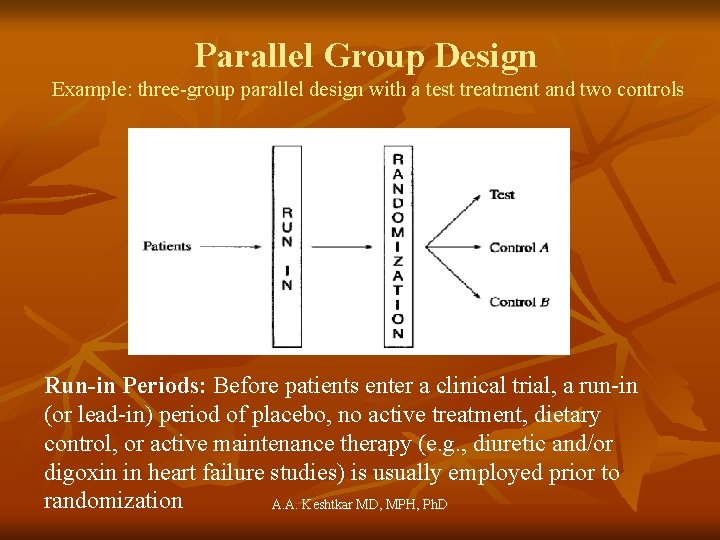
Parallel Group Design Example: three-group parallel design with a test treatment and two controls Run-in Periods: Before patients enter a clinical trial, a run-in (or lead-in) period of placebo, no active treatment, dietary control, or active maintenance therapy (e. g. , diuretic and/or digoxin in heart failure studies) is usually employed prior to randomization A. A. Keshtkar MD, MPH, Ph. D
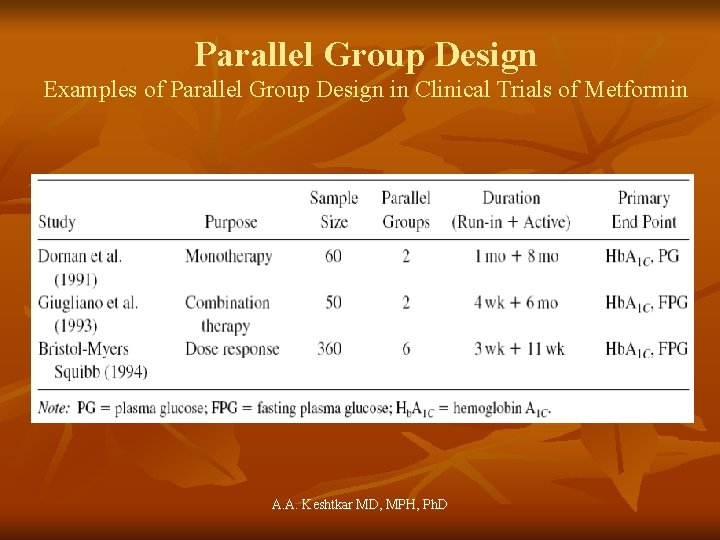
Parallel Group Design Examples of Parallel Group Design in Clinical Trials of Metformin A. A. Keshtkar MD, MPH, Ph. D

Cluster Randomized Design Definition: in cluster randomized designs, randomization is performed at the cluster level rather than at the subject level. Thus, the unit of analysis may and may not be the same as the unit of randomization. A. A. Keshtkar MD, MPH, Ph. D

