Pathology and Imaging In Biomarker Development C Carl

Pathology and Imaging In Biomarker Development C. Carl Jaffe, MD, FACC Cancer Imaging Program National Cancer Institute

Biomarker NIH Workshop definition (2001): a characteristic that is objectively measured … as an indicator of normal biologic or pathogenic processes or pharmacological responses to a therapeutic intervention CIP NCIA

Linguistic distinctions • biomarker • prognostic • predictive • ‘qualified’ biomarker • ‘surrogate’ marker CIP NCIA

Types of Biomarkers • Prognostic -- portend disease outcome at time of diagnosis without reference to any specific therapy • Predictive -- predict outcome of a particular therapy • Monitoring-- measure response to treatment and early detect disease progression or relapse CIP NCIA

Predictive vs Prognostic • Predictive markers can be used to make decisions about specific treatments • are essential for adaptive trial design • a predictive marker may not be prognostic if it does not predict outcome in untreated patients • Prognostic markers may not be predictive • i. e. doesn’t interact with particular treatment CIP NCIA
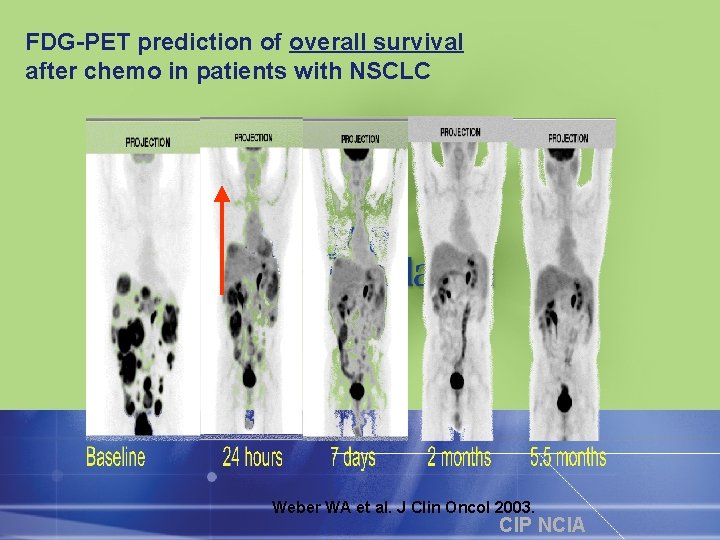
FDG-PET prediction of overall survival after chemo in patients with NSCLC Weber WA et al. J Clin Oncol 2003. CIP NCIA

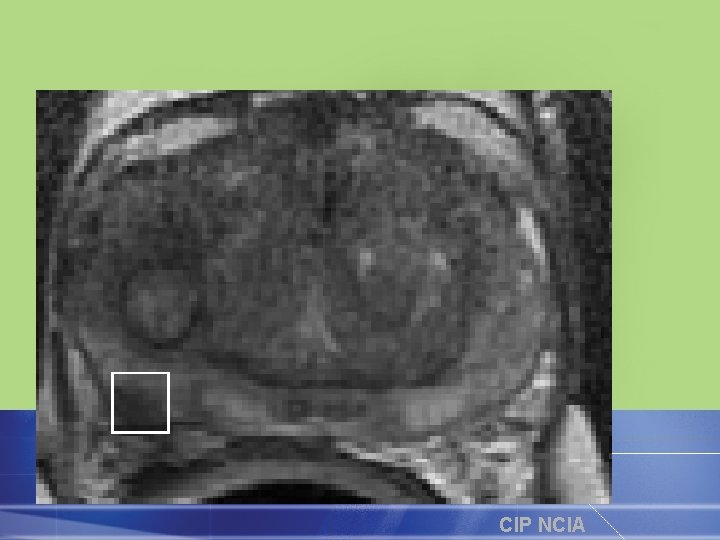
FDG-PET Monitoring Response to Gleevec in GIST Baseline 24 hrs 7 days 2 mos 5. 5 mos Dana-Farber Cancer Institute CIP NCIA

“Surrogate” biomarker • Biomarker used in place of definitive endpoint • May be observed earlier than definitive endpoint CIP NCIA

CIP NCIA

Context: Current Oncology Drugs Failure rate and development costs are high: >80% of drugs entering clinical development fail to get marketing approval 50% of new drugs reaching Phase III trials fail Development costs per drug from discovery through Phase III has been estimated at $0. 8– 1. 7 billion requiring 8– 10 years of time For new molecularly targeted oncology drugs, there are specific development issues Very promising oncology drugs may be effective only in selected cancer patients or risk groups Inhibition of critical signal transduction pathways may lead to collateral toxicity CIP NCIA

