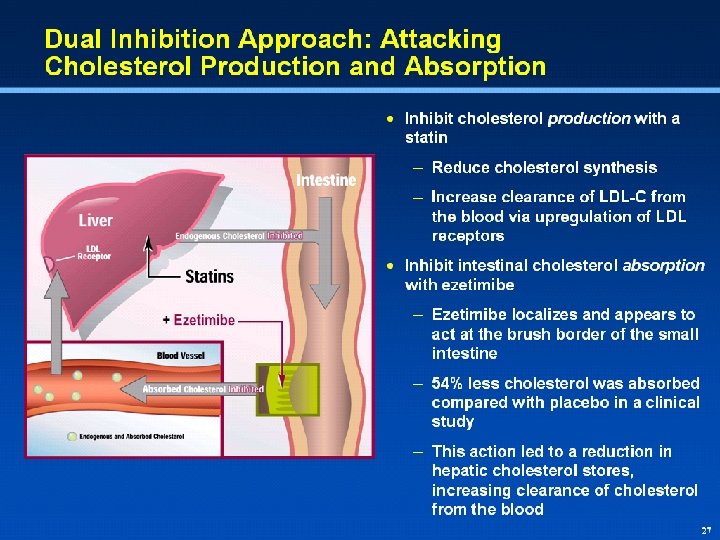
Pathophysiology and Mangement of Dyslipidemias Heidi Chamberlain Shea

Pathophysiology and Mangement of Dyslipidemias Heidi Chamberlain Shea, MD Endocrine Associates of Dallas
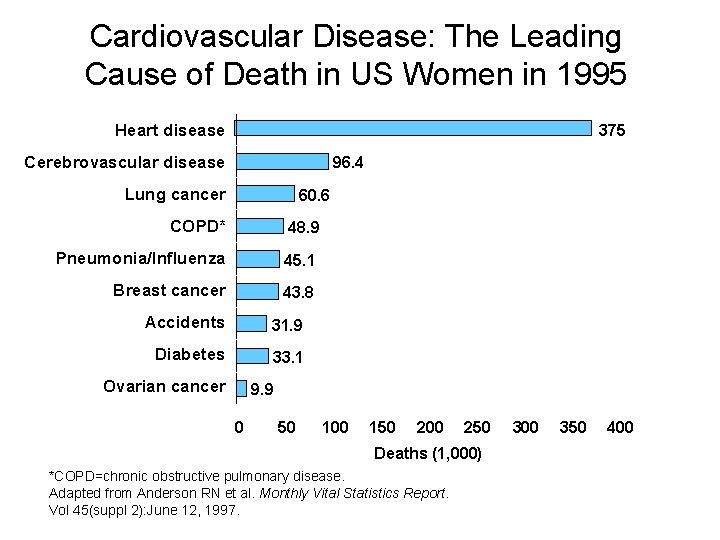
Cardiovascular Disease: The Leading Cause of Death in US Women in 1995 Heart disease 375 Cerebrovascular disease 96. 4 Lung cancer 60. 6 COPD* 48. 9 Pneumonia/Influenza 45. 1 Breast cancer 43. 8 Accidents 31. 9 Diabetes 33. 1 Ovarian cancer 9. 9 0 50 100 150 200 250 Deaths (1, 000) *COPD=chronic obstructive pulmonary disease. Adapted from Anderson RN et al. Monthly Vital Statistics Report. Vol 45(suppl 2): June 12, 1997. 300 350 400

