Pathophysiology of Ageing Mikls Molnr 6357 Semmelweis University







































- Slides: 56

Pathophysiology of Ageing Miklós Molnár 6357 Semmelweis University Inst. of Pathophysiology


Cumulatív % of Increase Aging as a global phenom Years

Introduction to Human Aging u Number and percentage of the elderly · In the US, more persons over 65 than under 25 years of age · Median age was ~32 in 1995; was 36 in 2000, and will be ~42 by 2040

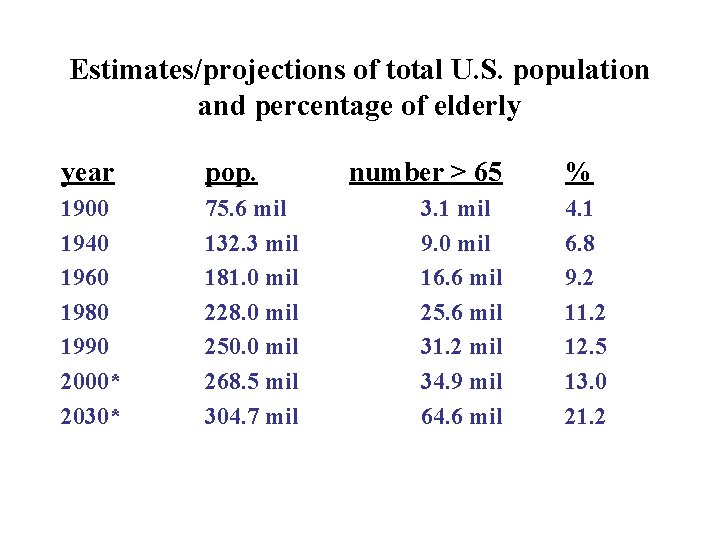
Estimates/projections of total U. S. population and percentage of elderly year pop. 1900 1940 1960 1980 1990 2000* 2030* 75. 6 mil 132. 3 mil 181. 0 mil 228. 0 mil 250. 0 mil 268. 5 mil 304. 7 mil number > 65 3. 1 mil 9. 0 mil 16. 6 mil 25. 6 mil 31. 2 mil 34. 9 mil 64. 6 mil % 4. 1 6. 8 9. 2 11. 2 12. 5 13. 0 21. 2

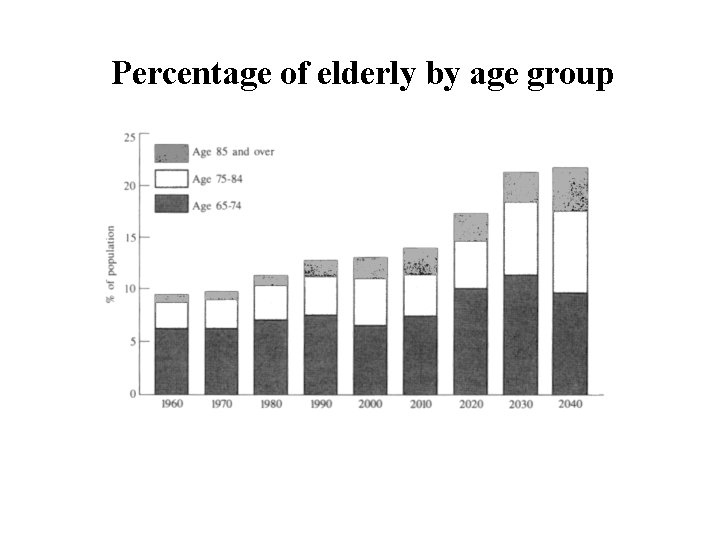
Percentage of elderly by age group

Problems for the younger members of our society · 30% of health care resources used by persons >65 years (will increase to 50% by 2030) · Older persons require more social services and specialized recreational facilities; these require money (increased taxes) · The burden of support falls now and will fall more heavily on the younger generations.

Ages · Chronologic age · Biologic age – – – – mitochondrial DNA damage β-galactosidase (fro skin biopsy) Glycolysation products presbyopia Rate of DNA unrounding vitalcapacity etc.

