Patents health focus on access WHO TBS June

Patents & health: focus on access – WHO TBS June 2017 Dr Peter Beyer, Senior Advisor 1| Public health, innovation and intellectual property

Patent l Gives the right to the inventor to exclude others from commercially using the invention against disclosure l Claims define the scope and can differ from one country to the other depending on examination l Only what is claimed is protected: a patent on use of an antibiotic to treat ophthalmological infections does not prevent marketing a generic tablet; patent on polymorph B does not prevent a tablet using polymorph A 2| Public health, innovation and intellectual property

Patents l There is nothing such as a worldwide patent! l WIPO Patent Cooperation Treaty allows for worldwide filing, but applicant receives individual national/regional patents 3| Public health, innovation and intellectual property

One drug = one patent? "…a key element of life cycle management strategies is to extent patent protection for as long as possible by filing secondary patents to keep generics off the market" (Burdon and Sloper, The art of using secondary patents to improve protection, International Journal of Medical Marketing, Volume 3, Number 3, June 2003, pp. 226 -238(13)
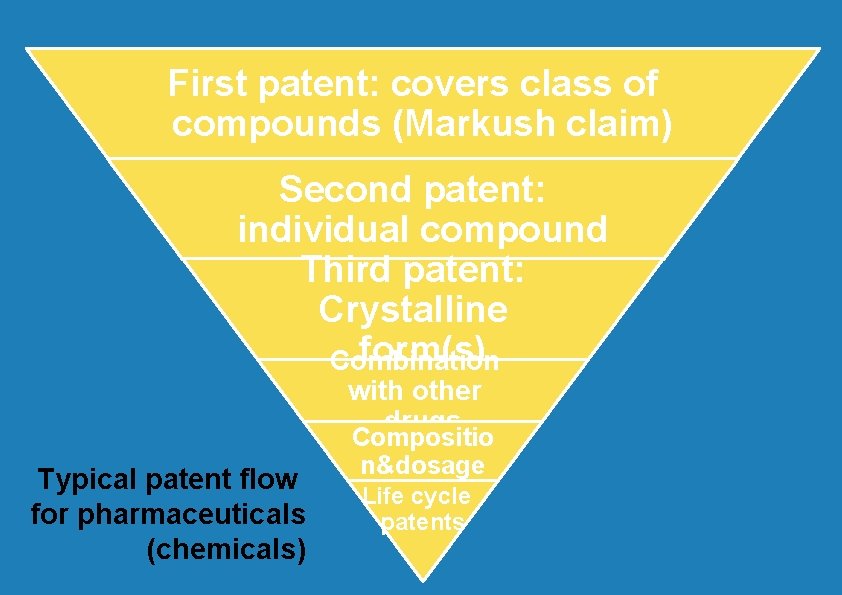
First patent: covers class of compounds (Markush claim) Second patent: individual compound Third patent: Crystalline form(s) Combination with other drugs Typical patent flow for pharmaceuticals (chemicals) Compositio n&dosage Life cycle patents
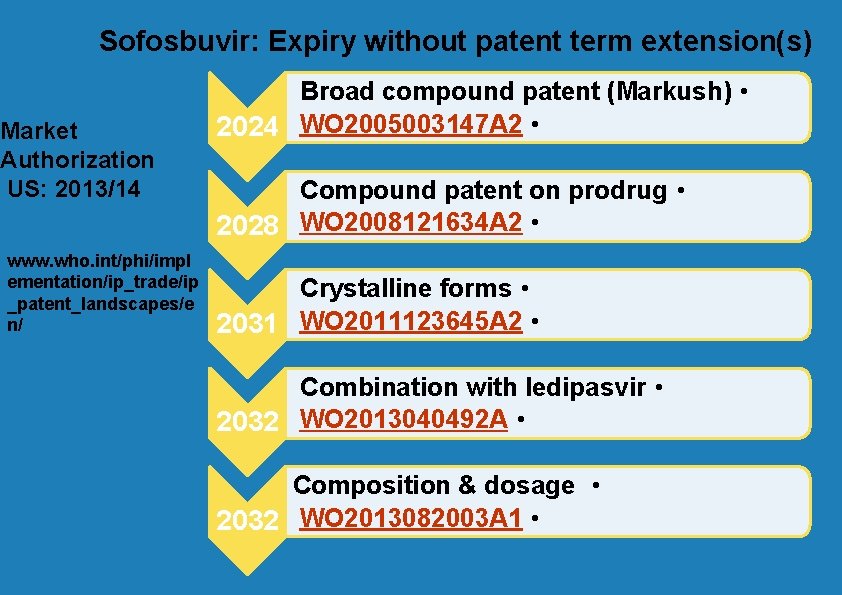
Sofosbuvir: Expiry without patent term extension(s) Market Authorization US: 2013/14 www. who. int/phi/impl ementation/ip_trade/ip _patent_landscapes/e n/ Broad compound patent (Markush) • 2024 WO 2005003147 A 2 • Compound patent on prodrug • 2028 WO 2008121634 A 2 • Crystalline forms • 2031 WO 2011123645 A 2 • Combination with ledipasvir • 2032 WO 2013040492 A • Composition & dosage • 2032 WO 2013082003 A 1 •

