Passive Fire Protection Lec 8 PFP Systems 5


























































- Slides: 61

Passive Fire Protection Lec 8 -PFP Systems 5 -Intumescent coating systems Dr. Zayed Al-Hamamre Chemical Engineering Department | University of Jordan | Amman 11942, Jordan Tel. +962 6 535 5000 | 22888

Introduction Ø Today most buildings and structures have some degree of fire protection in order to o protect lives, o delay possible structural collapse allowing for evacuation, o provide areas of temporary refuge in the case of fire, o ensure the integrity of escape routes by preventing or delaying the escalation of a fire and protect high-value assets. Ø Passive fire protection involves: o components of structural methods and materials such as concrete, mineral fiber boards, o vermiculite cements o intumescent coatings. Chemical Engineering Department | University of Jordan | Amman 11942, Jordan Tel. +962 6 535 5000 | 22888 1 -2

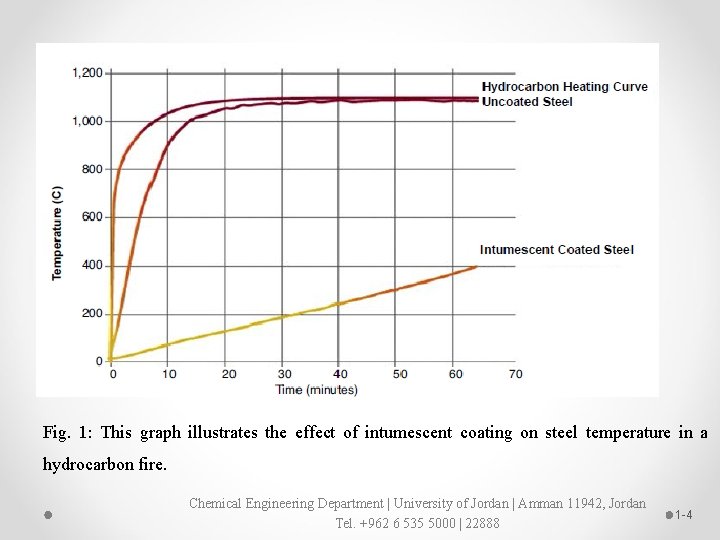
Intumescent coatings Ø Intumescent are often referred to as thin-film or thick-film coatings. Ø Thin-film intumescent can be solvent- or water-based products and have dry film thicknesses (DFTs) of less than 5 millimeters. Ø Thick-film coatings are typically solvent-free, epoxy-based with DFTs of up to 25 mm Ø Intumescent coatings have been used to protect the steelwork in buildings and other structures from fire for longtime ago. Ø These coatings work by swelling up in the event of fire and physically creating a barrier between the steel and the fire for up three hours. Ø Steel loses its structural strength at about 500 o. C and these coatings can delay the time it takes to reach this temperature (Fig. 1). Chemical Engineering Department | University of Jordan | Amman 11942, Jordan Tel. +962 6 535 5000 | 22888 1 -3

Fig. 1: This graph illustrates the effect of intumescent coating on steel temperature in a hydrocarbon fire. Chemical Engineering Department | University of Jordan | Amman 11942, Jordan Tel. +962 6 535 5000 | 22888 1 -4

Ø Thick film epoxies can also be used to form castings, typically in two half shells to protect narrow diameter pipework where spraying would create large volumes of overspray. Ø The acceptance and use of intumescent coatings increased dramatically in Europe in the 1970 s as the major oil companies learned of their ability to protect structural steel from the extreme heat caused by hydrocarbon fires, including jet fires caused by leaking hydrocarbons. Ø However, the intumescent paints of the prior art have suffered from a number of disadvantages, The prior art paints: 1. Have been slow drying paints so that a relatively long drying period was required before any article onto which the paint was applied could be handled. 2. Were also relatively soft and therefore were prone to chip when subjected to rough handling. Chemical Engineering Department | University of Jordan | Amman 11942, Jordan Tel. +962 6 535 5000 | 22888 1 -5

3. Lacked good brushability so that it was difficult to apply these paints to articles without a large expenditure in time and effort. 4. Water permeable and therefore were prone to degrade when exposed to moist environments such as the sea. 5. The char formed by prior art paints were weak and could easily be blown away if the fire had a velocity component or if there was a wind. Process Heat Transfer Chemical Engineering Department | University of Jordan | Amman 11942, Jordan Tel. +962 6 535 5000 | 22888 1 -6