WHO Classification of age groups · Middle-aged (45 -59 years) · Elderly (60 -74 years) · Old (75 -90 years) · Very old (90 - )

Introduction to Human Aging u General effects of aging · · Aging is a continuing, normal process. Begins at maturity, ends with death Effects of aging increase at age 40. Aging is influenced by interactions of genetics and environmental factors.

Biologic Changes Occuring by Age · Universal, in every species · Progressively on going process · Internal causes · Viability of the body is decreasing

So what is normal biological ageing? Ageing is characterised primarily by a reduction in the capability to adapt to changes in the environment. This adaptation includes an ability to recognise the abnormal substances which we come into contact with in daily life. Hence if this capacity is reduced then the organism becomes ever greatly prone to disease and damage.

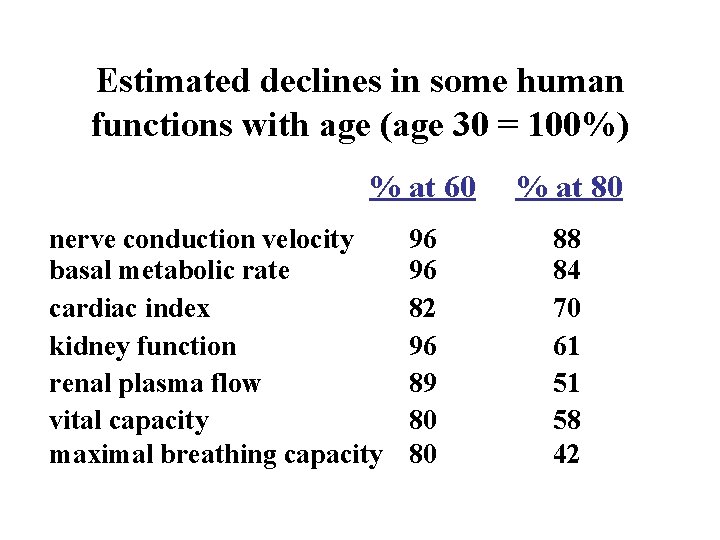
Estimated declines in some human functions with age (age 30 = 100%) % at 60 nerve conduction velocity basal metabolic rate cardiac index kidney function renal plasma flow vital capacity maximal breathing capacity 96 96 82 96 89 80 80 % at 80 88 84 70 61 51 58 42

The Deficits of Aging Basic physiologic changes: · less water, more fat · decreased circulation · Immune system failure · neuronal loss, myofibril loss, cartilage loss · Limited fibroblast replication lifetime limit = 50 replications

Deficits of Aging: Sensory Hearing – Tinnitus (ringing): blocking frequencies – Presbycusis: limited ability to pick out speech in a noisy environment Visual – Macular degeneration, cataracts

Deficits of Aging: Sensory · Taste > Loss of most taste buds except sugar and salt · Smell > Loss of olfactory stimulation > Stimulation helps preserve what’s lost

Deficits of Aging: Anatomic · Bones > 1 -2% mineral loss of bone matrix after age 65 > Exercise > Medication · Muscles > 1 -2% loss of myofibrils per year after 65 > If you can increase strength by 25% through exercise, you can add a decade of function, compensated by increasing myofibril size

Deficits of Aging: Functional · Reflexes > Slowed with age · Balance > Loss of proprioceptive neurons from feet and neck receptors Result = FALLS (40% of >70 y will fall) > Compensate with exercise and balance drills

Deficits of Aging: Functional · Cognition > Decrease in short term memory > Can recall, but need more time for recall > Affected by diabetes, HTN · Learning > Visuospatial is diminished but auditory is well preserved > Learn slowly, but better able to put it to wider use

Deficits of Aging: Psychiatric · Depression · Dementia > Often with depression, frequently medication related > Incidence rises with survival - Age 65 -74, 2 -3% - Age 75 -84, 22% - Age >85, 50%

