Patent Ductus Arteriosus Occlusion Device Oral Presentation 4












- Slides: 16

Patent Ductus Arteriosus Occlusion Device Oral Presentation #4 Group 6 David Brogan, Darci Phillips & Daniel Schultz Advisor: Dr. Thomas Doyle 4/11/2003

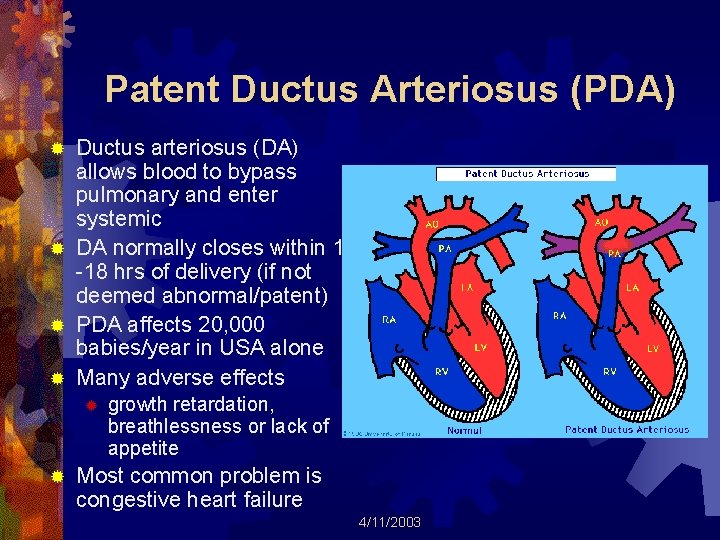
Patent Ductus Arteriosus (PDA) Ductus arteriosus (DA) allows blood to bypass pulmonary and enter systemic ® DA normally closes within 10 -18 hrs of delivery (if not deemed abnormal/patent) ® PDA affects 20, 000 babies/year in USA alone ® Many adverse effects ® ® ® growth retardation, breathlessness or lack of appetite Most common problem is congestive heart failure 4/11/2003

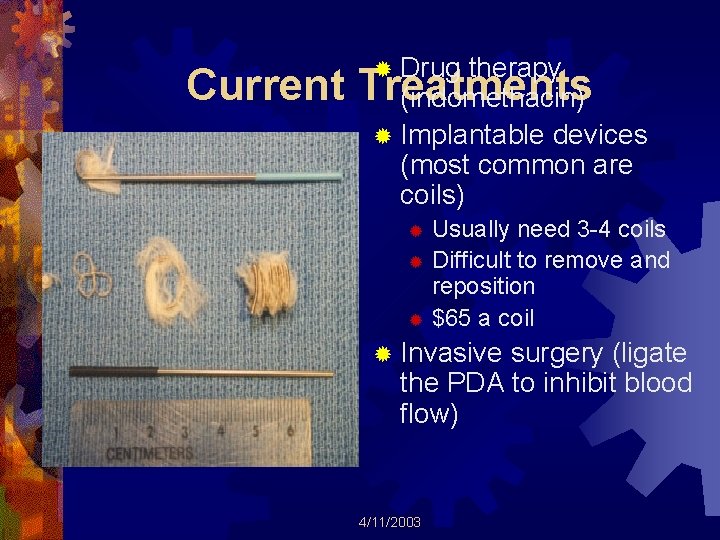
Current ® Drug therapy Treatments (indomethacin) ® Implantable devices (most common are coils) Usually need 3 -4 coils ® Difficult to remove and reposition ® $65 a coil ® ® Invasive surgery (ligate the PDA to inhibit blood flow) 4/11/2003

Current Best Competitor ® Amplatzer Occluder Duct Most effective ® Highest success rate of current devices ® In final stage of FDA approval ® Drawbacks: ® ® Not pliable PDA must conform to shape of ADO $2500 4/11/2003

Project Goals ® To design, develop and patent a PDA occlusion device that can… ® Be delivered via a catheter ® Conform to the shape of the PDA and cause occlusion ® Can be repositioned easily ® Be cost effective (<$200) ® Provide an initial success rate of 100% ® More patient friendly procedure 4/11/2003

