Patient rights Implementation and contribution to the quality

Patient rights: Implementation and contribution to the quality of healthcare services. Organisers: Dr Androulla Eleftheriou Executive Director, Thalassaemia International Federation Hamburg, 13 October 2019

TABLE OF CONTENTS 1. Historical background 2. Human Rights: The Global Picture 3. Patients’ Rights in EU countries 4. Patients’ Rights & EU Policies 5. Prerequisites to Safeguard Patients’ Rights 6. Implementation of Patients’ Rights 7. Patients’ Rights & Healthcare Services 8. Patients’ Rights & Chronic Patients 9. Advocating for Patients’ Rights
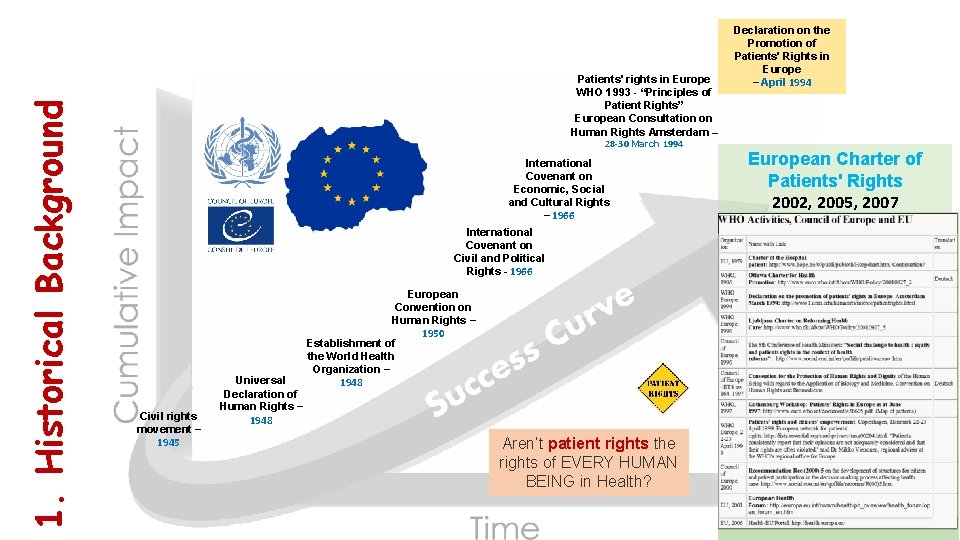
1. Historical Background Patients' rights in Europe WHO 1993 - “Principles of Patient Rights” European Consultation on Human Rights Amsterdam – 28 -30 March 1994 International Covenant on Economic, Social and Cultural Rights – 1966 International Covenant on Civil and Political Rights - 1966 Civil rights movement – 1945 Universal Declaration of Human Rights – 1948 European Convention on Human Rights – 1950 Establishment of the World Health Organization – 1948 Aren’t patient rights the rights of EVERY HUMAN BEING in Health? Declaration on the Promotion of Patients' Rights in Europe – April 1994 European Charter of Patients' Rights 2002, 2005, 2007

