Pathway Jetstream G 3 A Rotational Flushing and











- Slides: 22

Pathway Jetstream G 3: A Rotational Flushing and Aspirating Atherectomy System William A. Gray MD Director of Endovascular Services Associate Professor of Clinical Medicine Columbia University Medical Center The Cardiovascular Research Foundation

DISCLOSURES William A. Gray, MD Consulting Fees – Access. Closure, Inc. , Co. Aptus, Inc. , Mercator Med. Systems, Inc. , Ovalis, Inc. , Paragon Medical, Inc. , Pathway Medical Technologies, Inc. , Sage Medical, Inc. Grants/Contracted Research – Atritech, Inc. , Cordis, a Johnson & Johnson company, CREST/ National Institutes of Health, W. L. Gore & Associates, Inc. Ownership Interest (Stocks, Stock Options or Other Ownership Interest) – Access. Closure, Inc. , Co. Aptus, Inc. , Ovalis, Inc. , Paragon Medical, Inc. , Pathway Medical Technologies, Inc. , Sage Medical, Inc.

Pathway Jetstream G 3™ System Jetstream® Catheter Control Pod Console Blades “Down” 2. 1 mm Blades “Up” 3. 0 mm § OTW 135 cm Front Cutting, High Speed Rotational Catheter §. 014 GW /7 F sheath compatible § Differential cutting tip removes all plaque types § Aspiration ports collect plaque & thrombus § Two sizing options (single device) § Integrated system control within the sterile field § Rotational Speed § One-step size expansion § 70 K rpm (target therapy speed) § Cassette set up and data display outside sterile field

Pathway PVD study 1 st generation device • Purpose: A prospective, single arm, multi- center study to assess performance of the Pathway PV™ Atherectomy System during percutaneous peripheral vascular interventions Drs. Scheinert and Zeller - co-PIs ¡ 172 patients at 9 European centers ¡ 100% data monitoring ¡ Angiographic core lab and CEC adjudicated ¡

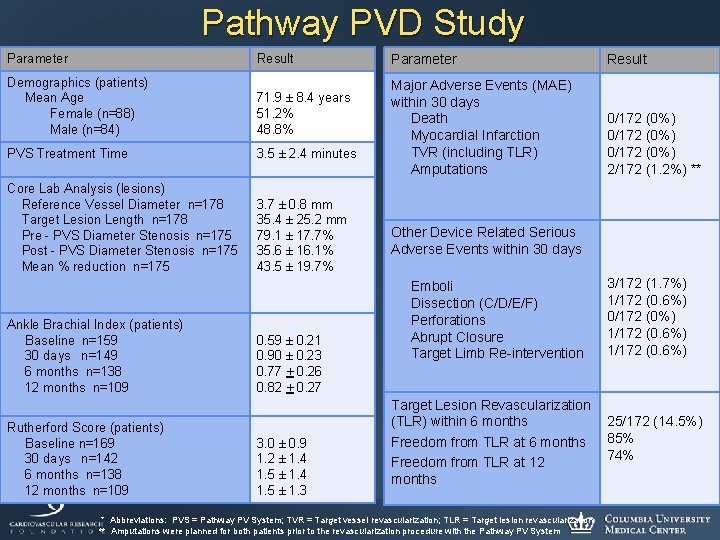
Pathway PVD Study Parameter Result Demographics (patients) Mean Age Female (n=88) Male (n=84) 71. 9 ± 8. 4 years 51. 2% 48. 8% PVS Treatment Time 3. 5 ± 2. 4 minutes Core Lab Analysis (lesions) Reference Vessel Diameter n=178 Target Lesion Length n=178 Pre - PVS Diameter Stenosis n=175 Post - PVS Diameter Stenosis n=175 Mean % reduction n=175 3. 7 ± 0. 8 mm 35. 4 ± 25. 2 mm 79. 1 ± 17. 7% 35. 6 ± 16. 1% 43. 5 ± 19. 7% Ankle Brachial Index (patients) Baseline n=159 30 days n=149 6 months n=138 12 months n=109 Rutherford Score (patients) Baseline n=169 30 days n=142 6 months n=138 12 months n=109 0. 59 ± 0. 21 0. 90 ± 0. 23 0. 77 + 0. 26 0. 82 + 0. 27 3. 0 ± 0. 9 1. 2 ± 1. 4 1. 5 ± 1. 3 Parameter Result Major Adverse Events (MAE) within 30 days Death Myocardial Infarction TVR (including TLR) Amputations 0/172 (0%) 2/172 (1. 2%) ** Other Device Related Serious Adverse Events within 30 days Emboli Dissection (C/D/E/F) Perforations Abrupt Closure Target Limb Re-intervention Target Lesion Revascularization (TLR) within 6 months Freedom from TLR at 12 months * Abbreviations: PVS = Pathway PV System; TVR = Target vessel revascularization; TLR = Target lesion revascularization ** Amputations were planned for both patients prior to the revascularization procedure with the Pathway PV System 3/172 (1. 7%) 1/172 (0. 6%) 0/172 (0%) 1/172 (0. 6%) 25/172 (14. 5%) 85% 74%