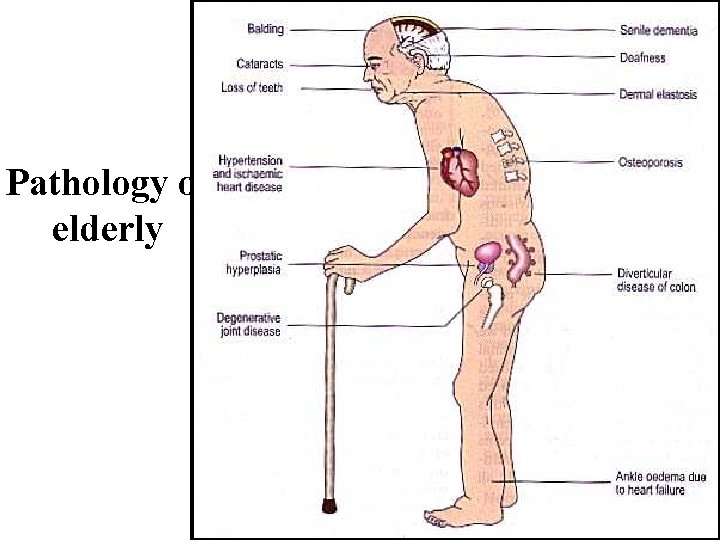
Pathology of elderly

Normal Brain surface:

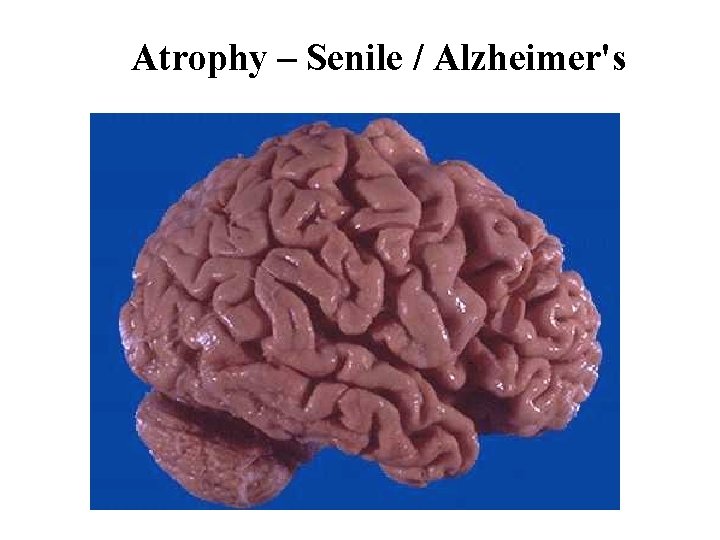
Atrophy – Senile / Alzheimer's

Ageing: “Progressive time related loss of structural and functional capacity of cells leading to death” · Senescence, Senility, Senile changes. · Ageing of a person is intimately related to cellular ageing. · Blood vessel damage precedes ageing.

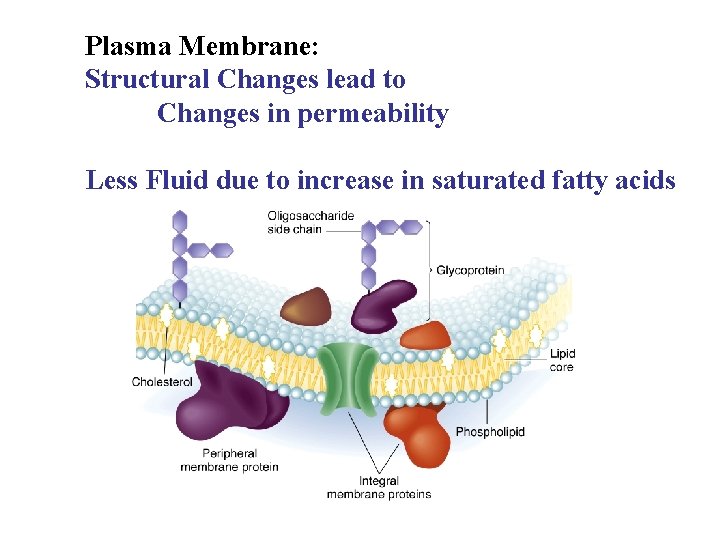
Plasma Membrane: Structural Changes lead to Changes in permeability Less Fluid due to increase in saturated fatty acids

Nuclear Changes Chromatin becomes more condensed (increase cross-links) (disulfide bonds between histones) Implication: Damage to DNA less likely repaired Lymphocytes in culture, add reducing agents to medium (break disulfide bonds) senescent cells divide again