New intumescent paint 1. Dries relatively rapidly. 2. Relatively hard and is capable of withstanding rough handling. 3. Possesses good brushability and which is therefore easily applied. 4. Resists degradation well when subjected to the environment of the sea. 5. Foams quickly and has a relatively long cook-off time. 6. The formed char has consistency and is not easily blown away by a fire which has a velocity component or by a wind. Chemical Engineering Department | University of Jordan | Amman 11942, Jordan Tel. +962 6 535 5000 | 22888 1 -7

Basic ingredients Ø Many intumescent systems have been proposed and it is established that the conventional system contains four basic ingredients i. A base polymer: o A binder such as chlorinated natural rubber, solid vinyl-toluene/ butadiene resin, alkyds, urea-formaldehyde resins, epoxy resins, poly vinylacetate, polyvinylidene chloride and polyvinyl chloride, Glass fiber-reinforced epoxy resins i. Fire retardant materials: 1. A mineral acid catalyst: o The inorganic acid source, carbon source and blowing agent can be selected from a range of materials Chemical Engineering Department | University of Jordan | Amman 11942, Jordan Tel. +962 6 535 5000 | 22888 1 -8

o The inorganic acid (inorganic material yielding a mineral acid) such as sulphuric or phosphoric at a temperature between 200° C. and 250° C. o A preferred example is ammonium polyphosphate. 2. A source of carbon: o The carbon source material is an organic compound containing hydroxyl groups which will react with the acid, and a high proportion of carbon so that when charred during heating a useful amount of carbon is produced. o Examples of such materials are starches, polyhydric alcohols such as erythritol, pentaerythritol, di- and tri-pentaerythritol, and sugars such as glucose, maltose. The preferred material is dipentaerythritol. Chemical Engineering Department | University of Jordan | Amman 11942, Jordan Tel. +962 6 535 5000 | 22888 1 -9

3. A blowing agent o The blowing agent is required to release non-flammable gases at the temperature where the coating has begun to melt and char so that a foam can be formed from the charring mass. o The 2 -amino s-triazine salts will enhance the intumescent properties of the composition, and also its fire resistance. o The most preferred salts have been found to be melamine monooxalate, and dimelamine oxalate. o On the other hand some advantages in intumescent properties and/or fire resistance are to be gained over compositions omitting these salts when using such salts as acetoguanamine oxalate, propioguanamine oxalate, melamine succinate, and melamine maleate. Chemical Engineering Department | University of Jordan | Amman 11942, Jordan Tel. +962 6 535 5000 | 22888 1 -10

Ø In order that the composition's intumescent properties are retained in damp conditions it is preferred that the materials used have low water solubilities to resist their being leached out of the composition. Ø On the other hand if a composition is for use in low humidities only then this requirement is much reduced. Ø Other additive might be also included: o A lubricant and source of chlorine and a material to provide slippage such as a chlorinated paraffin, o A solvent such as a 50: 50 mixture of toluene and xylol, o An anti-settling agent, o A coloring agent such as titanium dioxide or a mixture of yellow oxide and black iron oxide; and o A surfactant. Chemical Engineering Department | University of Jordan | Amman 11942, Jordan Tel. +962 6 535 5000 | 22888 1 -11

The binder Ø The basic function of the coating binder: 1. Is to bind together these intumescent ingredients, and to provide adhesion to the substrate, so that they are held in intimate contact, in order to perform their function when required to do so in a fire situation. 2. Contributes to the formation of a uniform cellular foam structure since the molten binder helps trap the gases given off by the decomposing blowing agents, thus ensuring a controlled expansion of the char. Ø It is important that the ingredients retain their functionality over a long period of time, so the binder also has to protect the often water-sensitive intumescent ingredients by providing the necessary resistance to water, UV light, abrasion, etc. Chemical Engineering Department | University of Jordan | Amman 11942, Jordan Tel. +962 6 535 5000 | 22888 1 -12