Evolutionary changes in Pathway device Proximal Aspiration Ports Jetstream G 2™ Jetstream™

Pathway Jetstream G 3™ System G 2 NXT Distal Cutter: G 3 Distal Cutter: 10 Flute Design 54% more efficient at differential cutting compared to Jetstream G 2™ NXT* *Calculated in Blades Down (Bench test data on file at Pathway Medical)

Pathway Jetstream G 3™ System • 7 French Compatibility • Optimized Aspiration* • Enhanced Cutting Effectiveness* JETSTREAM G 3 Improvements *As compared to Jetstream G 2™ NXT in bench testing in challenging lesion model **As compared to Jetstream G 2™ NXT in bench testing Increased Aspiration Efficiency** 11% Improved differential cutting efficiency (Blades Down)* 54%

JETSTREAM G 3™ Animation

Pathway Jetstream G 3™ System • Definitive Therapy for Calcium & CTOs ¡ Excellent differential cutting efficiency • Optimized Cutting tip • Improved torque ¡ Front-end cutting • Active Aspiration ¡ Only atherectomy device with this feature • Minimizes risk of distal embolization • Short Procedure Times ¡ ¡ 54% Increase in Cutting Efficiency* Minimal Fluoroscopy /Reduced Lab Time Simplicity of Operation Atherectomy and Thrombectomy indications • “Single Device” Solution ¡ Multiple Vessel Sizes • Expandable Blade Technology • Treats disease from SFA to below the knee ¡ All Pathologies • Atheromatous, Calcium and Thrombus *Compared to Jetstream G 2™NXT in Blades Down during bench testing

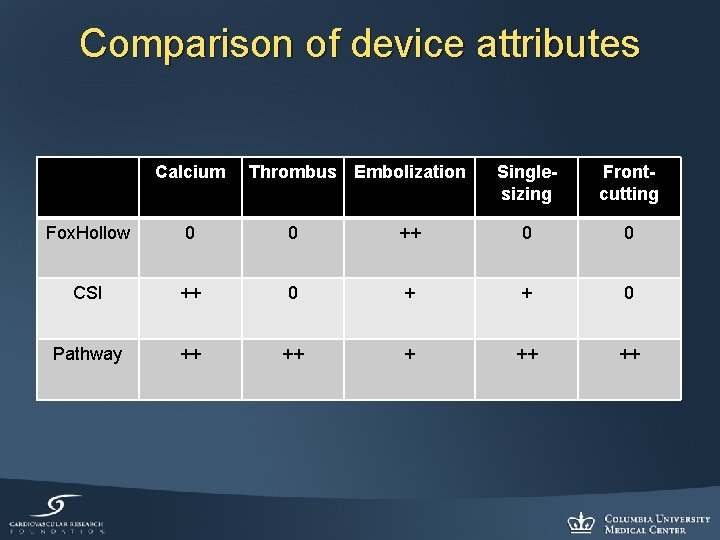
Comparison of device attributes Calcium Thrombus Embolization Singlesizing Frontcutting Fox. Hollow 0 0 ++ 0 0 CSI ++ 0 + + 0 Pathway ++ ++ ++

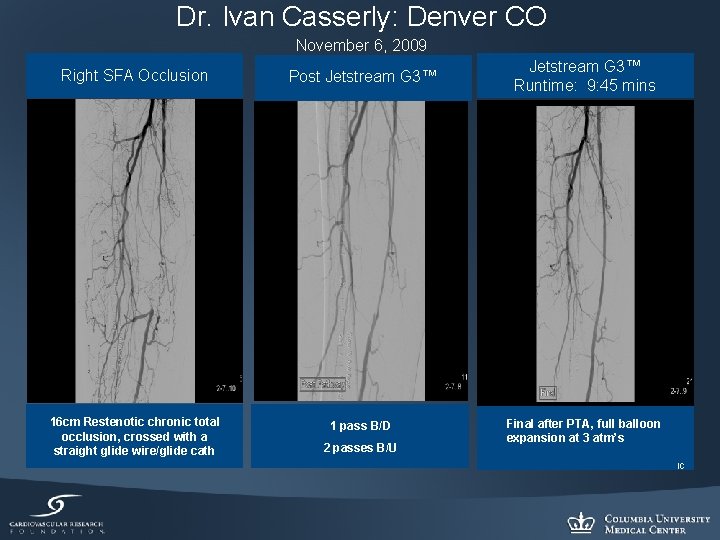
Dr. Ivan Casserly: Denver CO November 6, 2009 Right SFA Occlusion Post Jetstream G 3™ Runtime: 9: 45 mins 16 cm Restenotic chronic total occlusion, crossed with a straight glide wire/glide cath 1 pass B/D Final after PTA, full balloon expansion at 3 atm’s 2 passes B/U IC