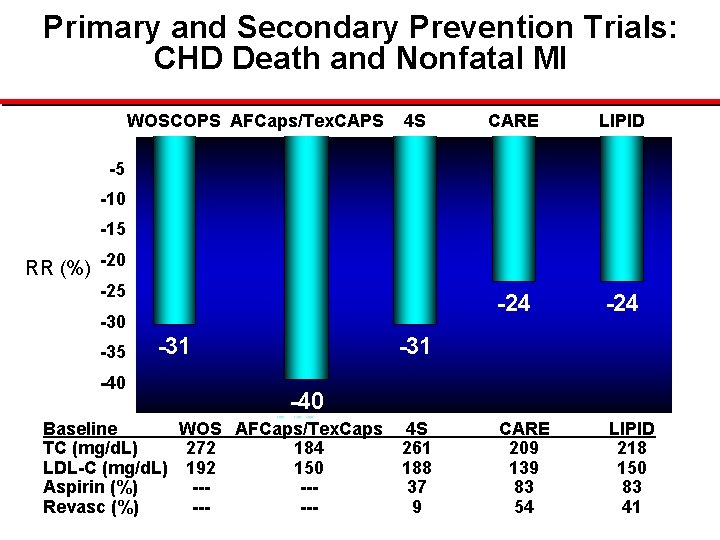
Primary and Secondary Prevention Trials: CHD Death and Nonfatal MI 0 WOSCOPS AFCaps/Tex. CAPS 4 S CARE LIPID -24 CARE 209 139 83 54 LIPID 218 150 83 41 -5 -10 -15 RR (%) -20 -25 -30 -35 -40 -31 -40 -45 Baseline WOS AFCaps/Tex. Caps TC (mg/d. L) 272 184 LDL-C (mg/d. L) 192 150 Aspirin (%) ----Revasc (%) ----- 4 S 261 188 37 9
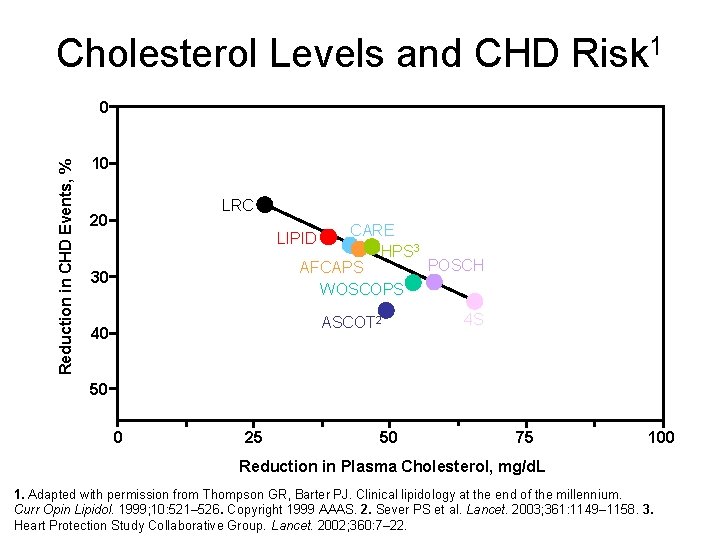
Cholesterol Levels and CHD Risk 1 Reduction in CHD Events, % 0 10 LRC 20 CARE HPS 3 POSCH AFCAPS WOSCOPS LIPID 30 ASCOT 2 40 4 S 50 0 25 50 75 100 Reduction in Plasma Cholesterol, mg/d. L 1. Adapted with permission from Thompson GR, Barter PJ. Clinical lipidology at the end of the millennium. Curr Opin Lipidol. 1999; 10: 521– 526. Copyright 1999 AAAS. 2. Sever PS et al. Lancet. 2003; 361: 1149– 1158. 3. Heart Protection Study Collaborative Group. Lancet. 2002; 360: 7– 22.
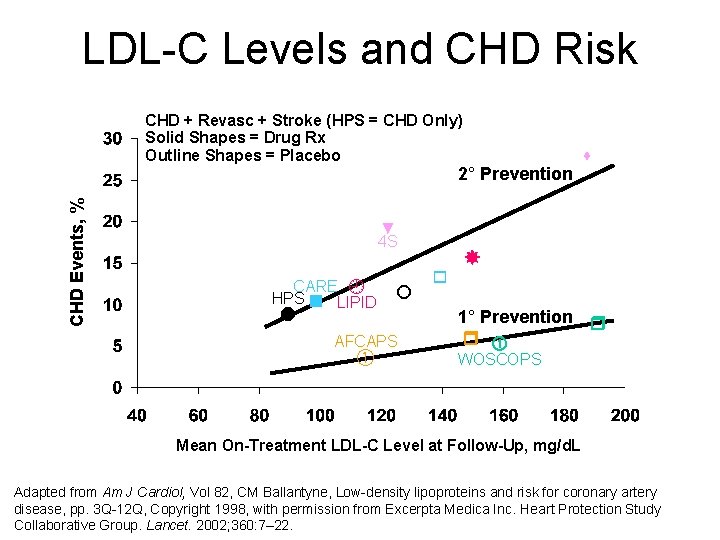
LDL-C Levels and CHD Risk CHD + Revasc + Stroke (HPS = CHD Only) Solid Shapes = Drug Rx Outline Shapes = Placebo CHD Events, % 2° Prevention ▼ 4 S CARE HPS LIPID AFCAPS 1° Prevention WOSCOPS Mean On-Treatment LDL-C Level at Follow-Up, mg/d. L Adapted from Am J Cardiol, Vol 82, CM Ballantyne, Low-density lipoproteins and risk for coronary artery disease, pp. 3 Q-12 Q, Copyright 1998, with permission from Excerpta Medica Inc. Heart Protection Study Collaborative Group. Lancet. 2002; 360: 7– 22.
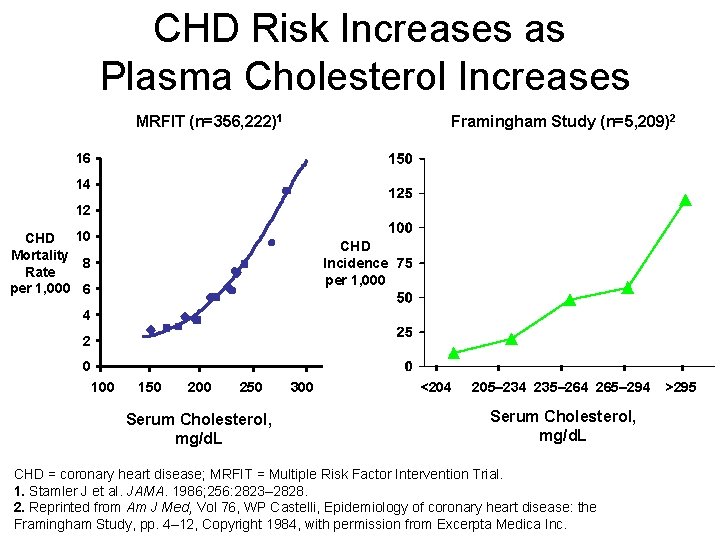
CHD Risk Increases as Plasma Cholesterol Increases MRFIT (n=356, 222)1 Framingham Study (n=5, 209)2 16 14 12 CHD 10 Mortality 8 Rate per 1, 000 6 CHD Incidence per 1, 000 4 2 0 100 150 200 250 Serum Cholesterol, mg/d. L 300 <204 205– 234 235– 264 265– 294 Serum Cholesterol, mg/d. L CHD = coronary heart disease; MRFIT = Multiple Risk Factor Intervention Trial. 1. Stamler J et al. JAMA. 1986; 256: 2823– 2828. 2. Reprinted from Am J Med, Vol 76, WP Castelli, Epidemiology of coronary heart disease: the Framingham Study, pp. 4– 12, Copyright 1984, with permission from Excerpta Medica Inc. >295
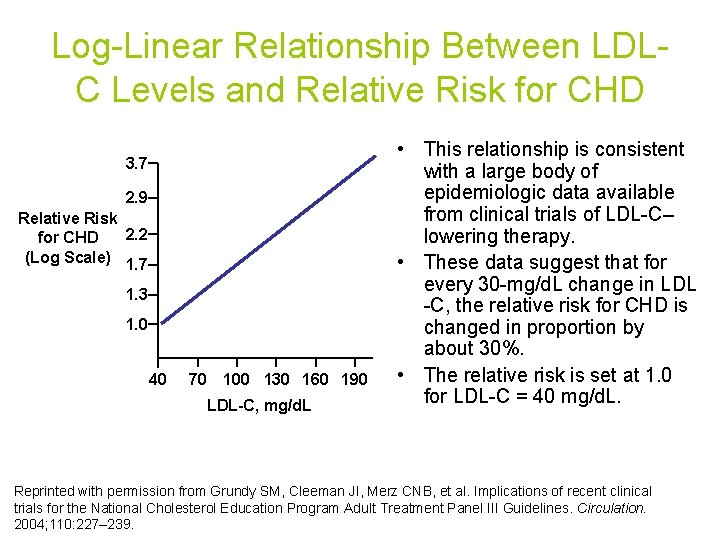
Log-Linear Relationship Between LDLC Levels and Relative Risk for CHD 3. 7 2. 9 Relative Risk 2. 2 for CHD (Log Scale) 1. 7 1. 3 1. 0 40 70 100 130 160 190 LDL-C, mg/d. L • This relationship is consistent with a large body of epidemiologic data available from clinical trials of LDL-C– lowering therapy. • These data suggest that for every 30 -mg/d. L change in LDL -C, the relative risk for CHD is changed in proportion by about 30%. • The relative risk is set at 1. 0 for LDL-C = 40 mg/d. L. Reprinted with permission from Grundy SM, Cleeman JI, Merz CNB, et al. Implications of recent clinical trials for the National Cholesterol Education Program Adult Treatment Panel III Guidelines. Circulation. 2004; 110: 227– 239.
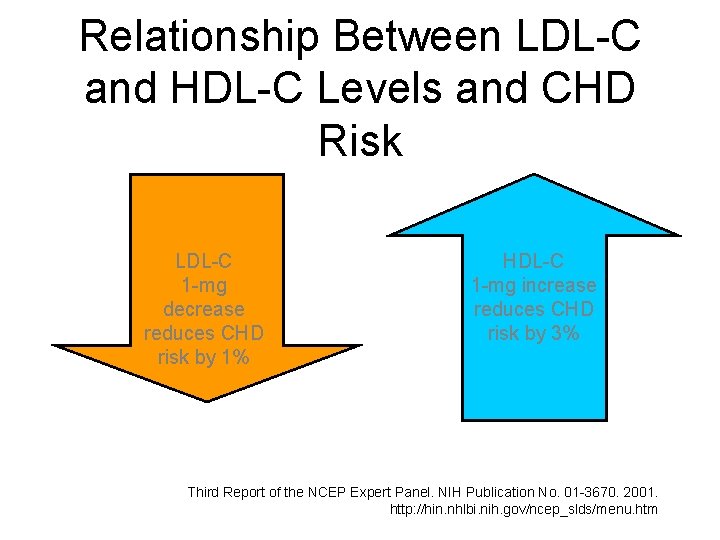
Relationship Between LDL-C and HDL-C Levels and CHD Risk LDL-C 1 -mg decrease reduces CHD risk by 1% HDL-C 1 -mg increase reduces CHD risk by 3% Third Report of the NCEP Expert Panel. NIH Publication No. 01 -3670. 2001. http: //hin. nhlbi. nih. gov/ncep_slds/menu. htm
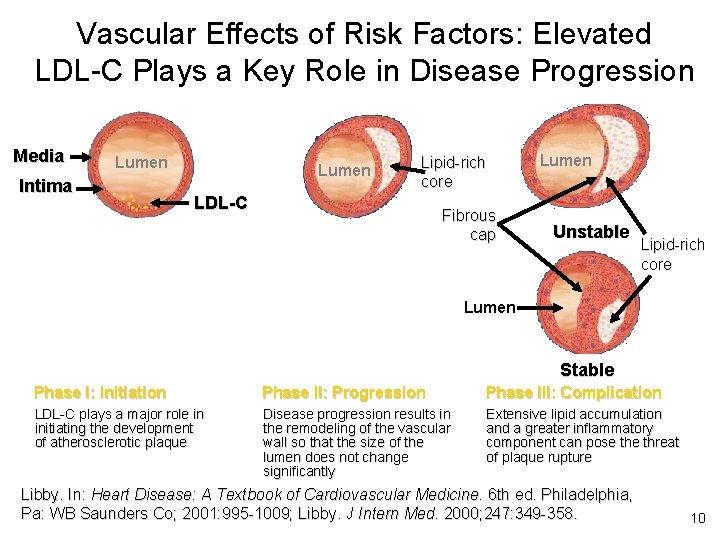
Vascular Effects of Risk Factors: Elevated LDL-C Plays a Key Role in Disease Progression Media Lumen Intima Lumen Lipid-rich core LDL-C Fibrous cap Lumen Unstable Lipid-rich core Lumen Stable Phase I: Initiation Phase II: Progression Phase III: Complication LDL-C plays a major role in initiating the development of atherosclerotic plaque Disease progression results in the remodeling of the vascular wall so that the size of the lumen does not change significantly Extensive lipid accumulation and a greater inflammatory component can pose threat of plaque rupture Libby. In: Heart Disease: A Textbook of Cardiovascular Medicine. 6 th ed. Philadelphia, Pa: WB Saunders Co; 2001: 995 -1009; Libby. J Intern Med. 2000; 247: 349 -358. 10