Patentability criteria: incremental innovation vs. "evergreening" l India: patent applications rejected as known substances are not considered novel under Section 3 d Patent Act (e. g. paediatric formulations, combination patents, polymorphs) unless patent applicant can prove enhanced efficacy Examples: patents refused on: polymorph of imatinib mesylate; suspension of nevirapine hemihydrate; tenofovir disoproxil 7| Public health, innovation and intellectual property

What is a voluntary license? l The owner of a patent can allow others to use the invention, in particular to manufacture, sell, export or import the patented product l Such a mutual agreement is called voluntary license 8| Public health, innovation and intellectual property

What is a voluntary license? l The owner of a patent can allow others to use the invention, in particular to manufacture, sell, export or import the patented product l Such a mutual agreement is called voluntary license l It lays down the conditions, namely – the territory (list of countries in which the licensee may sell the invention) – Royalties – Exclusive or non-exclusive 9| Public health, innovation and intellectual property
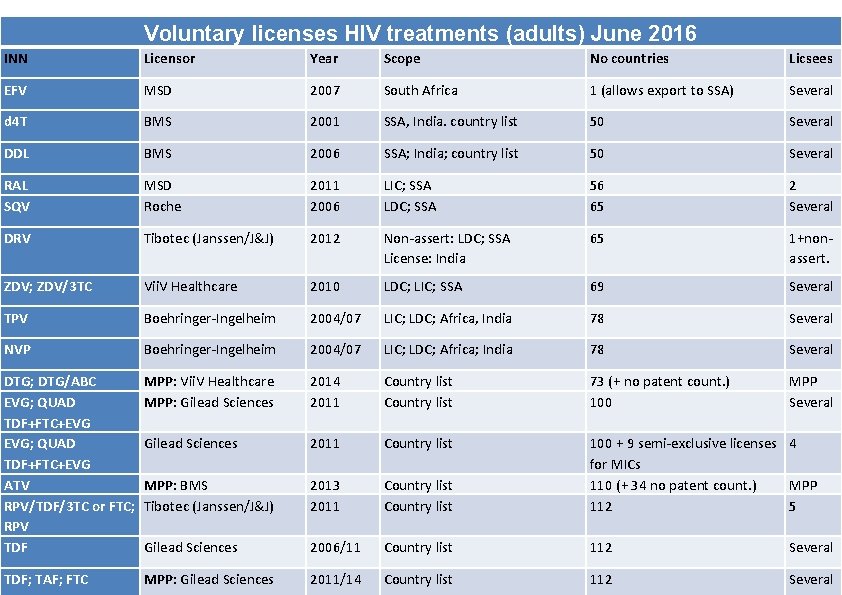
Voluntary licenses HIV treatments (adults) June 2016 INN Licensor Year Scope No countries Licsees EFV MSD 2007 South Africa 1 (allows export to SSA) Several d 4 T BMS 2001 SSA, India. country list 50 Several DDL BMS 2006 SSA; India; country list 50 Several RAL SQV MSD Roche 2011 2006 LIC; SSA LDC; SSA 56 65 2 Several DRV Tibotec (Janssen/J&J) 2012 Non-assert: LDC; SSA License: India 65 1+nonassert. ZDV; ZDV/3 TC Vii. V Healthcare 2010 LDC; LIC; SSA 69 Several TPV Boehringer-Ingelheim 2004/07 LIC; LDC; Africa, India 78 Several NVP Boehringer-Ingelheim 2004/07 LIC; LDC; Africa; India 78 Several DTG; DTG/ABC EVG; QUAD TDF+FTC+EVG ATV RPV/TDF/3 TC or FTC; RPV TDF MPP: Vii. V Healthcare MPP: Gilead Sciences 2014 2011 Country list 73 (+ no patent count. ) 100 MPP Several Gilead Sciences 2011 Country list MPP: BMS Tibotec (Janssen/J&J) 2013 2011 Country list 100 + 9 semi-exclusive licenses 4 for MICs 110 (+ 34 no patent count. ) MPP 112 5 Gilead Sciences 2006/11 Country list 112 Several TDF; TAF; FTC MPP: Gilead Sciences 2011/14 Country list 112 Several