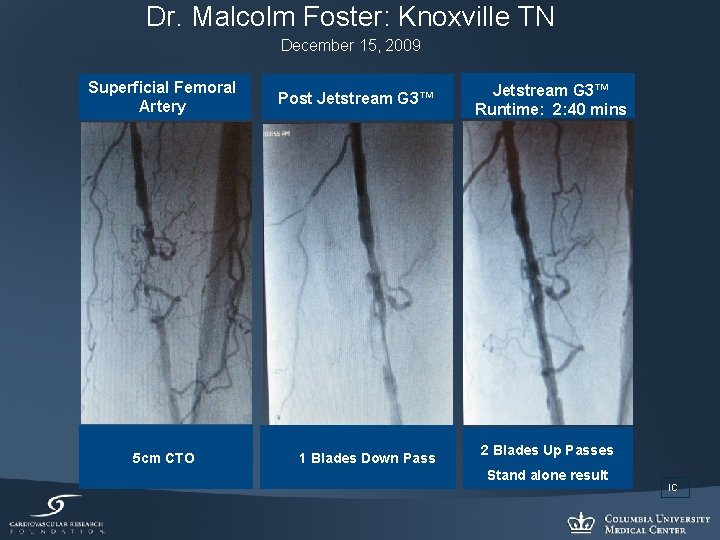
Dr. Malcolm Foster: Knoxville TN December 15, 2009 Superficial Femoral Artery 5 cm CTO Post Jetstream G 3™ 1 Blades Down Pass Jetstream G 3™ Runtime: 2: 40 mins 2 Blades Up Passes Stand alone result IC

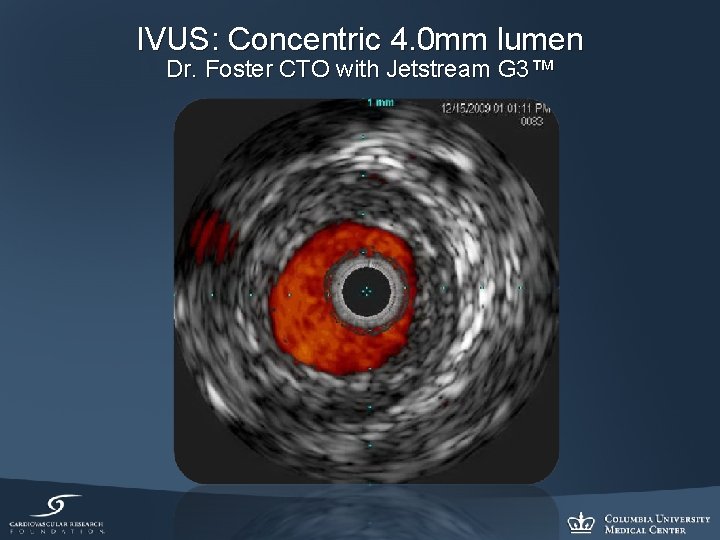
IVUS: Concentric 4. 0 mm lumen Dr. Foster CTO with Jetstream G 3™