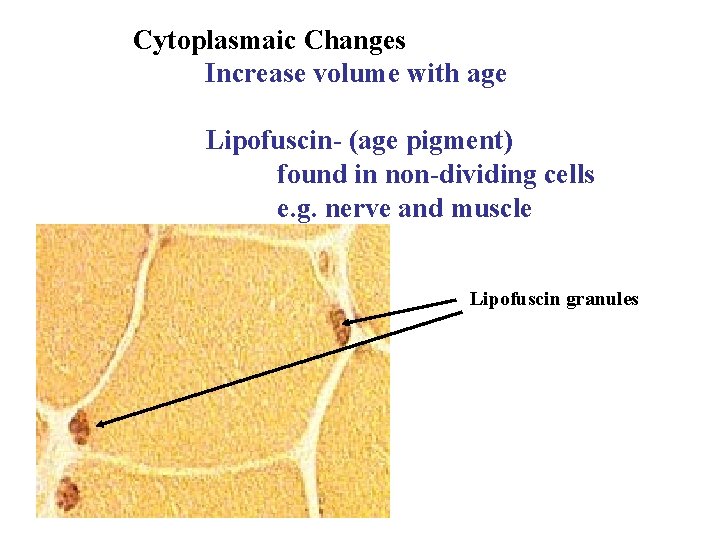
Cytoplasmaic Changes Increase volume with age Lipofuscin- (age pigment) found in non-dividing cells e. g. nerve and muscle Lipofuscin granules

Ribosomal Changes r. RNA decreases with age general decline in protein synthesis

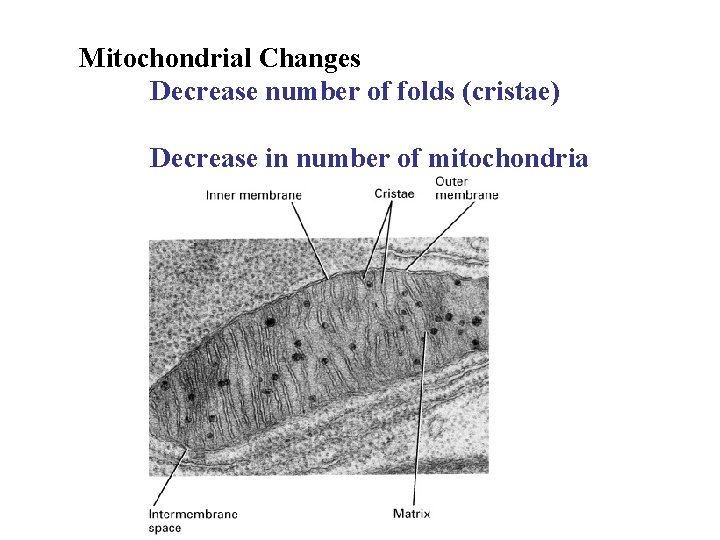
Mitochondrial Changes Decrease number of folds (cristae) Decrease in number of mitochondria

Lysosomal Changes Decrease in activity leads to accumulation of cellular garbage e. g. lipofuscins Release of enzymes leads to cell death

Pre-programmed Cell Death (apoptosis) Apoptosis vs. Necrosis - external cause (trauma) random breaks in DNA Apoptosis- internal cause (cellular suicide) non-random 180 base fragments Apoptosis - natural developmental process e. g. interdigital tissue (webbing) neurons

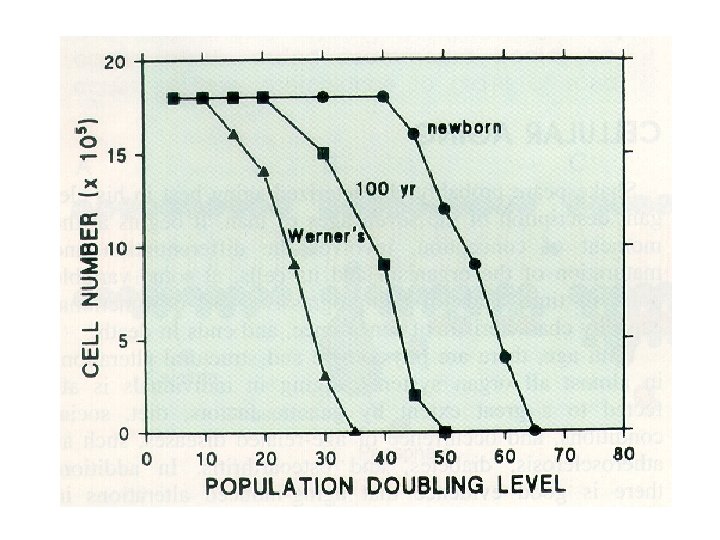
Factors affecting Ageing: · Genetic 60% & Environmental 40% · Clock genes, (fibroblast culture) · Werner’s syndrome. · Age gene on Chromosome 1. · “Age” is a character from female parent. · Mammalian mitochondria come from ovum.