2. Human Rights: The Global Picture The rights of patients are protected ONLY in the context of: i. Hospital regulations; ii. Codes of conduct of doctors; iii. Disability laws. With only a few exceptions, most countries do not have any legal framework for safeguarding patients’ rights and their contribution in decision -making by expressing their positions and views.
3. Patients’ Rights in EU countries A patients’ rights law means an important shift towards a more patient-oriented approach. Key Documents EU Declaration on the Promotion of Patients’ Rights (1994): Derived from the WHO European Consultation on the Rights of Patients Convention on Human Rights and Biomedicine (1997): The only international legally binding instrument on the protection of human rights in the biomedical field § All EU countries have developed a legal/political approach at a national level to defining and implementing patients’ rights. § Only a few EU Member States are lacking a special law on patients’ rights (Austria, Bulgaria, Ireland, Italy and Malta). Source: Patients’ rights in the European Union European Charter of Patients’ Rights (2002): Drafted by the Active Citizenship Network Directive 2011/24/EU on the application of patients' rights in cross-border healthcare (2011) The main problem is the actual enforcement of patients’ rights. Some countries developed alternative enforcement mechanisms: • Monitoring bodies (Bulgaria) • Advocates (Hungary) • Ombudspersons (Poland) • Legal representation of individuals by patient associations (France)
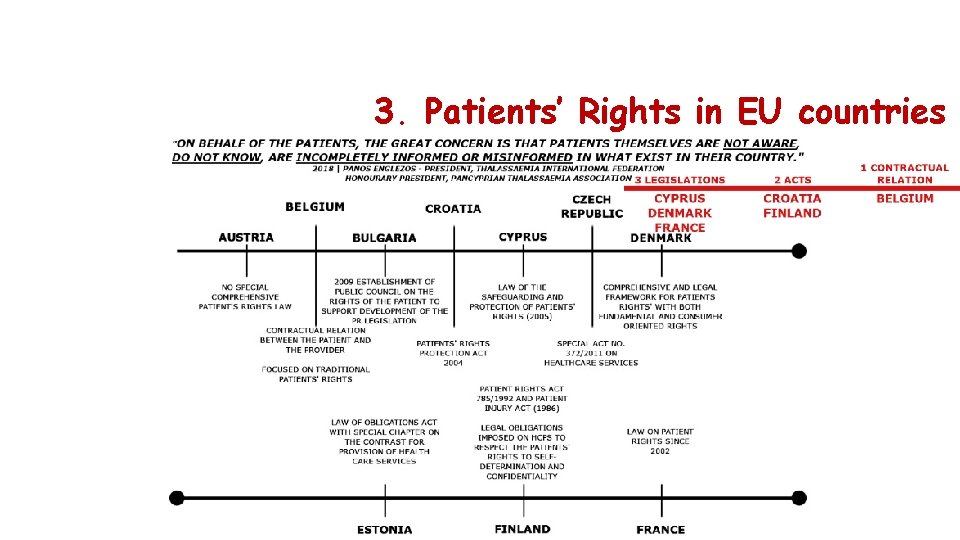
3. Patients’ Rights in EU countries
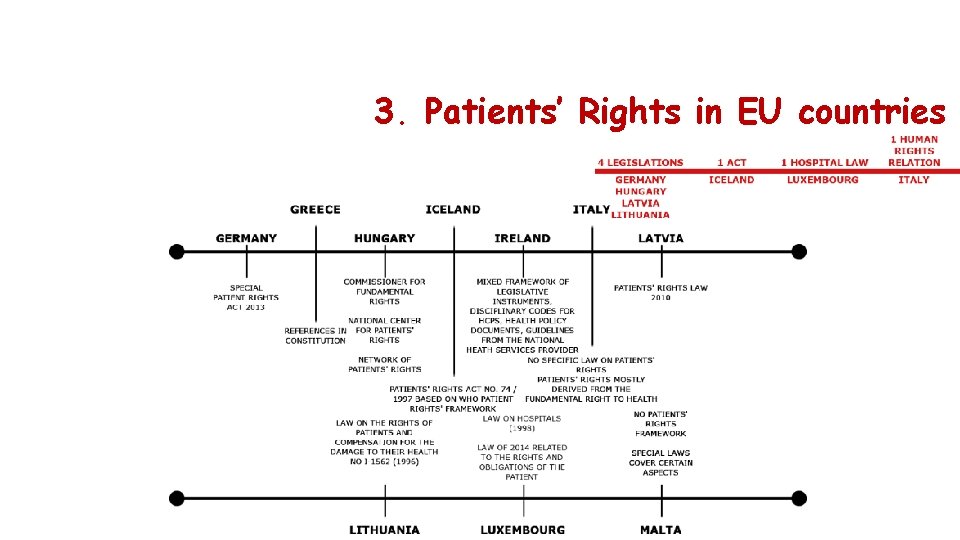
3. Patients’ Rights in EU countries
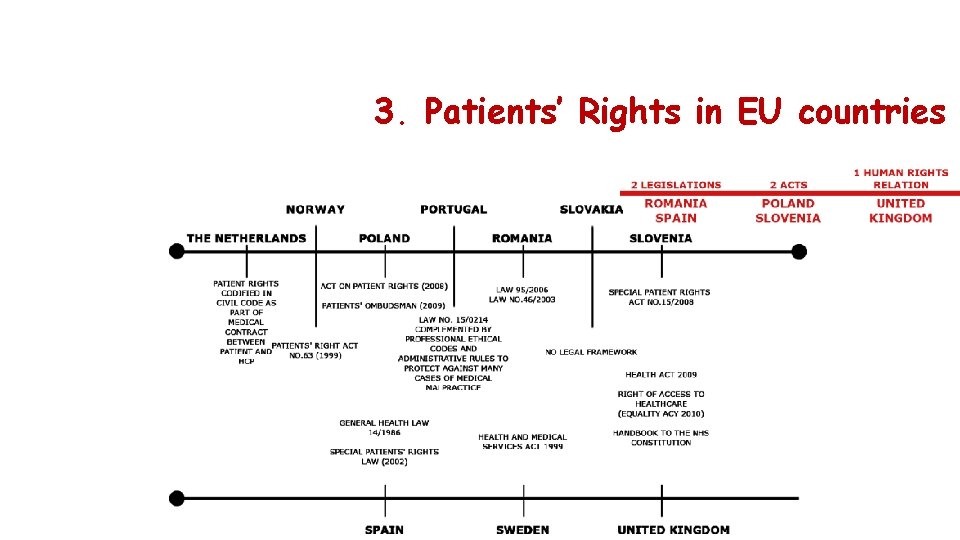
3. Patients’ Rights in EU countries