Discussion Points • Review physiology of cholesterol metabolism • Classification of dyslipidemias • Secondary causes of dyslipidemias • Adult Treatment Panel III (ATP III) • Lipid lowering therapies
Cholesterol Metabolism • Importance – Vital component of all cell structure – Source of energy • Dangers – Abnormal metabolism results in vascular disease
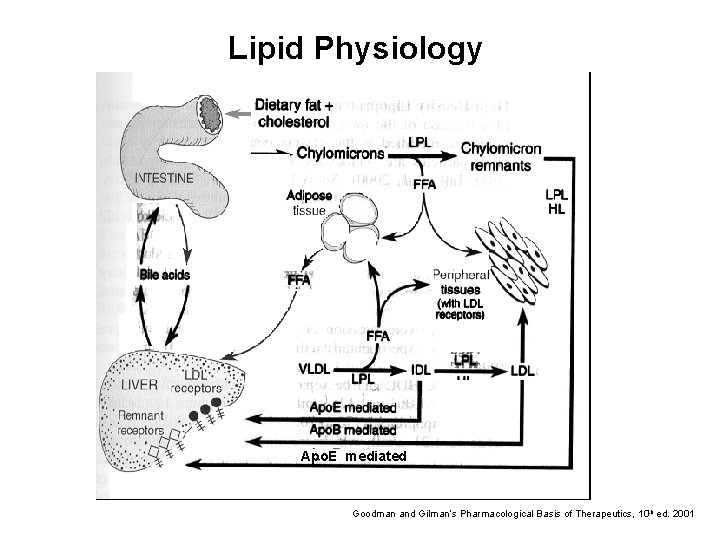
Lipid Physiology Apo. E mediated Goodman and Gilman’s Pharmacological Basis of Therapeutics, 10 th ed. 2001
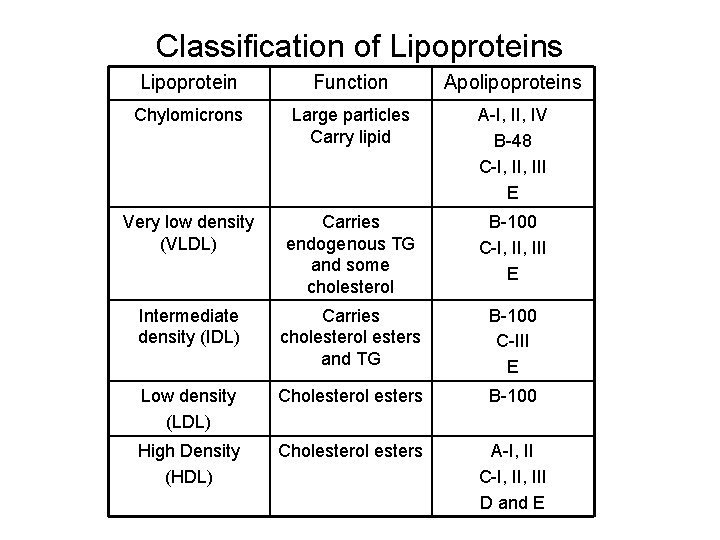
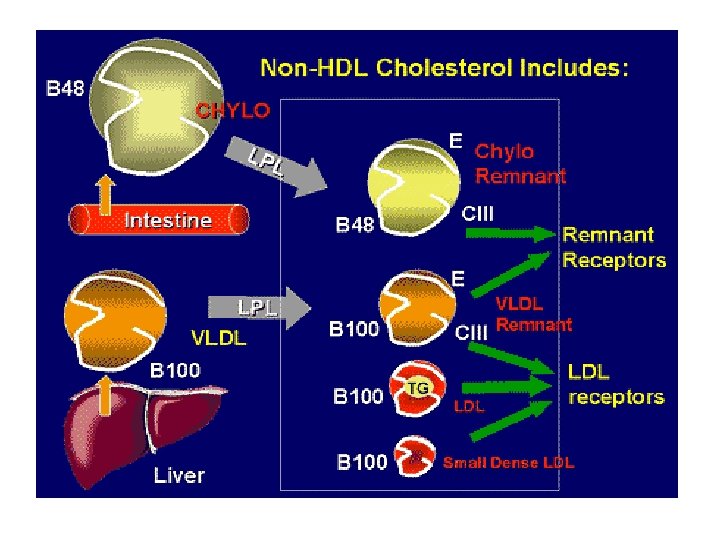
Classification of Lipoproteins Lipoprotein Function Apolipoproteins Chylomicrons Large particles Carry lipid A-I, IV B-48 C-I, III E Very low density (VLDL) Carries endogenous TG and some cholesterol B-100 C-I, III E Intermediate density (IDL) Carries cholesterol esters and TG B-100 C-III E Low density (LDL) Cholesterol esters B-100 High Density (HDL) Cholesterol esters A-I, II C-I, III D and E