What is a compulsory license? National patent laws mandate national authorities to allow third parties to use a patented invention without the authorization of the patent owner under certain conditions to address certain unwanted consequences – – unfair competition, not working the invention, abusively high prices, health emergencies and others l WTO TRIPS contains certain conditions, but does not limit the grounds 11 | Public health, innovation and intellectual property
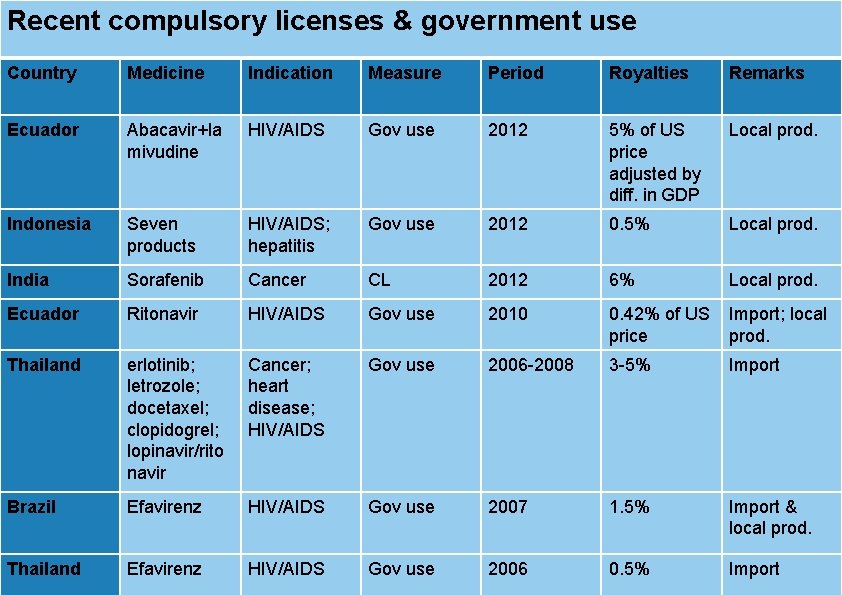
Recent compulsory licenses & government use Country Medicine Indication Measure Period Royalties Remarks Ecuador Abacavir+la mivudine HIV/AIDS Gov use 2012 5% of US price adjusted by diff. in GDP Local prod. Indonesia Seven products HIV/AIDS; hepatitis Gov use 2012 0. 5% Local prod. India Sorafenib Cancer CL 2012 6% Local prod. Ecuador Ritonavir HIV/AIDS Gov use 2010 0. 42% of US price Import; local prod. Thailand erlotinib; letrozole; docetaxel; clopidogrel; lopinavir/rito navir Cancer; heart disease; HIV/AIDS Gov use 2006 -2008 3 -5% Import Brazil Efavirenz HIV/AIDS Gov use 2007 1. 5% Import & local prod. health, innovation and intellectual property 2006 12 | Public Thailand Efavirenz HIV/AIDS Gov use 0. 5% Import
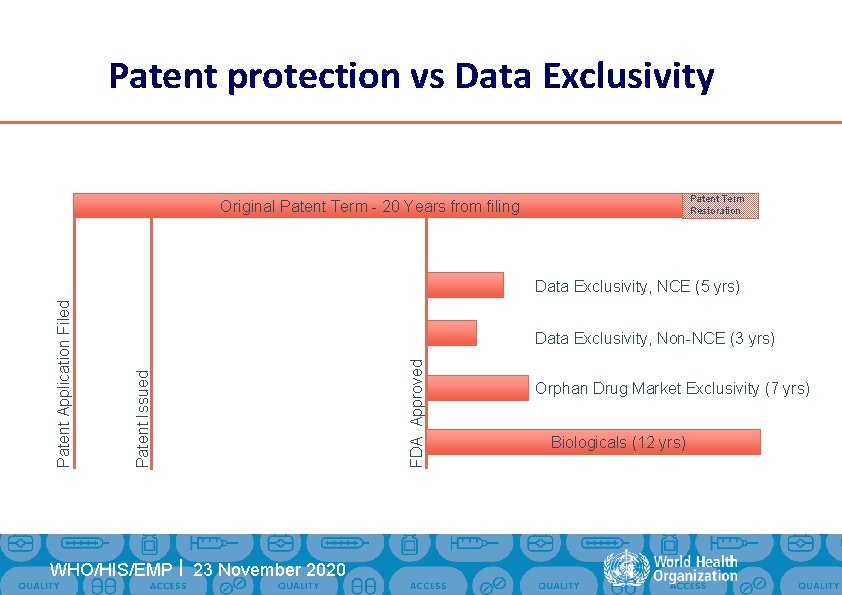