3. Patients’ Rights in EU countries PART ONE: FUNDAMENTAL RIGHTS PART TWO: FOURTEEN RIGHTS OF THE PATIENT PART THREE: RIGHTS OF ACTIVE CITIZENSHIP PART FOUR: GUIDELINES FOR IMPLEMENTING THE CHARTER

3. Patients’ Rights in EU countries

4. Patients’ Rights & EU Policies • A DYNAMIC PROCESS in a changing environment, mainly in EU but also globally. • Different patients’ rights derive from: ü At European level: EU Directives, Regulations, Recommendations ü At international level: WHO (Resolutions), Council of Europe (Recommendations), UN (Ratifications) These cover an array of topics: • • • Rare Diseases Patients’ Orphan Medicines Rights Clinical tests Pharmacovigilance Blood Quality and Safety Recognition of Professional Qualifications Falsified Medicines Application of patients' rights in cross-border healthcare European Reference Networks

4. Patients’ Rights & EU Policies Blood Safety & Donation Pharmacovigilance Directive 2002/98/EC of the European Parliament and of the Council of 27 January 2003 setting standards of quality and safety for the collection, testing, processing, storage and distribution of human blood and blood components and amending Directive 2001/83/EC Commission Directive 2004/33/EC of 22 March 2004 implementing Directive 2002/98/EC of the European Parliament and of the Council as regards certain technical requirements for blood and blood components Commission Directive 2005/61/EC of 30 September 2005 implementing Directive 2002/98/EC of the European Parliament and of the Council as regards traceability requirements and notification of serious adverse reactions and events Commission Directive 2005/62/EC of 30 September 2005 implementing Directive 2002/98/EC of the European Parliament and of the Council as regards Community standards and specifications relating to a quality system for blood establishments Commission Directive 2009/135/EC of 3 November 2009 allowing temporary derogations to certain eligibility criteria for whole blood and blood components donors laid down in Annex III to Directive 2004/33/EC in the context of a risk of shortage caused by the Influenza A(H 1 N 1) pandemic Commission Directive 2014/110/EU of 17 December 2014 amending Directive 2004/33/EC as regards temporary deferral criteria for donors of allogeneic blood donations Commission Directive (EU) 2016/1214 of 25 July 2016 amending Directive 2005/62/EC as regards quality system standards and specifications for blood establishments Directive 2001/83/EC of the European Parliament and the Council of 6 November 2001 on the Community code relating to medicinal products for human use Regulation (EC) No 726/2004 of the European Parliament and of the Council of 31 March 2004 laying down Community procedures for the authorisation and supervision of medicinal