Ø In composition the resin binder may be in the form of an unsaturated polyester resin solution in monomer, o The composition also including catalyst and cure promoter for the resin, intumescent additives and optionally other additives such as pigment. o The unsaturated polyester is typically utilized in the composition in the form of a solution of the polymer in a monomer such as styrene, the amount of monomer preferably being in the range 20 to 60 wt %. o By unsaturated polyester we mean a polyester of the kind which contains carbon unsaturation along its backbone chain. o The unsaturated polyester is preferably plasticized to enhance its capability as binder in the intumescent system. o As plasticizer an unsaturated polyester resin may be used, this also being a solution in styrene monomer, of similar styrene content to the other polyester Chemical Engineering Department | University of Jordan | Amman 11942, Jordan Tel. +962 6 535 5000 | 22888 1 -13

o Alternatively the plasticizer may be a known plasticizing compound such as an alkyl phosphate, a halogenated paraffin, and ester type plasticizers such as di-octyl phthalate and di-octyl maleate. o The preferred plasticizers contain either phosphorus or halogen or both, these compounds being fire-retardant plasticizers which we have now found to improve the efficiency of the intumescent system. o Halogen containing polyesters are particularly preferred when the plasticizer contains no combined halogen or phosphorus, although polyesters containing no halogen can be used successfully with halogen or phosphorus containing plasticizers. Ø The particularly preferred plasticizers are tris 2, 3 -dibromo propyl phosphate, tris monochloro ethyl phosphate and tri-ethyl phosphate Chemical Engineering Department | University of Jordan | Amman 11942, Jordan Tel. +962 6 535 5000 | 22888 1 -14

Ø The unsaturated polyester resin preferably is dissolved in styrene monomer to facilitate cross-linking of the resulting coating, the amount of styrene in the coating composition being dependent on the particular resin being used, but preferably in the range 20 to 60 percent by weight based on the total weight of solution. Butyl rubber Ø An ambient curing butyl rubber is prepared from a composition comprising a liquid copolymer of isobutylene and isoprene, an ambient curing agent such as p-quinone dioxime and lead oxide and a synergistic combination of hexabromobenzene, antimony oxide and a chlorinated paraffin as a flame retardant which renders the composition self-extinguishing without affecting the physical properties of the rubber. Ø The composition is useful as a sprayable protective coating over urethane foam roofs. Chemical Engineering Department | University of Jordan | Amman 11942, Jordan Tel. +962 6 535 5000 | 22888 1 -15

Ø The outstanding weatherability, flexibility and water resistance of butyl rubber makes it useful as a roofing material where it can be formulated to serve as a primary seal or as a top coat for existing roofs. Its low water vapor and gas transmission rates make it especially useful for foam roofs or in insulated freezer buildings where food is stored in inert gas atmosphere. Ø Asphalt coatings are approximately 25 times more permeable to' moisture than butyl coatings. Ø However, the strength of the cured prepolymers is lower than that of a conventional butyl rubber coatings and theflammability characteristics are not acceptable. Ø When the prepolymer composition is compounded with typical flame retardant agents such as antimony trioxide and organic chloride, the retardantsmust be present in a high level that the physical properties of the final-elastomer are seriously degraded. Chemical Engineering Department | University of Jordan | Amman 11942, Jordan Tel. +962 6 535 5000 | 22888 1 -16

Ø A flame retardant, low permeability roofing coating is especially needed‘ when utilized to cover urethane foam roofs since if water can permeate into the foam layer, at sub-freezing temperatures the water will freeze, expand rupture the foam which will deteriorate and eventually lead to rupture of the surface coating. Chemical Engineering Department | University of Jordan | Amman 11942, Jordan Tel. +962 6 535 5000 | 22888 1 -17

Chlorinated parafiin Ø The chlorinated parafiin is merely a mixture of paraffins which have been chlorinated. o The chlorine content of the paraffin should be in the neighborhood of about 70% and cannot vary greatly from this percentage. o This material provides slippage to the paint composition and also acts as a source of chlorine. o It is theorized that this material either gives of chlorine gas directly or does so thru a series of steps. o This gas acts as a fire retardant by pushing the flame away from the article which the instant paint coats. o This gives the binder a better chance to form a char which will act as the fire retardant material once the liberation of the gas has ceased. Chemical Engineering Department | University of Jordan | Amman 11942, Jordan Tel. +962 6 535 5000 | 22888 1 -18

The Solvent Ø The purpose of the solvent is to aid in mixing the other components so that a uniminutes. Ø The solvent will evaporate when the paint is applied and dried. Ø The dry paint will contain all the other ingredients hereinbefore and hereinafter listed except for the solvent which should nearly completely evaporate. Ø A preferred solvent for the paint of this invention is 50 -50 mixture of toluene and xylol. Ø Toluene and xylol are the preferred solvents because they dry very rapidly, Examples of other possible solvents which can be used in the composition of this invention are mineral spirits, MEK and high flash naphtha. Chemical Engineering Department | University of Jordan | Amman 11942, Jordan Tel. +962 6 535 5000 | 22888 1 -19