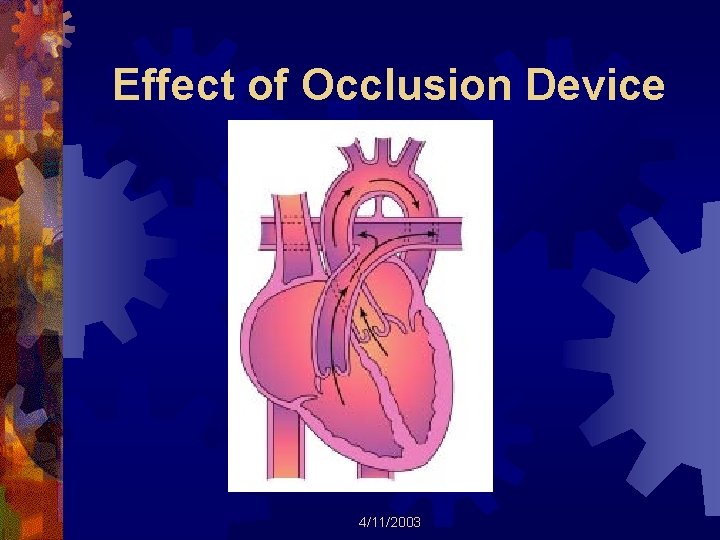
Effect of Occlusion Device 4/11/2003

Our Current Prototype 4/11/2003

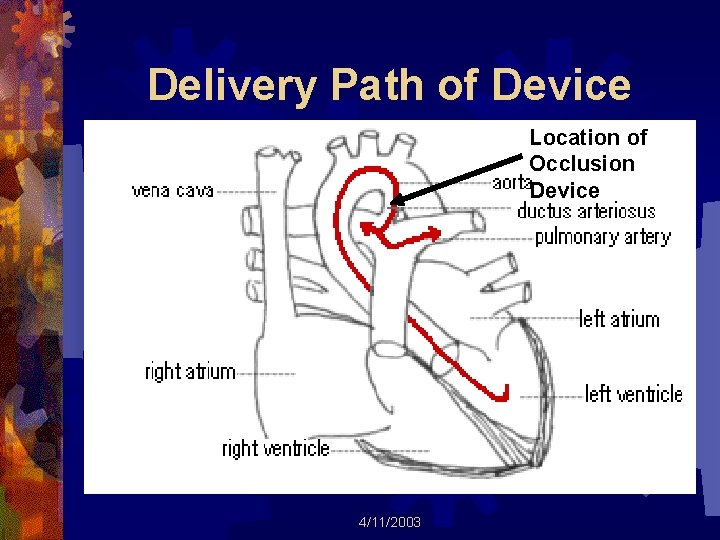
Delivery Path of Device Location of Occlusion Device 4/11/2003

Foam Issues ® We can make a polyurethane foam with methylene bisphenyl diisocyanate (MDI), polytetramethylene glycol (PTMEG), 1, 4 butanediol and water. ® Have ordered MDI and 1, 4 -butanediol, but have not found a vendor yet to supply the PTMEG. ® Contacted Lyonell and are awaiting shipment on the PTMEG. ® Contacted PTG about prototyping and building the polyurethane foam. 4/11/2003

Work Completed ® Conducted extensive research on other treatment methods (to avoid short comings on our design) ® Met with Dr. Doyle to discuss our progress and future goals ® Have placed order foam chemicals (will arrive by Wednesday) ® Have ordered and received Nitinol memory wire in two different diameter thicknesses ® Have secured an In-Vitro PDA Simulation device for testing 4/11/2003

Current Status ® Making final design refinements to device ® Developing life-size PDA device prototype ® Developing equations to model PDA testing apparatus ® Making arrangements with Mechanical Engineering professor to have Nitinol wire machined here at Vanderbilt ® Completing Design Safe and Innovation Work Bench assignments ® Making necessary modifications to website 4/11/2003

In-Vitro Modeling Specs. ® Pressure Drop : 100 mm Hg ® Calculate flow inside PA using Hagen. Poiseuille Eqn. 4 = -ΔP * p*r /(8*μ*L) ® r = 2 -10 mm ®Q ® All variables are known, thus Q can be calculated easily 4/11/2003

Needs Obtain missing foam ingredient (PTMEG) ® Meet with Dr. Doyle to discuss further progress of device past this semester ® Confirm a partnership with PTG to have working/actual prototype manufactured ® 4/11/2003

Future Direction ® Build scaled prototype with correct biomaterials ® Figure device out best way to secure Nitinol within ® Finish conducting pressure and durability tests in PDA simulated environment ® Refine design based on testing 4/11/2003

Recommendations ® Much depending on the outcome of the next two weeks ® If we were to conclude at this exact moment in time, we would recommend… ® Follow-up on patent ® ® ® We have discussed applying for a preliminary patent with Brian Cox and Dr. Doyle Preliminary Patent needs to be filed by April 22, 2003 Continue with PTG arrangements ® Non-Disclosure agreement in the works with PTG Further strength and pressure tests ® Refine design one final time ® 4/11/2003

Contact Information ® David Brogan ® david. m. brogan@vanderbilt. edu ® 1 -210 -364 -4546 ® Darci Phillips ® darci. j. phillips@vanderbilt. edu ® 1 -615 -386 -9015 ® Daniel Schultz ® daniel. j. schultz@vanderbilt. edu ® 1 -615 -421 -6067 4/11/2003