Progeria

Factors affecting Ageing: · Environmental factors (40%) · Trauma · Diseases – Atherosclerosis, diabetes · Diet – malnutrition, obesity etc. · Psychological & Social health – stress.

Theories of Aging Possible Mechanisms (How ? )

3 Criteria of Aging Theories 1. Must occur in all individuals of the population 2. Produce Changes in function/ structure 3. Changes increase with age (progressive)

3 Catagories of Theories 1. Wear and Tear (Damage) Theory 2. Physical/ Chemical Changes 3. Genetically Programmed

Why Do We Age? Why do not we die aerlier?

Wear and Tear Theories Weismann (1891) Ordinary insults and injuries of daily living accumulate and decrease function to some sub-vital level e. g. loss of teeth starvation molecular level: enzymes accumulation of harmful metabolites (cell garbage theory) e. g. aldehydes, free radicals, lipofuscins interfere with cell function

Wear and Tear Theories (cont. ) finite energy theories · animals with high metabolic rates have shorter life spans · rats on calorie restrictive diets live longer

Wear and Tear Theories Refuted · Animals in protected environments have no change in maximum life span. · Time-dependent changes cannot initiate aging · Cellular/ Genetic evidence Reformulated as Failure to Repair Theories

Physical/ Chemical Changes · Cross Linkage Theory (Post-translational modification) – macromolecules cross linked (denatured) leading to a decline in function e. g. proteins- collagen, elastin How ? Disulfide bonds Advanced Glycation End-Products (AGEs) accelerated in diabetics DNA cross-linkage occurs also

Physical/ Chemical Changes · Altered Protein Theory – protein folding no change in primary structure decline in catalytic activity with age e. g. enolase in nematodes denature/renature experiments · increased carbonyl content (ketones, aldehydes) of proteins (oxidative)

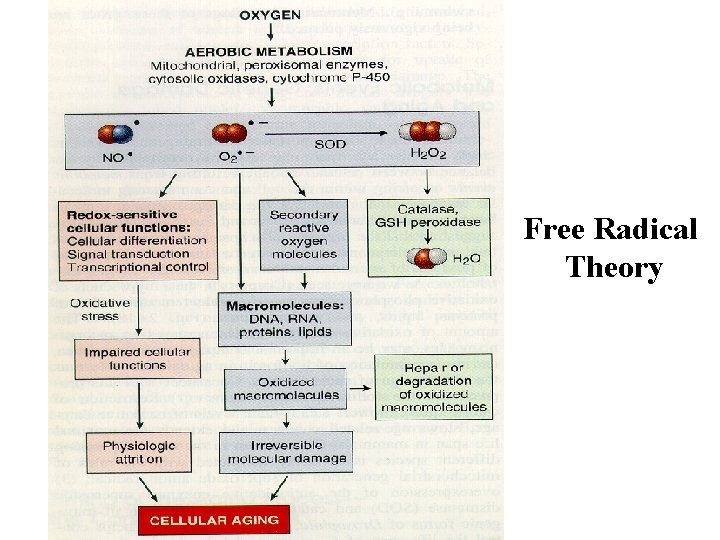
Physical/ Chemical Changes · · Free Radical Theory (Oxidative Damage) – Free Radicals: contain unpaired electrons making them highly reactive therefore only exist for a short time. e. g. Super oxide, hydroxyl , peroxide Lipid peroxidation- damage to cell membranes Protein cross linkage DNA damage Antioxidants - Vitamins A, C, E, Cellular Defenses- Catalase, Superoxide Dismutase