Ø An anti-settling agent is also necessary in the paint composition. o Any of the well known anti-settling agents used in the paint arts can be used with the instant paint composition since the actual composition of the anti-settling agent is not a critical feature. Ø A coloring agent is also added to give the desired color and to impart body to the paint. o If a white paint is desired Ti can be added. o On the other hand if an olive drab color is desired a mixture of yellow oxide and black iron oxide is added. Ø A surfactant is also added to the hereinbefore described fire retardant intumescent paint composition. o Any of the well known surfactants, such as lecithin, used in the paint arts can be used in the paint composition since the nature of this ingredient is also not critical. Process Heat Transfer Chemical Engineering Department | University of Jordan | Amman 11942, Jordan Tel. +962 6 535 5000 | 22888 1 -20

Ø The composition of paint must have a series of components which will react to liberate Water. Ø These materials include (a) melamine (b) dipentaerythritol or tripentaerythritol and (c) ammonium polyphosphate or tris (2, 3 -dibromopropyl) phosphate. Ø It is important that the ammonium polyphosphate have a sufficient phosphate content (28. 5 -33%) Ø The ammonium polyphosphate or tris (2, 3 -dibromopropyl) phosphate acts as a source of phosphorus when the paint is exposed to fire and the phosphorus thereupon reacts with the alcohol with melamine acting as a catalyst. Ø The product which is formed breaks down at a lower temperature than does the original products and liberates water. Chemical Engineering Department | University of Jordan | Amman 11942, Jordan Tel. +962 6 535 5000 | 22888 1 -21

Ø Because there at least three different chemical reactions taking place (formation of char, chlorine and water) and each is needed if the paint is to be fire retardant it is very important that the various constituents of the paint be present in the proper proportions. Ø If improper proportions are used any one of the reactions which must take place to yield the desired fire retardant properties may not take place. Ø Thus, for example, if insufiicient binder is present the char that does form will be chalky and will tend to blow away. In view of this a very delicate balance exists between the various substituents and there is a relatively narrow variation in the quantity of each material which can be used to form a paint which is fire retardant and intumescent. Chemical Engineering Department | University of Jordan | Amman 11942, Jordan Tel. +962 6 535 5000 | 22888 1 -22

Ø In the compositions the ratio of resin binder to total intumescent additives is preferably in the range (by weight) of 75: 25 to 40: 60. Ø Similarly, amongst the intumescent additives the ratio (by weight) of phosphate to total carbon source + blowing agent is preferably in the range 85: 15 to 30: 70, Ø The ratio of polyhydric compound to blowing agent (by weight) is preferably in the range 90: 10 to 10: 90. Chemical Engineering Department | University of Jordan | Amman 11942, Jordan Tel. +962 6 535 5000 | 22888 1 -23

Ø A fire retardant intumescent paint comprising: (a) 6. 457. 50% of a binder selected from the group consisting of chlorinated natural rubber with a chlorine content of about 67%, solid yinyl-toluene/butadiene resin and mixtures thereof; (b) 14 -15% melamine; (c) 99. 5% of an alcohol selected from the group consisting of dipentaerythritol and tripentaerythritol; (d) 28. 5 -33% of a phosphorus containing material selected from the group consisting of ammonium polyphosphate with a phosphate content of about and tris(2, 3 -dibromopropyl) phosphate; (e) 1. 3 -3. 5 of a chlorinated paraflin with a chlorine content of about 70%; (f) at least 25% of a solvent; (g) 02 -06% of an anti-settling agent; (h) 0. 25 -0. 75 of a surfactant; and (i) 4. 46. 5%. of coloring agents provided that when said paint is white said binder exceeds 6. 70%. Chemical Engineering Department | University of Jordan | Amman 11942, Jordan Tel. +962 6 535 5000 | 22888 1 -24

How Do Intumescent Coatings Work? Ø Intumescent coatings react to fire by o Expanding to form a carbon “char” with low thermal conductivity, which essentially forms an insulating layer reducing the rate of heat transfer and extending the time necessary to reach the critical failure temperature of the underlying steel. Ø When an intumescent coating is subjected to heat, a series of chemical reactions occurs: o The ammonium polyphosphate decomposes to produce phosphoric acid; o the phosphoric acid causes dehydration of the pentaerythritol or dipentaerythritol to produce a thick carbon char. o The char provide a higher level of insulation than the coating system in its unreacted state Chemical Engineering Department | University of Jordan | Amman 11942, Jordan Tel. +962 6 535 5000 | 22888 1 -25

o The blowing agent decomposes, releasing non-flammable gases that cause the carbon char to foam, thus producing a meringue-like structure that is a highly effective insulator against heat. Ø These are the basic reactions taking place, although more complex interactions also occur. For example, filler particles are incorporated into the formulation to act as nucleating sites or “bubble growth” sites and the resin binder plays a large part in softening and charring. Reinforcing mesh can be used to support the formed char. Chemical Engineering Department | University of Jordan | Amman 11942, Jordan Tel. +962 6 535 5000 | 22888 1 -26

v Reactions during intumescence Chemical Engineering Department | University of Jordan | Amman 11942, Jordan Tel. +962 6 535 5000 | 22888 1 -27