Fredrickson Classification of Hyperlipidemias Type Predominant lipoprotein Predominant lipid Xanthoma CHD risk Pancreatitis I Chylomicrons Triglycerides >1000 Eruptive - + IIa LDL Cholesterol >300 Tendon; Xanthelasma + - IIb VLDL + LDL TG and CH <1000 >300 + - III Remnants TG and CH + - IV VLDL CH + - V Chylomicrons + VLDL TG and CH >1000 >300 - + Palmar and tuberous Eruptive


Major Genetic Hyperlipoproteinemias Single gene mutations Disorder Mutant Gene Frequency Inheritance Lipoprotein patterns Familial Hypercholesterolemia (FH) LDL receptor 1/500 AD IIa Familial defective Apo B 1/1000 AD IIa Familial combined hyperlipidemia ? 1/100 AD IIa, IIb, IV Familial Type III Apo E 1/10, 000 AR III

Causes for Hyperlipidemia Secondary • • • Obesity/diet Diabetes (IV, V) Alcohol (IV) Hypothyroidism (II, III) Estrogen therapy (IV) Glucocorticoids (IIa, IIb) Hypopituitarism (IIb) Acromegaly (IV) Anorexia Nervosa (IIa) Lipodystrophy (IV) • Nephrotic syndrome (IIa, IIb) • Hepatitis (IV) • Systemic Lupus Erythematosus (I) • Biliary obstruction or cholestasis (Lp) • Acute intermittent porphyria (IIa) • Glycogen storage disease (IV) • Monoclonal gammopathy (IIa, III, IV)

National Cholesterol Education Program Adult Treatment Panel III (ATP III) Guidelines


NCEP ATP III Treatment Algorithm Assess CHD and CHD risk equivalents No CHD or risk equivalents With CHD or risk equivalents • Treat to LDL-C goal <100 mg/d. L Count CHD risk factors 2 risk factors Framingham risk factor score 1 risk factor • Treat to LDL-C goal <160 mg/d. L >20% 10 -year risk of CHD • CHD risk equivalent • Treat to LDL-C goal <100 mg/d. L 10% to 20% 10 -year risk of CHD • Treat to LDL-C goal <130 mg/d. L <10% 10 -year risk of CHD • Treat to LDL-C goal <130 mg/d. L Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults. JAMA. 2001; 285: 2486 -2497. 28

Emerging Risk Factors • • • Lipoprotein (a) Homocysteine Prothrombotic factors Proinflammatory factors Impaired fasting glucose Subclinical atherosclerosis

Major Risk Factors That Modify LDL Goal (Exclusive of LDL Cholesterol) • Cigarette smoking • Hypertension (BP 140/90 mm. Hg or on antihypertensive medication) • Low HDL cholesterol (< 40 mg/d. L)† • Family history of premature CHD – CHD in male first degree relative < 55 years – CHD in female first degree relative < 65 years • Age (men 45 years; women 55 years) † HDL cholesterol 60 mg/d. L counts as a “negative” risk factor; its presence removes one risk factor from the total count.