Cluster Randomized Design Two commonly encountered mistakes in most cluster randomized trials: 1. Although the trials adopt a cluster randomization, the analysis of data completely ignores this fact and uses subject as the unit of analysis. 2. Sample size estimation fails to take into consideration the variance inflation factor A. A. Keshtkar MD, MPH, Ph. D
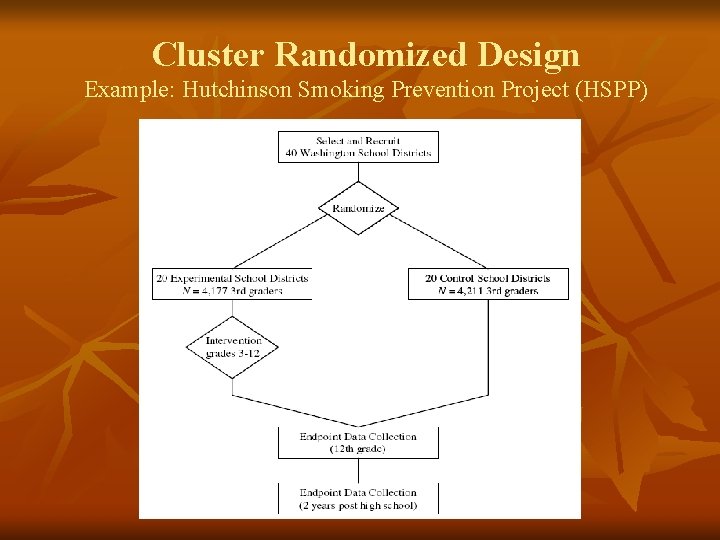
Cluster Randomized Design Example: Hutchinson Smoking Prevention Project (HSPP) A. A. Keshtkar MD, MPH, Ph. D

Crossover Design • Definition: a modified randomized block design in which each block receives more than one treatment at different dosing periods. A block can be a patient or a group of patients. Patients in each block receive different sequences of treatments • A crossover design is called a complete crossover design if each sequence contains all treatments under investigation. A. A. Keshtkar MD, MPH, Ph. D
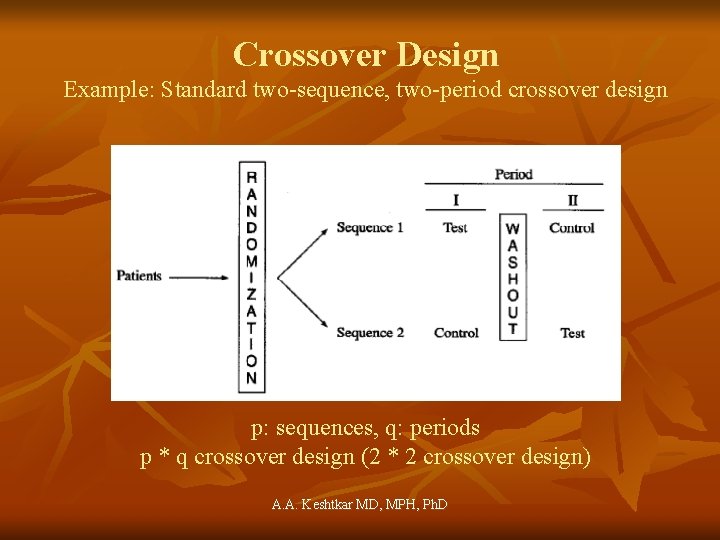
Crossover Design Example: Standard two-sequence, two-period crossover design p: sequences, q: periods p * q crossover design (2 * 2 crossover design) A. A. Keshtkar MD, MPH, Ph. D

Crossover Design Advantages: (1) It allows a within-patient comparison between treatments, since each patient serves as his or her own control (2) It removes the interpatient variability from the comparison between treatments (3) With a proper randomization of patients to the treatment sequences, it provides the best unbiased estimates for the differences between treatments. A. A. Keshtkar MD, MPH, Ph. D

Crossover Design is implemented under situations: (1) objective measures and interpretable data for both efficacy and safety are obtained (2) chronic (relatively stable) disease are under study (3) prophylactic drugs with relatively short half-life are being investigated (4) relatively short treatment periods are considered (5) baseline and washout periods are feasible A. A. Keshtkar MD, MPH, Ph. D

Crossover Design Examples of Crossover Design in Clinical Trials of Metformin A. A. Keshtkar MD, MPH, Ph. D

Randomization and Blinding A. A. Keshtkar MD, MPH, Ph. D

Randomization Concept: In nonrandomized controlled trial bias often occurs due to preconceived ideas or perceptions acquired during the study by: (1)the investigator and supporting staff who might influence reporting response to therapy or adverse events (2)the patient who might influence compliance, cooperation, or provision of information. A. A. Keshtkar MD, MPH, Ph. D