Biomarker Consortium OBQI - public-private partnerships • coordinated by Foundation for the NIH through the Biomarker Consortium, - a larger public-private partnership to promote discovery, development, qualification, and regulatory acceptance of biomarkers; • make research results and data arising under consortium projects publicly available • develop safe, innovative, and effective medicines and diagnostics to improve medical care, and improve public health. CIP NCIA

CIP NCIA

CIP NCIA

In this context – How might Imaging Informatics and Digital Imaging help? • • Image storage and transmission Distributed network communication Database biospecimens Integrate the broader healthcare record and enterprise • Enable performance auditing CIP NCIA

ca. BIG objectives software suite that provides a means of capturing, storing and sharing medical images. confederated archive for images and related data connected interoperably Clinical Research Molecular Biology Imaging Pathology CIP NCIA

050107 ca. TISSUE Suite An enhanced application for biospecimen management CIP NCIA

ca. Tissue Suite • Enhanced Collection Protocol Definition Pre-define specimen processing schemes Define multiple study arms and time points • Facilitated Specimen Accession Pre-defined specimen and specimen-related data creation • Collection Protocol Consent Tracking • Pathology Annotation (CAE) CAP protocol pathology annotation for major organ systems • ca. TIES-like Pathology Report Annotation • Custom Annotation (Dynamic Extensions) • Advanced Query “Wizard” Create and save complex, pre-defined or parameterized searches • Specimen Requisition and Request Tracking CIP NCIA
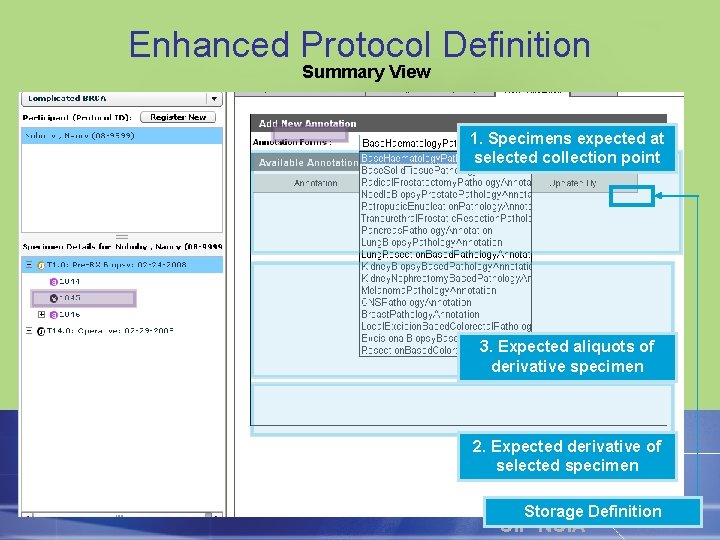
Enhanced Protocol Definition Summary View 1. Specimens expected at selected collection point 3. Expected aliquots of derivative specimen 2. Expected derivative of selected specimen Storage Definition CIP NCIA
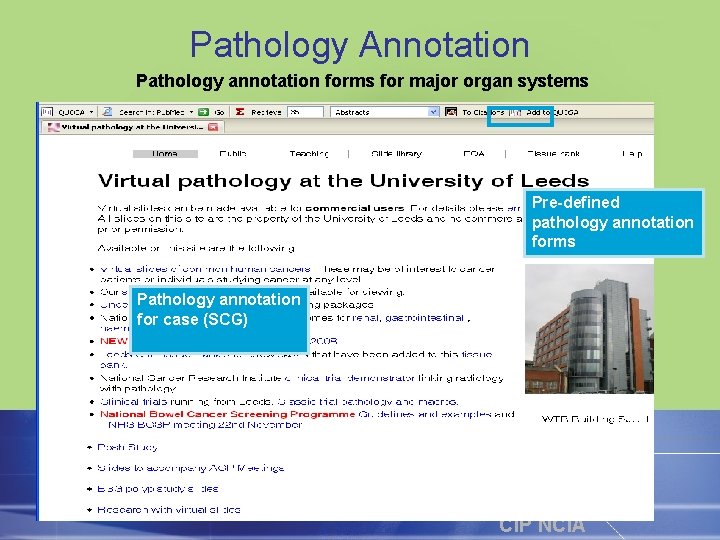
Pathology Annotation Pathology annotation forms for major organ systems Pre-defined pathology annotation forms Pathology annotation for case (SCG) CIP NCIA

ca. Tissue Suite v 1. 0 • Demonstration Site: http: //catissuecore. wustl. edu • Application release: 4/15/2008 • What’s next – Usability enhancements Security and control for multi-bank user environment Improved custom form generation Temporal queries Other enhancements based on user feedback CIP NCIA