Case Study: 52 yr old Male with Multiple Risk Factors • • • Asymptomatic Non smoker BMI 26 HIV positive BP 136/84 – Diltiazem • Lipid Profile – – TC: 240 mg/d. L TG: 180 mg/d. L HDL: 42 mg/d. L LDL: 158 mg/d. L
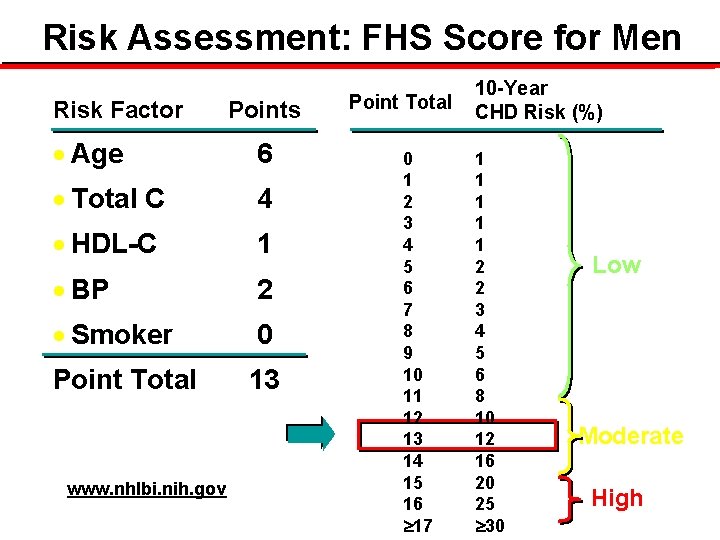
Risk Assessment: FHS Score for Men Risk Factor Points · Age 6 · Total C 4 · HDL-C 1 · BP 2 · Smoker 0 Point Total 13 www. nhlbi. nih. gov Point Total 0 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 10 -Year CHD Risk (%) 1 1 1 2 2 3 4 5 6 8 10 12 16 20 25 30 Low Moderate High
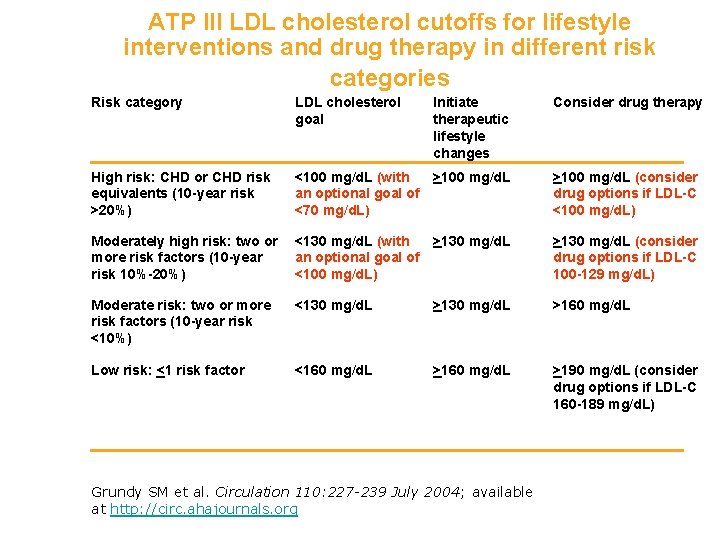
ATP III LDL cholesterol cutoffs for lifestyle interventions and drug therapy in different risk categories Risk category LDL cholesterol goal Initiate therapeutic lifestyle changes High risk: CHD or CHD risk equivalents (10 -year risk >20%) <100 mg/d. L (with >100 mg/d. L an optional goal of <70 mg/d. L) >100 mg/d. L (consider drug options if LDL-C <100 mg/d. L) Moderately high risk: two or more risk factors (10 -year risk 10%-20%) <130 mg/d. L (with >130 mg/d. L an optional goal of <100 mg/d. L) >130 mg/d. L (consider drug options if LDL-C 100 -129 mg/d. L) Moderate risk: two or more risk factors (10 -year risk <10%) <130 mg/d. L >160 mg/d. L Low risk: <1 risk factor <160 mg/d. L >190 mg/d. L (consider drug options if LDL-C 160 -189 mg/d. L) Grundy SM et al. Circulation 110: 227 -239 July 2004; available at http: //circ. ahajournals. org Consider drug therapy

ATP lll UPDATE • Update based on findings of 5 new trials which addressed issues not adequately addressed in prior statin trials: – Heart Protection Study (HPS) – Prospective Study of Pravastatin in the Elderly at Risk (PROSPER) – Anti-Hypertensive and Lipid Lowering Treatment to Prevent Heart Attack Trial - Lipid Lowering Trial (ALLHAT-LLT) – Anglo-Scandinavian Cardiac Outcomes Trial – Lipid Lowering Arm (ASCOT – LLA) – Pravastatin or Atorvastatin Evaluation and Infection – Thrombolysis in Myocardial Infarction 22 (PROVE IT – TIMI 22) Grundy SM et al. Circulation 110: 227 -239 July 2004; available at http: //circ. ahajournals. org

Rationale For Optional Very Low LDL goal (<70) • ATP lll considered an LDL level of 100 mg/dl to be a minimal goal of treatment for high risk patients. • HPS & PROVE-IT suggest that additional benefit may be obtained by reducing LDL levels to substantially below 100 mg/dl. • Several other clinical trials are underway to determine the efficacy of lowering LDL to very low levels. • Recent clinical trials with statin therapy have shown no significant side effects from LDL lowering. Grundy SM et al. Circulation 110: 227 -239 July 2004; available at http: //circ. ahajournals. org