products for human and veterinary use and establishing a European Medicines Agency Directive 2010/84/EU of the European Parliament and of the Council of 15 December 2010 amending, as regards pharmacovigilance, Directive 2001/83/EC on the Community code relating to medicinal products for human use Orphan Medicinal Products Regulation (EC) No 141/2000 of the European Parliament and of the Council of 16 December 1999 on orphan medicinal products Clinical Trials Directive 2001/20/EC of the European Parliament and of the Council of 4 April 2001 on the approximation of the laws, regulations and administrative provisions of the Member States relating to the implementation of good clinical practice in the conduct of clinical trials on medicinal products for human use Cross-border Healthcare Directive 2011/24/EU of the European Parliament and of the Council of 9 March 2011 on the application of patients’ rights in cross-border healthcare Rare Diseases Council Recommendation of 8 June 2009 on an action in the field of rare diseases Migrants’ Health European Agenda on Migration Falsified Medicines Commission delegated Regulation (EU) 2016/161 of 2 October 2015 supplementing Directive 2001/83/EC of the European Parliament and of the Council by laying down detailed rules for the safety features appearing on the packaging of medicinal products for human use

4. Patients’ Rights & EU Policies Patients living in EU countries have the right: 1. TO RECEIVE HEALTHCARE WHEN VISITING ANOTHER EU COUNTRY 2. TO BE REIMBURSED FOR HEALTHCARE SOUGHT IN ANOTHER EU COUNTRY 3. TO RECEIVE INFORMATION ON SAFETY AND QUALITY STANDARDS IN EU COUNTRIES 4. TO BE TREATED BY QUALIFIED HEALTHCARE PROFESSIONALS 5. TO GET A COPY OF THEIR MEDICAL RECORDS 6. TO HAVE THEIR PRESCRIPTION RECOGNISED IN ALL EU COUNTRIES 7. TO BE TREATED WITH SAFE AND EFFECTIVE MEDICINES 8. TO BE ABLE TO REPORT SUSPECTED SIDE EFFECTS OF MEDICINES 9. TO BE TREATED WITH SAFE MEDICAL DEVICES 10. TO BENEFIT FROM COMMON HIGH STANDARDS OF QUALITY FOR BLOOD, ORGANS, TISSUES AND CELLS
5. Prerequisites to Safeguard Patients’ Rights ü BE ACTIVELY INVOLVED in their development, monitoring, implementation and updating (when necessary) ü BE KNOWLEDGEABLE of exactly WHAT your RIGHTS are within the applicable national legislative and other framework. Each country (EU Member State) needs to develop their own national laws based on the European Charter of Patients’ Rights.
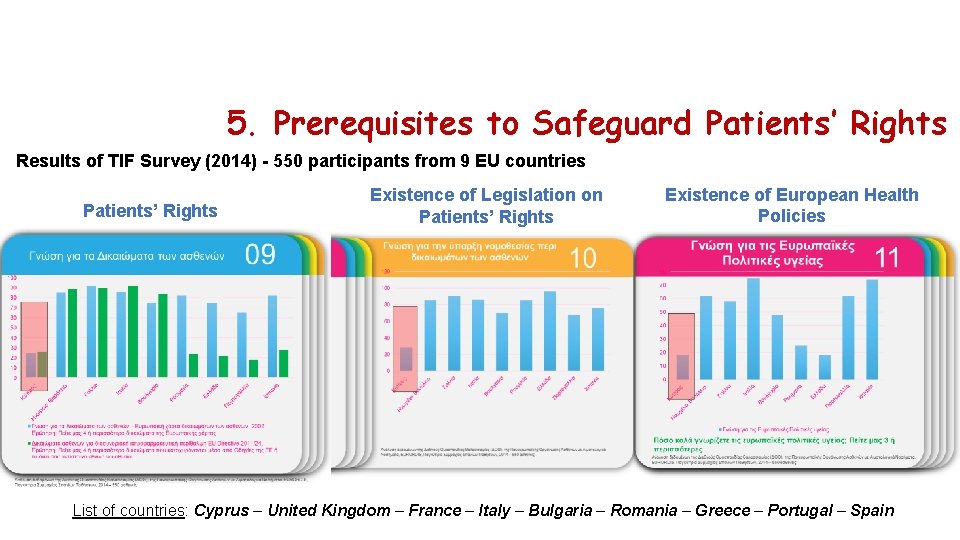
5. Prerequisites to Safeguard Patients’ Rights Results of TIF Survey (2014) - 550 participants from 9 EU countries Patients’ Rights Existence of Legislation on Patients’ Rights Existence of European Health Policies List of countries: Cyprus – United Kingdom – France – Italy – Bulgaria – Romania – Greece – Portugal – Spain