Patent protection vs Data Exclusivity Patent Term Restoration Original Patent Term - 20 Years from filing HIS/EMP | Communications Planning | 23 November WHO/HIS/EMP 2020 FDA Approved Data Exclusivity, Non-NCE (3 yrs) Patent Issued Patent Application Filed Data Exclusivity, NCE (5 yrs) Orphan Drug Market Exclusivity (7 yrs) Biologicals (12 yrs)

WHO Patent Landscapes sofosbuvir (updated) ledipasvir (updated) Daclatasvir (updated) dasabuvir ombitasvir paritaprevir simeprevir Harvoni Viekira Pak with ritonavir Gilead Sciences BMS Abb. Vie Janssen www. who. int/phi/implementation/ip_trade/ip_patent_landscapes/en/ 14 | Public health, innovation and intellectual property

The of intellectual property in local production in developing countries http: //www. who. int/phi/publications/int_pro p_local_prod_opportunities_chanllenge s/en/ www. who. int/phi/publications/category/en/ 15 | Public health, innovation and intellectual property

WHO Global Report on Access to Hepatitis C Treatment http: //www. who. int/phi/publicat ions/category/en/ 16 | Public health, innovation and intellectual property

Promoting Access to Medical Technologies and Innovation www. who. int/phi/promoting_access_me dical_innovation/en/ www. who. int/phi/publications/category/e n/ Dr Peter Beyer Senior Advisor World Health Organization beyerp@who. int Tel. +41 -22 -791 25 07 17 | Public health, innovation and intellectual property
- Slides: 17