Randomization Methods The old or unacceptable methods (early 1970 s): the investigators or patients may be aware of which treatment the patients receive, subjective bias can consciously or unconsciously occur in both the assignment of patients to treatments and the evaluation of clinical outcomes for the treatment under investigation A. A. Keshtkar MD, MPH, Ph. D

Randomization Methods: 1. Complete Randomization: • Using Simple Randomization • Properties: • The chance that a patient receives either the test drug or the placebo is 50%. • Randomization of assignments is performed independently for each of the N patients A. A. Keshtkar MD, MPH, Ph. D

Randomization Methods: 1. Complete Randomization: • Generated by: • Random numbers table • Statistical softwares • Disadvantage: • Treatment impalance • Decreasing statistical power A. A. Keshtkar MD, MPH, Ph. D


Randomization Methods: 2. Permuted-Block Randomization: • As block size increases, the potential selection bias decreases • Although the use of random block size can reduce the selection bias, it cannot completely eliminate the bias. • The only way to eliminate the selection bias is to enforce a double-blinded procedure during the entire course of study for which both investigators and patients are blinded to block size and treatment assignments. A. A. Keshtkar MD, MPH, Ph. D

Blinding (Masking): § Randomization + Blinding = No bias in RCT § Types of blindness: § Open-label § Single blind § Double blind § Triple blind A. A. Keshtkar MD, MPH, Ph. D

Follow-up and Outcome Assessment A. A. Keshtkar MD, MPH, Ph. D

Follow-up Duration Determinants: § Treatment duration § The trial outcomes (early vs late outcomes) § Biologic concepts A. A. Keshtkar MD, MPH, Ph. D

Interim (repeated) Follow-up objectives: § Decreasing loss to follow-up § Measuring (assessing) multiple outcomes (endpoints) § Measuring (assessing) early and late outcomes (endpoints) §Patients’ safety (adverse events) § Longitudinal data analysis A. A. Keshtkar MD, MPH, Ph. D

Outcomes Assessment Classification: § Primary vs Secondary endpoints § Efficacy/effectiveness vs Safety § Clinical types: § Treatment response (efficacy) § Survival § Mortality § Adverse events § Quality of Life § Compliance, adherence § Satisfaction § Economic A. A. Keshtkar MD, MPH, Ph. D §…. .

Outcomes Assessment Classification: (cont) § Statistical types: § Continuous § Categorical (dichotomous) § Categorical (ordinal) § Time to event A. A. Keshtkar MD, MPH, Ph. D

Protocol Deviation Classification: § Ineligible patients; should be excluded § Non-compliance; the sources should be charactrized § lack of patient’s cooperation § treatment cessation or modification (adverse events) § Withdraw (discontinuation of medication); the evaluation should be continued § medical § non-medical A. A. Keshtkar MD, MPH, Ph. D

Ethics in Clinical Trials A. A. Keshtkar MD, MPH, Ph. D









General Consideration for Statistical Analysis A. A. Keshtkar MD, MPH, Ph. D

Choosing an appropriate method of analysis n There are several aspects of the data in the choosing an appropriate method of analysis: n n Number of treatment groups Independent or dependent outcome(s) data The type of outcome(s) data The distribution of outcome(s) data A. A. Keshtkar MD, MPH, Ph. D

Number of treatment groups n n Comparing two groups of treatment (2 arms parallel) Comparing several groups (>2) of treatment A. A. Keshtkar MD, MPH, Ph. D

Independent or dependent outcome(s) data n Independent outcome(s) data: n n Parallel design Dependent outcome(s) data: n n n Historical control (before after) Matched pair parallel Crossover (within each sequence) A. A. Keshtkar MD, MPH, Ph. D

The type of data § Statistical types: § Continious § Categorical (dichotomous) § Categorical (ordinal) § Time to event A. A. Keshtkar MD, MPH, Ph. D

Distribution of data n n n For independent groups, parametric methods require the observations within each group to have an approximately Normal distribution. If the raw data do not satisfy this condition, a transformation may be successful. Otherwise, a non-parametric method should be used. A. A. Keshtkar MD, MPH, Ph. D


Clinical Trial Registration www. irct. ir A. A. Keshtkar MD, MPH, Ph. D


- Slides: 73