Free Radical Theory

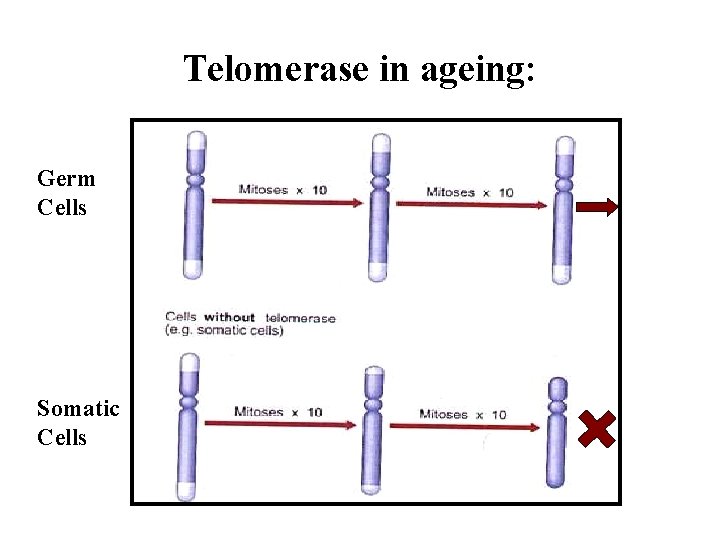
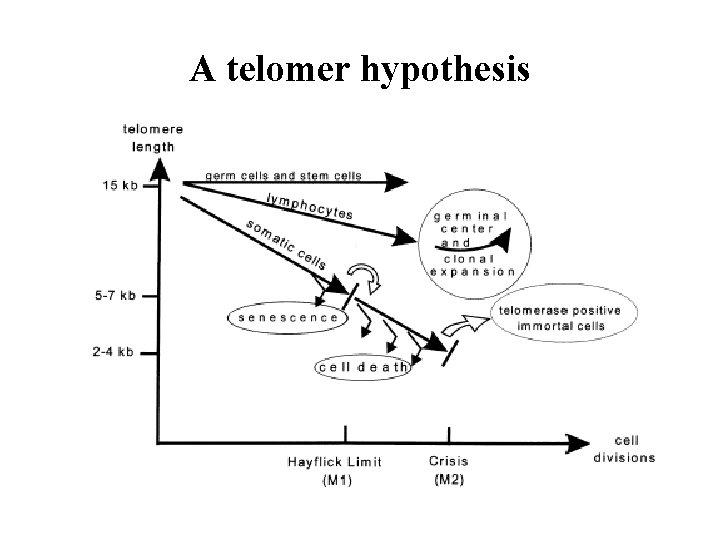
Aging By Program Assumptions: Biological Clock Molecular Clock- the Telomere ? Life Span Inheritable Twin Studies Biological Clock: Hypothalamus Decline in Signal Decreased Sensitivity to Feedback Programmed senescence does not require central control - cell culture evidence

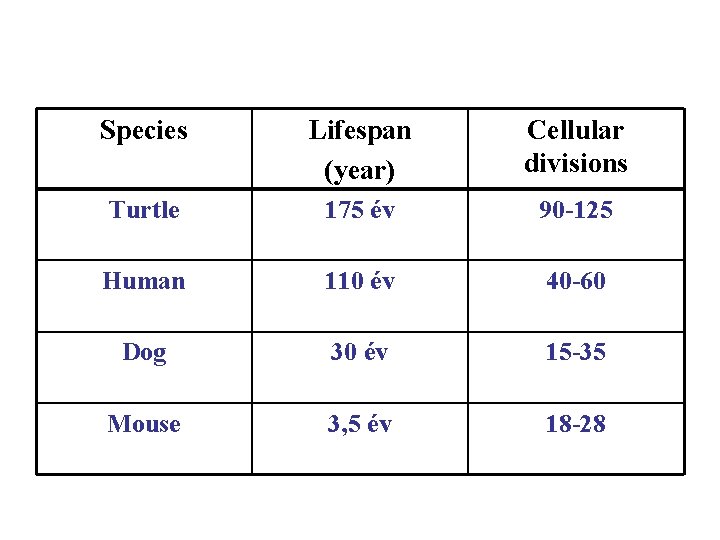
Species Lifespan (year) Cellular divisions Turtle 175 év 90 -125 Human 110 év 40 -60 Dog 30 év 15 -35 Mouse 3, 5 év 18 -28