FACTORS THAT FAVOR A DECISION TO REDUCE LDL LEVELS TO <70 MG/DL • VERY HIGH RISK-established CHD plus: – Multiple major risk factors, especially diabetes – Severe and poorly controlled risk factors, especially continued smoking – Multiple risk factors of the metabolic syndrome – Patients with acute coronary syndromes Grundy SM et al. Circulation 110: 227 -239 July 2004; available at http: //circ. ahajournals. org

Specific Dyslipidemias: Elevated Triglycerides Non-HDL Cholesterol: Secondary Target • Non-HDL cholesterol = VLDL + LDL cholesterol – Total Cholesterol – HDL cholesterol • VLDL cholesterol: – Atherogenic remnant lipoproteins • Non-HDL cholesterol: – Secondary target of therapy when serum triglycerides are 200 mg/d. L (esp. 200– 499 mg/d. L) • Non-HDL cholesterol goal: – LDL-cholesterol goal + 30 mg/d. L

Specific Dyslipidemias: Low HDL Cholesterol Management of Low HDL Cholesterol • LDL cholesterol is primary target of therapy • Weight reduction and increased physical activity (if the metabolic syndrome is present) • Non-HDL cholesterol is secondary target of therapy (if triglycerides 200 mg/d. L) • Consider nicotinic acid or fibrates (for patients with CHD or CHD risk equivalents)

Benefit Beyond LDL Lowering: The Metabolic Syndrome as a Secondary Target of Therapy General Features of the Metabolic Syndrome • Abdominal obesity • Atherogenic dyslipidemia – – – • • Elevated triglycerides Small LDL particles Low HDL cholesterol Raised blood pressure Insulin resistance ( glucose intolerance) Prothrombotic state Proinflammatory state
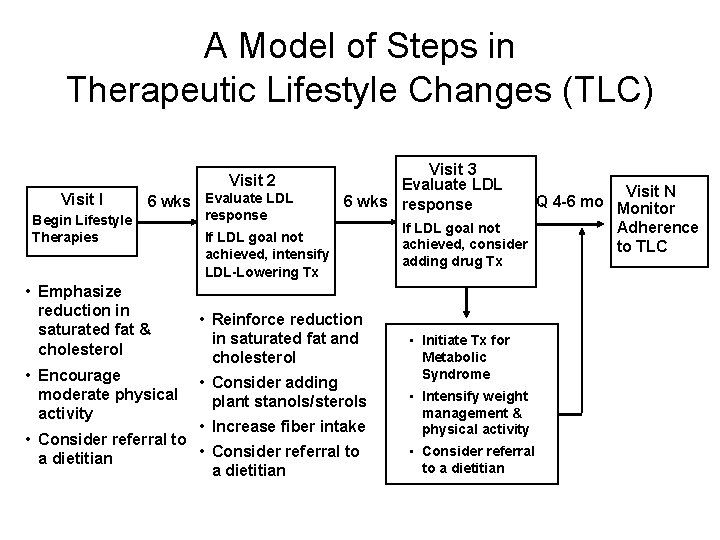
A Model of Steps in Therapeutic Lifestyle Changes (TLC) Visit 2 Visit I 6 wks Evaluate LDL Begin Lifestyle Therapies • Emphasize reduction in saturated fat & cholesterol • Encourage moderate physical activity response Visit 3 Evaluate LDL 6 wks response If LDL goal not achieved, intensify LDL-Lowering Tx • Reinforce reduction in saturated fat and cholesterol • Consider adding plant stanols/sterols • Increase fiber intake • Consider referral to a dietitian Visit N Q 4 -6 mo Monitor If LDL goal not Adherence achieved, consider to TLC adding drug Tx • Initiate Tx for Metabolic Syndrome • Intensify weight management & physical activity • Consider referral to a dietitian

Implications of New Therapeutic Guidelines • Recent clinical trials indicate that for every 1% reduction of LDL levels the relative risk for major CHD events is reduced by approximately 1%. • Currently available statins at doses typically used in these trials will lower LDL levels by 30 -40% which translates into a similar percentage reduction in CHD risk over a 5 year period. • “It is reasonable to employ statin doses adequate to achieve a reduction in risk for major coronary events of 30 -40%. • The statin doses that produce such reductions are termed standard doses in ATPIII, 7/04. Grundy SM et al. Circulation 110: 227 -239 July 2004; available at http: //circ. ahajournals. org

Clinical Trial Findings: The Statins • Statins LDL-C by 25%-35% (achieved in clinical event trials and regression studies) • Benefits at various LDL-C levels; evident soon after therapy in some studies • in LDL-C required for in CHD morbidity/mortality • in all-cause mortality in 2° prevention and in cardiovascular mortality in 1° prevention • Studies support treatment in various patient groups – women – elderly – diabetics

Safety Summary for HMG-Co. A Reductase Inhibitors • Excellent patient acceptance • Few drug-drug interactions • Few side effects – most common are gastrointestinal: mild to moderate – at high doses, elevated ALT/AST in 1% to 2% – myopathy reported in 0. 1% (CK >10 x ULN) • Only rare cases of toxicity • No increases in total or non-CHD mortality Lovastatin Study Groups I through IV. Arch Intern Med. 1993; 153: 1079 -1087. Second Report of the Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults (ATP II). September 1993; NIH Publication 93 -3095.