6. Implementation of Patients’ Rights (as per the European Charter of Patients’ Rights) Patients’ rights (Charter or Law) in place The BEST TOOL for the citizen / patient to ensure that dignity, quality and respect are elements characterising the healthcare services they receive. Implementation and adherence to Patients’ Rights (Charter/Law) contributes to the better understanding of the needs for corrective measures – often related to: i. Reductions in waste and inefficiency of healthcare systems; ii. Public health reforms.

7. Patients’ Rights & Healthcare Services “Arguably, the most important public policies we have in the United States have come from nonprofit organizations lobbying for their causes. . . ” David F. Arons, in Nonprofit Governance and Management … and also from the persistence and commitment of non-profit patient organisations to implement and safeguard the rights of patients. Prof. George Stamatoyiannopoulos Medical Geneticist University of Washington “It is in the context of safeguarding the rights of patients with thalassaemia and other severe haemoglobin disorders that the pillars of work and activities of TIF have been based and it is this element that has contributed immensely to the success achieved albeit at different levels across the world, in the prevention and care of this disorder. ”

8. Patients’ Rights and Chronic Patients’ Rights (in a national Charter or Law) need to be translated to reflect the Rights of patients with chronic diseases, e. g. β-thalassaemia (TDT): In Thalassaemia, for example: • Right to preventative measures, quality translates to: ü Right to safe and adequate blood ü Right to appropriate monitoring ü Right to effective chelation treatment ü Right to multidisciplinary care • Right to innovation ü Right to be fully informed and participate in clinical studies ü Right to access, personalised care, choice of care and its provider, choice of drug, sharing decisions

8. Patients’ Rights and Chronic Patients In case of suboptimal care, delayed or inappropriate monitoring TRANSLATE YOUR NEEDS into a violation of your Rights DEMAND PROGRESS & CHANGES, if your needs are NOT MET adequately, in terms of quality and time. DIGNITY and RESPECT to the patient AT ALL TIMES (BEFORE, DURING and AFTER)

9. Advocating for patients’ rights Legislation on Patients’ Rights is YOUR WEAPON for ACHIEVING CHANGES and improvements in the quality standards of health services. It is the OBLIGATION of every Government to RESPECT the rights of its citizens as patients and consequently to take corrective measures and QUALITY improvements → Quality of Services & Quality of Life You can play a key role in the fight against waste in healthcare by acting as sentinels to track and report corruption, irregularities and inefficiency.

“Not a single bedpan should fall on a hospital floor without the clatter being heard in Whitehall” Aneurin Bevan Former Secretary of State for Health United Kingdom Violating the Human Right to Health and the Rights of Patients SHOULD NOT BE ACCEPTABLE TO ANYONE = The patients’ community (at national and international levels) need to ACT and REACT.

- Slides: 22