CIP NCIA
CIP NCIA
CIP NCIA
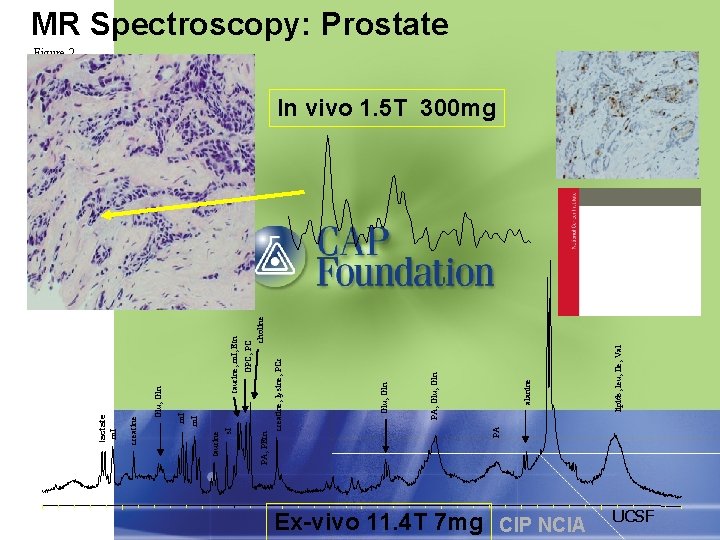
taurine, m. I, Etn GPC, PC choline Ex-vivo 11. 4 T 7 mg CIP NCIA lipids, leu, Ile, Val alanine PA, Glu, Gln creatine, lysine, PCr PA PA, PEtn taurine s. I m. I Glu, Gln creatine lactate m. I MR Spectroscopy: Prostate Figure 2 In vivo 1. 5 T 300 mg lactate UCSF
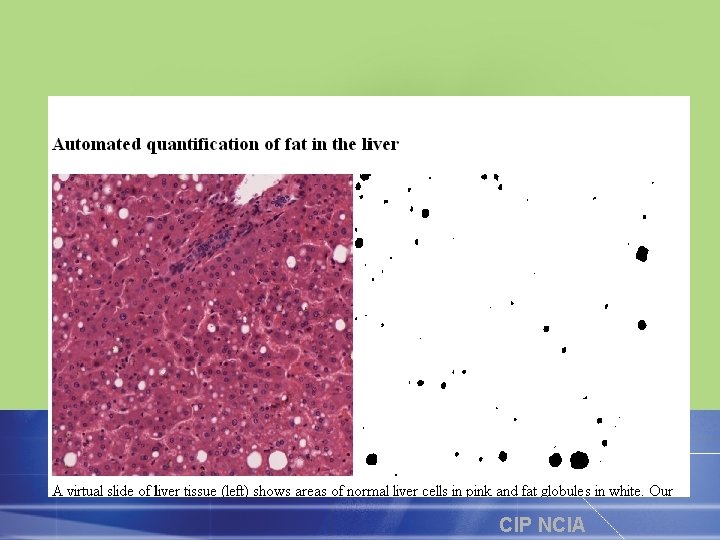
CIP NCIA