Statins: Standard Dosing • • • Atorvastatin Lovastatin Pravastatin Simvastatin Fluvastatin Rosuvastatin • • • 10 mg 40 mg 20 -40 mg 40 -80 mg 5 -10 mg Grundy SM et al. Circulation 110: 227 -239 July 2004; available at http: //circ. ahajournals. org

Fibric Acid Derivatives • Utility – HDL, TG – Coronary events (Helsinki Heart Study, VA-HIT, BIP, DAIS) • Limitations – Gallstones, myositis, hepatotoxicity, renal impairment – Drug interactions with warfarin – Gemfibrozil • Glucose monitoring – Increases bioavailability of statins

Fibric Acids Drug • Gemfibrozil • Fenofibrate • Clofibrate BID Dose 600 mg BID 200 mg QD 1000 mg
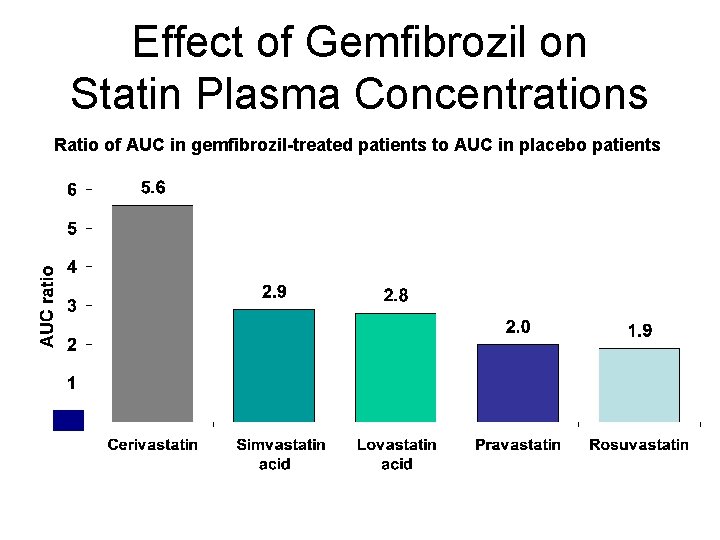
Effect of Gemfibrozil on Statin Plasma Concentrations Ratio of AUC in gemfibrozil-treated patients to AUC in placebo patients
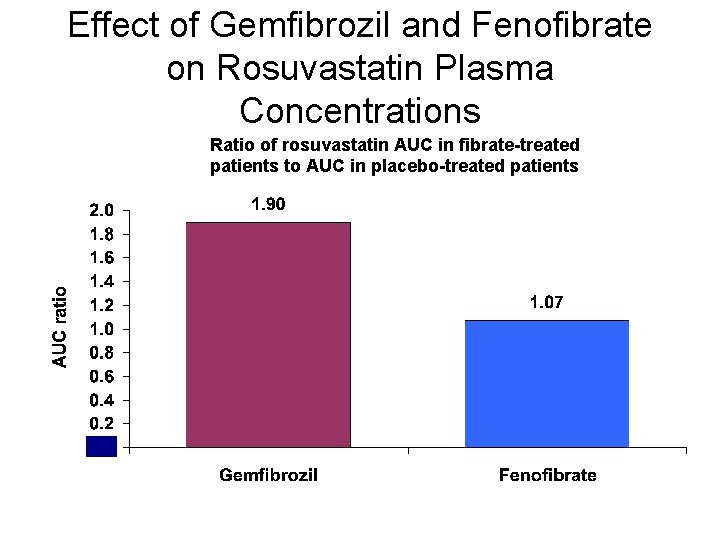
Effect of Gemfibrozil and Fenofibrate on Rosuvastatin Plasma Concentrations Ratio of rosuvastatin AUC in fibrate-treated patients to AUC in placebo-treated patients

Nicotinic Acid • Utility – – – HDL TC, LDL, TG, Apo B Coronary events (Coronary Drug Project) – Combines with conventional bile acid sequestrants • Limitations – Severe flushing, headache, GI and hepatotoxicity – Contraindicated: LFT’s, liver disease – Frequent LFT- every 612 weeks in first year – Combination with statin increases risk of rhabdomyolysis
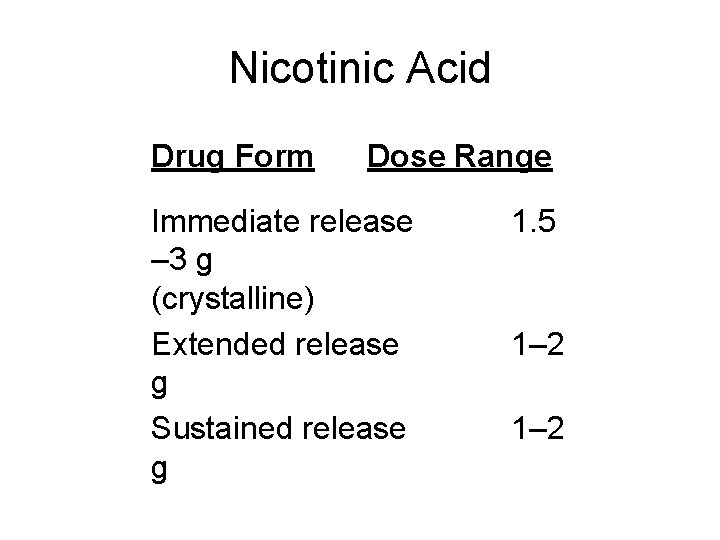
Nicotinic Acid Drug Form Dose Range Immediate release – 3 g (crystalline) Extended release g Sustained release g 1. 5 1– 2

Bile Acid Sequestrants • Utility – – LDL Coronary events • Limitations – Constipation and flatulence – Drug interactions: blocks absorption of statins and vitamins – 1 hour before or 4 hours after other meds – Granules unpalatable – Tablets are large – 41%-63% discontinuance

Bile Acid Sequestrants Drug Dose Range Cholestyramine 4– 16 g Colestipol 5– 20 g Colesevelam 2. 6– 3. 8 g