Chemical Engineering Department | University of Jordan | Amman 11942, Jordan Tel. +962 6 535 5000 | 22888 1 -28

Intumescent for wood substrate Ø It is difficult to impart durable flame retardance to a wood substrate. Ø Wood substrates present to flame-proofing problem which is materially different from that presented by fibrous hydrophilic organic substrates Ø In the fibrous hydrophilic organic substrates substantially independent fibers are tangled together, leaving the interstices capable of being filled by an aqueous medium by capillary action between all of their surfaces. Ø The individual fibers contain a relatively small amount of cellulose, and the materials composed of them have a relatively low ignition temperature. Ø In wood substrates, however, the cellulosic fibers are bonded together to form a relatively impenetrable block susceptible to little capillary action. Chemical Engineering Department | University of Jordan | Amman 11942, Jordan Tel. +962 6 535 5000 | 22888 1 -29

Ø A piece of wood has small surface area in relation to the amount of surface area it contains and has a relatively high ignition temperature. Ø Because of these factors, a flame proofing agent which flame proofs fibrous hydrophilic organic materials is not likely to flameproof wood because its capacity to inhibit burning is likely to the destroyed by the time it is heated to the ignition temperature of the wood and/or because of the difficulty of causing a non-volatile substance to penetrate into the volume of a block of wood Ø the use of a wood substrate impregnated with a fire-retardant salt is restricted to low humidity applications due to the water solubility and hygroscopicity of most known fire-retardant salts. Ø Thus, if an ammonium phosphate-impregnated wood substrate is exposed to high (greater than 90 percent) humidity at ambient temperature, in approximately three days the fire retardant-impregnant (salt) will leach therefrom Chemical Engineering Department | University of Jordan | Amman 11942, Jordan Tel. +962 6 535 5000 | 22888 1 -30

Ø The salt will absorb sufficient water vapor to enable it to migrate to the wood substrate surface. Ø Not only does this leaching deplete the salt content of the wood substrate, rendering it less fire resistant, but it also severely disfigures the wood substrate's surface. Ø Many of the flame retardants which are used to impregnate wood substrates are acidic and hydroscopic. Ø The use of these compositions dehydrates the wood and that the wood, in order to make up for the dehydration caused by these hygroscopic agents, must absorb water from the moisture in the ambient air. Chemical Engineering Department | University of Jordan | Amman 11942, Jordan Tel. +962 6 535 5000 | 22888 1 -31

Ø Such moisture absorption will tend to o discolor the plywood and rust and stain connecting members such as nails; o dilutes the amount of agent used in the wood o causes the wood to be damp and moist thus not only corroding the wood but subjecting it to attack from insects and the like Ø An intumescent composition of a wood substrate contains o A cyclic nitrogen compound (selected from group consisting of hydrogen and -CH 3) o A copolycondensed vinylphosphonate, o An acid (phosphorus acid, phosphoric acid), and o Water Chemical Engineering Department | University of Jordan | Amman 11942, Jordan Tel. +962 6 535 5000 | 22888 1 -32

Ø It represents a unique intumescent composition which can impart durable flame retardance to wood substrates even after repeated exposure to water, which will form a coating film which can withstand repeated scrubbing or washing, and which will substantially decrease the amount of noxious fumes generated during pyrolysis of the treated wood substrate. Chemical Engineering Department | University of Jordan | Amman 11942, Jordan Tel. +962 6 535 5000 | 22888 1 -33

Applications Ø Designed for protecting electric power cables, communication cables, junction boxes - against ignition, propagation of fire, smoke emission o Expands over 65 times by volume for heat insulation, o Prevention of coated area ignition, o Sealing against fire spread, o Flame resistance & flame propagation retardance. Chemical Engineering Department | University of Jordan | Amman 11942, Jordan Tel. +962 6 535 5000 | 22888 1 -34