CIP NCIA
CIP NCIA

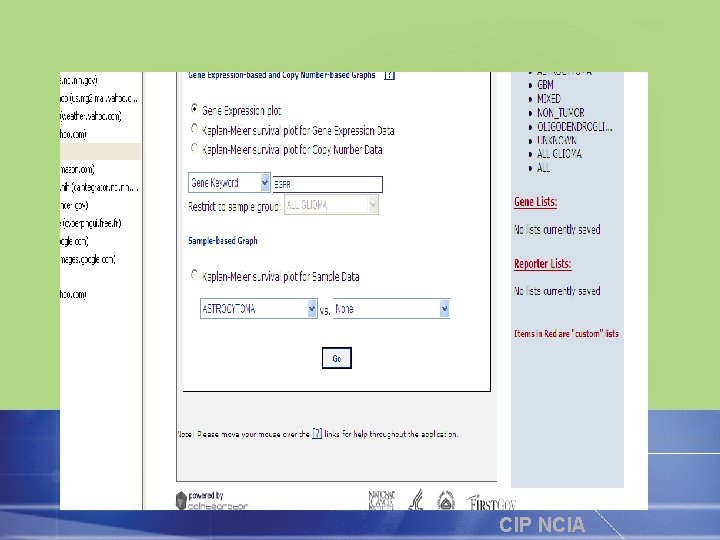
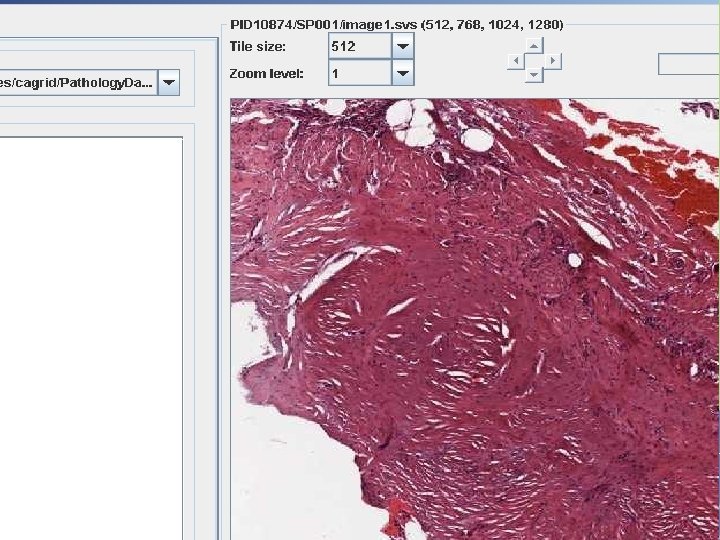
National Cancer Institute Imaging Archive • repository for oncology image data including ongoing and former clinical trials, reference image collections and phantom data • Image visualization, interpretation and mark-up tool • A project to develop free and open source software for acquisition, archival and flexible distribution of images and related data via: • • Internet portal ca. GRID DICOM Query Retrieve API CIP NCIA
CIP NCIA
CIP NCIA

How Does It Fit The “Big” Picture? • ca. BIG modules: • ca. Tissue: manage users, authentication/authorization, specimen registration, search, and specimen distribution. • ca. Microscope: image viewer, data services, and image streaming. • ca. Microsocpe • Will host the data service as a ca. Grid service • Uses Grid. FTP to stream large images CIP NCIA

What are the unresolved challenges ? CIP NCIA

Annotation is a challenge CIP NCIA
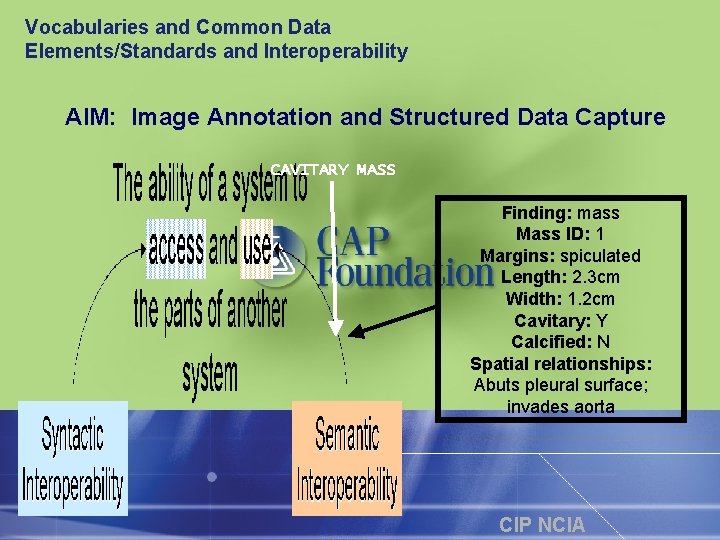
Vocabularies and Common Data Elements/Standards and Interoperability AIM: Image Annotation and Structured Data Capture CAVITARY MASS Finding: mass Mass ID: 1 Margins: spiculated Length: 2. 3 cm Width: 1. 2 cm Cavitary: Y Calcified: N Spatial relationships: Abuts pleural surface; invades aorta CIP NCIA

Common problem: Lack of a radiology Lexicon/Ontology • Limited radiology terminology in Snomed CT (Systematized Nomenclature of Medicine Clinical Terms) or UMLS (Unified Medical Language System) • Current general medical lexicons only include about 20% of terms used in radiology reports • Don’t have consensus on acquisition parameters such as MRI sequences including GRASS, ROAST, etc. to describe acquisition standards CIP NCIA

Lesson 5: Making a Tool ca. BIG™ Compatible What is Data Compatibility? • ca. BIG™ compatibility is about using standards to ensure interoperability among tools – so that data can be exchanged and understood between systems. CIP NCIA


TRANSFORMING PATHOLOGY: Emerging technology driving practice innovation
- Slides: 38