Telomerase in ageing: Germ Cells Somatic Cells

A telomer hypothesis

Replication Error of the 5’. End Telomer: 5 -15 kb TTAGGGRepeated sequences, shortened during the replications progressively

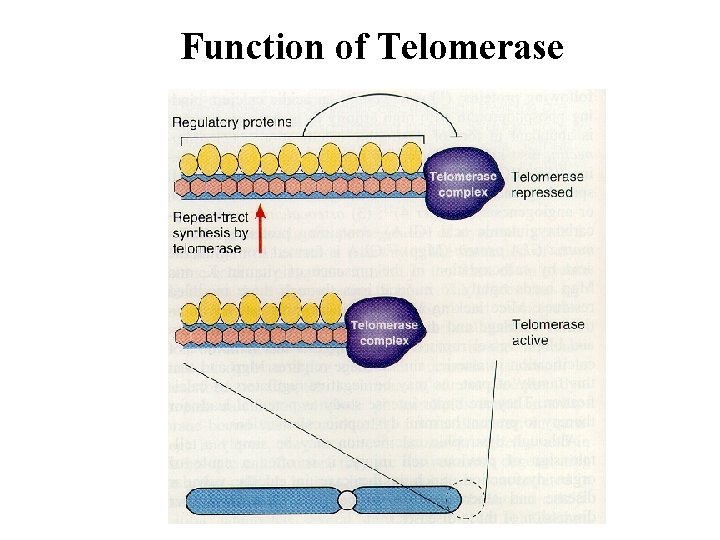
Function of Telomerase

Aging By Program · Gene Mutation Theory- accumulation of somatic cell mutations leads to a decline in function · Liver cells of older mice have more mutations than young · Short life span strains have more mutations at same age DNA repair mechanisms decrease in repair function? ?

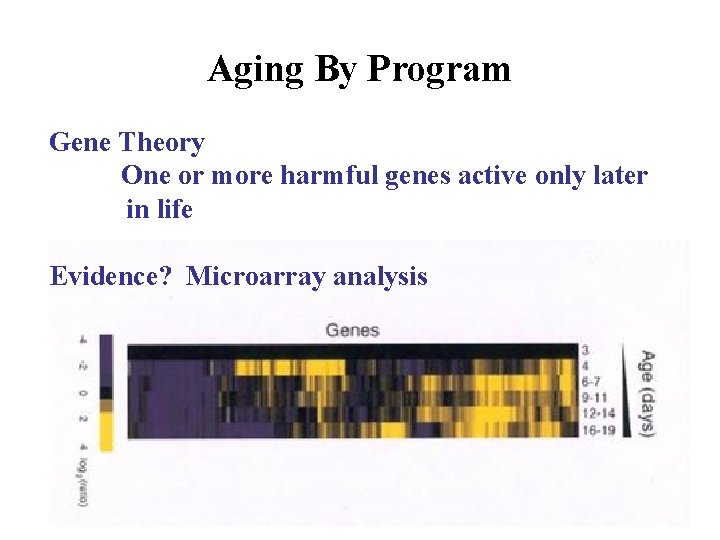
Aging By Program Gene Theory One or more harmful genes active only later in life Evidence? Microarray analysis

For practitioners, based on the biology of ageing · New diseases could have different signs · Clinical manifestations of diseases are more serious, · · · because decreases in reserve capacity – hyperthyreodism heart failure etc. Easier to cure, better respond to therapies Drug induced side effects are more frequent Because a reduction in the capability to adapt occurs the side effects of the diseases worsening the status of the patient more, however any treatment directed again the side effect can improve the life of the patients

For practitioners, based on the biology of ageing · What ever is abnormal in young may be has no clinical meaning in elderly, eg. bacteriuria, decreased glucose tolerance, incontinentia etc. – Bacteruria may not be the cause of the fever from unknown origin. · Difficult to find the right diagnosis because the clinical signs are more complex · Because elderly patients suffer more from the side effects of the diseases, they improve better by the treatment or prevention