Calcification

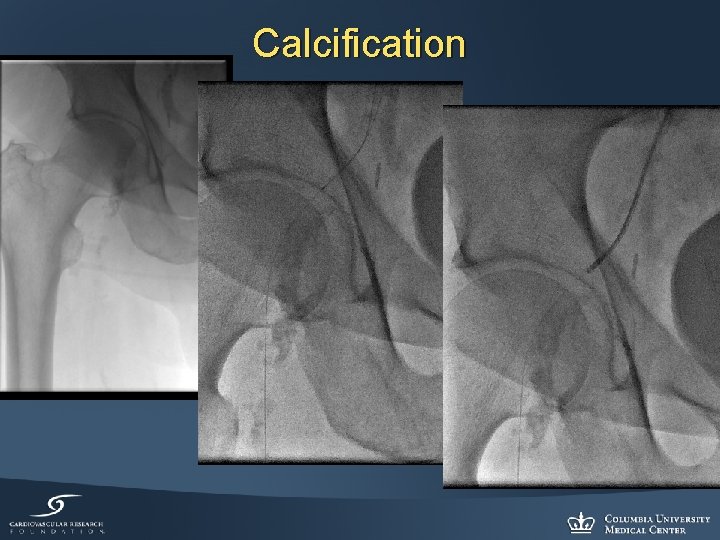
Calcification

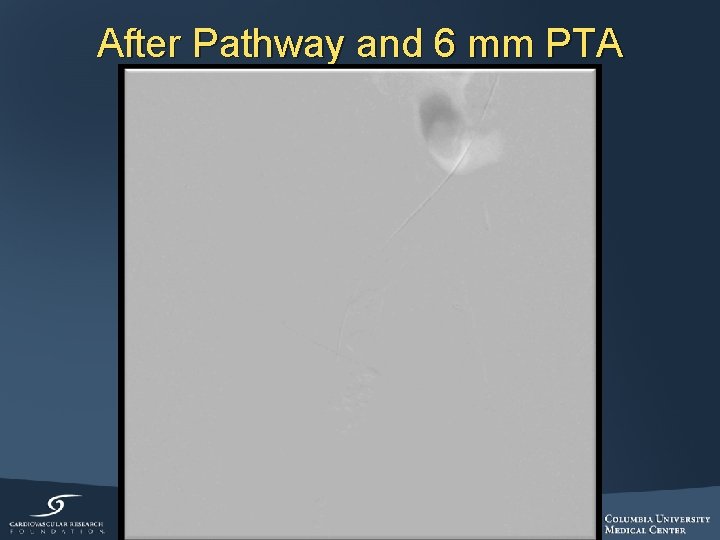
After Pathway and 6 mm PTA

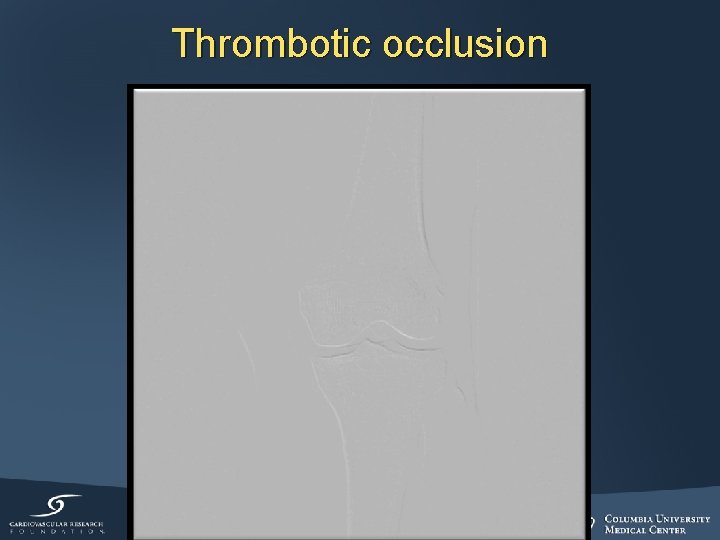
Thrombotic occlusion

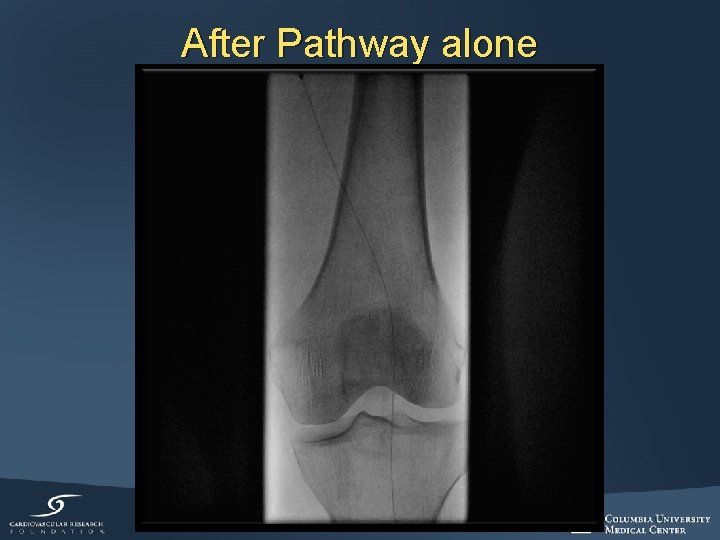
After Pathway alone

After Pathway and PTA

Conclusions • Pathway device adds to the tool set for PAD intervention with the capability to address: Calcification ¡ Thrombus-containing lesions ¡ ISR ¡ • Good stand alone results (PTA only) as demonstrated by low rates of stent usage • Evolutionary improvements have extended applications, safety and efficacy original device • New devices will address larger and smaller vascular needs

Procedural Outcomes § 99% device success § Mean device activation time = 3: 37 minutes § 33% of procedures were stand-alone § 57% used adjunctive balloon § Only 7% require adjunctive stent