NEW THERAPIES
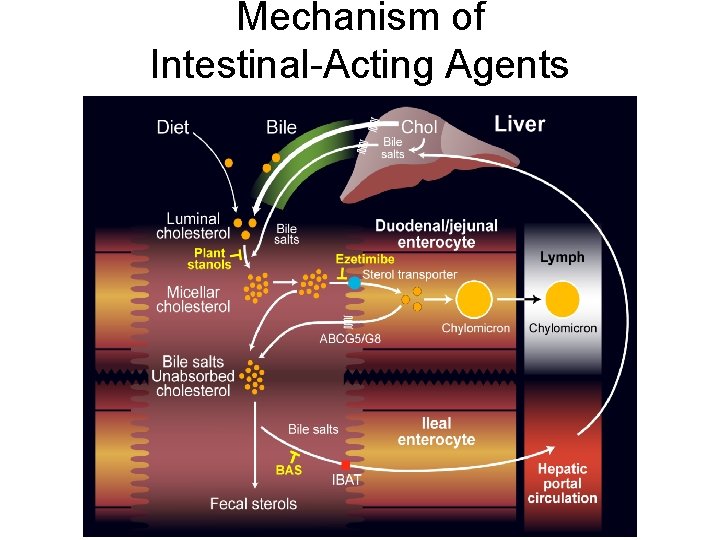
Mechanism of Intestinal-Acting Agents
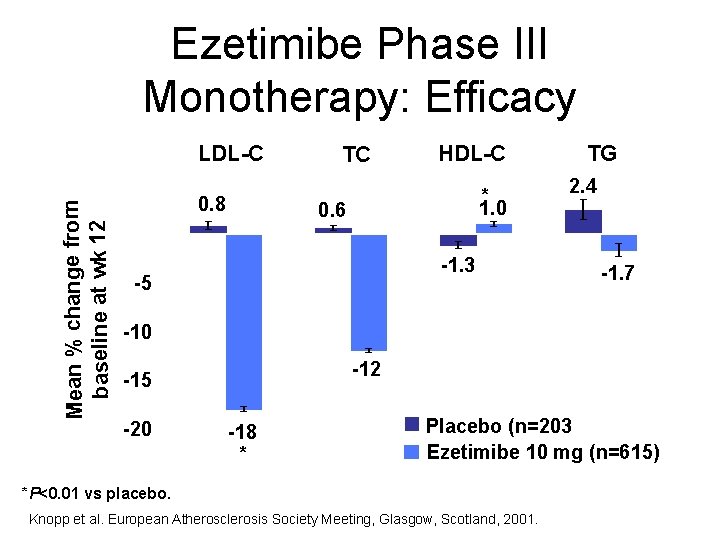
Ezetimibe Phase III Monotherapy: Efficacy LDL-C Mean % change from baseline at wk 12 5 0. 8 TC HDL-C * TG 2. 4 1. 0 0. 6 0 -1. 3 -5 -1. 7 -10 -12 -15 -20 -18 * Placebo (n=203) Ezetimibe 10 mg (n=615) *P<0. 01 vs placebo. Knopp et al. European Atherosclerosis Society Meeting, Glasgow, Scotland, 2001.
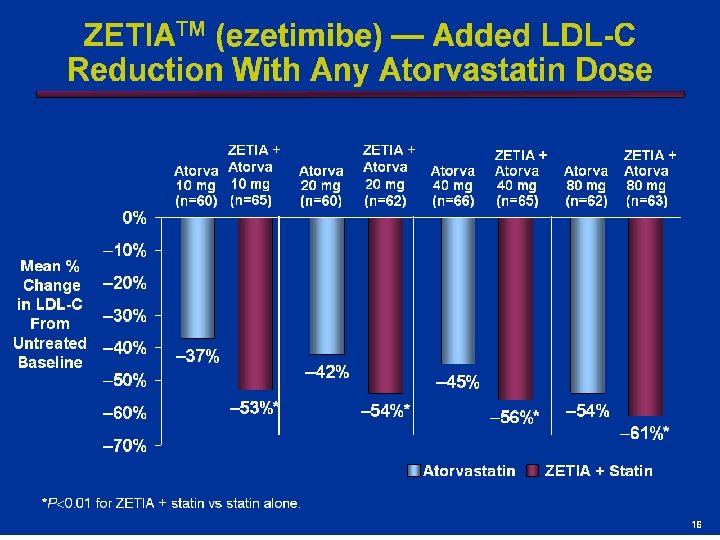
Lisa Shaw: Deleted graph headline per M/L
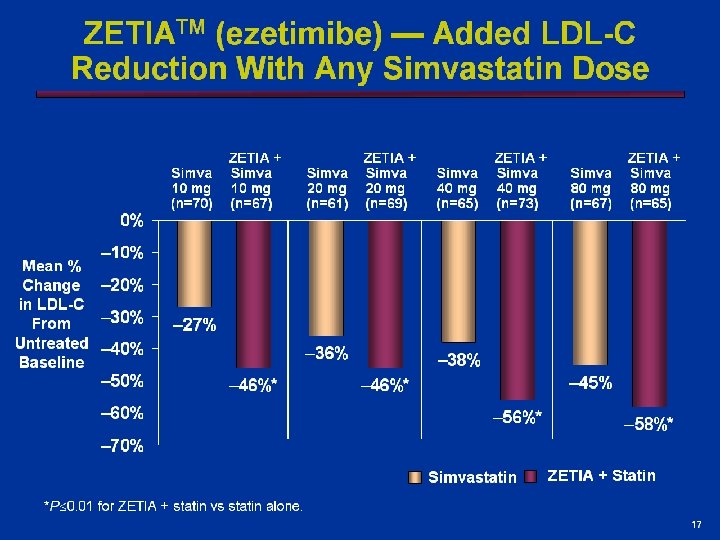
Lisa Shaw: Deleted graph headline per M/L

ACP Guidelines: Lipid Control in Diabetics Recommendation 1: Lipid-lowering therapy should be used for secondary prevention of cardiovascular mortality and morbidity for all patients (both men and women) with known coronary artery disease and type 2 diabetes. Recommendation 2: Statins should be used for primary prevention against macrovascular complications in patients (both men and women) with type 2 diabetes and other cardiovascular risk factors.

ACP GUIDELINES: Lipid Control in Diabetics Recommendation 3: Once lipid-lowering therapy is initiated, patients with type 2 diabetes mellitus should be taking at least moderate doses of a statin. Recommendation 4: For those patients with type 2 diabetes who are taking statins, routine monitoring of liver function tests or muscle enzymes is not recommended except in specific circumstances.
- Slides: 54