Applications Ø Passive fire suppression cable coatings preventing fire propagation & smoke generation 1 -35 Chemical Engineering Department | University of Jordan | Amman 11942, Jordan Tel. +962 6 535 5000 | 22888

Ø The passage of all building services like cables, electrical wiring and telephone cables etc. , shall be protected by enclosures in the form of Duct / Shafts with a fire resistance of not less than 2 hours. Chemical Engineering Department | University of Jordan | Amman 11942, Jordan Tel. +962 6 535 5000 | 22888 1 -36

Ø For the fire proofing of combustible cables at fire stops in walls, floors, below floor mounted panels, switchgears, junctions /crossings of cable routes. . . Ø Typically applied as a continuous coating on power & communication cables Ø Can also be applied intermittently to serve as a "Fire Break" within a cable run both interior & exterior applications (either top coat or use weather resistant - grade) Ø For both vertical & horizontal cable runs on both single & grouped electrical & communication cabling, Ø Within cables trays as well as outside of cable trays. Chemical Engineering Department | University of Jordan | Amman 11942, Jordan Tel. +962 6 535 5000 | 22888 1 -37

Chemical Engineering Department | University of Jordan | Amman 11942, Jordan Tel. +962 6 535 5000 | 22888 1 -38

Specific features Ø In the event of fire expands as a result of heat exposure and cures to an insulating char. Ø Flame spread on cables and insulation damage are delayed in the case of prolonged exposure to fire or heat. Ø Current rating of the coated electrical cables is not reduced. Ø Escape routes and power supply can be used for a longer time. Ø Significant extension of operative condition of cables. Ø Dripping or dropping of burning cable fragments is limited in amount and time depending on kind and intensity of the fire. Ø Protects Cables sheaths against heat ageing with larger area coverage. Ø Other characteristics include: smoke generated from the protected materials is < 400% minimized, free of asbestos, halogens. . . Chemical Engineering Department | University of Jordan | Amman 11942, Jordan Tel. +962 6 535 5000 | 22888 1 -39

Structural steel intumescent coatings Ø Thin Film High Solids Intumescent Coating provide maximum protection o preventing steel structures from crossing the critical temperature of 550 C, thus o delaying the col apse of loaded steel structures for up to 2 hours and o providing time for evacuation & fire fighting. Ø It can enables a high quality finish to be obtained similar to conventional decorative paints Ø Smooth fiber free robust decorative surface allows the architect to use the structural steel work as an integral part of the building design. Ø Easy cleaning & maintenance. Ø Rapid installation Ø Appearance of the structure is preserved without affecting the load bearing calculations. Chemical Engineering Department | University of Jordan | Amman 11942, Jordan Tel. +962 6 535 5000 | 22888 1 -40

Ø Solvent - based primer for fire protection coating for structural steel to protect steel against corrosion & prepare the surface. o Self extinguishing. o Fast drying. o Based on synthetic resins. Ø The drying time depends on temperature and humidity. Ø Applied to a measured thickness to provide the specified level of protection Ø Reacts under the influence of heat by swelling to many times of its original thickness and producing a layer of carbonaceous foam that acts as an insulating layer to delay the steel from reaching its critical temperature. Chemical Engineering Department | University of Jordan | Amman 11942, Jordan Tel. +962 6 535 5000 | 22888 1 -41

Ø Passive fire protection coatings for structural steel - preventing weakening & collapse. Chemical Engineering Department | University of Jordan | Amman 11942, Jordan Tel. +962 6 535 5000 | 22888 1 -42

Chemical Engineering Department | University of Jordan | Amman 11942, Jordan Tel. +962 6 535 5000 | 22888 1 -43

Chemical Engineering Department | University of Jordan | Amman 11942, Jordan Tel. +962 6 535 5000 | 22888 1 -44

Specification principles Ø The performance of a specific intumescent depends on coating thicknesses, the types of steel section, hollow sections and the section orientation, i. e. , beam or column. Ø Specifications of thickness of intumescent vary depending on the substrate and steel profile Ø Factors influencing the specification: o Type of section (open/ closed/cellular profile) o Massivity of the steel (Hp/A) o Exposure (i. e. 3 sided, 4 sided exposure) o Fire Rating FR (30/ 60/ 90/ 120 min) o Critical temperature (CT) Chemical Engineering Department | University of Jordan | Amman 11942, Jordan Tel. +962 6 535 5000 | 22888 1 -45

Chemical Engineering Department | University of Jordan | Amman 11942, Jordan Tel. +962 6 535 5000 | 22888 1 -46

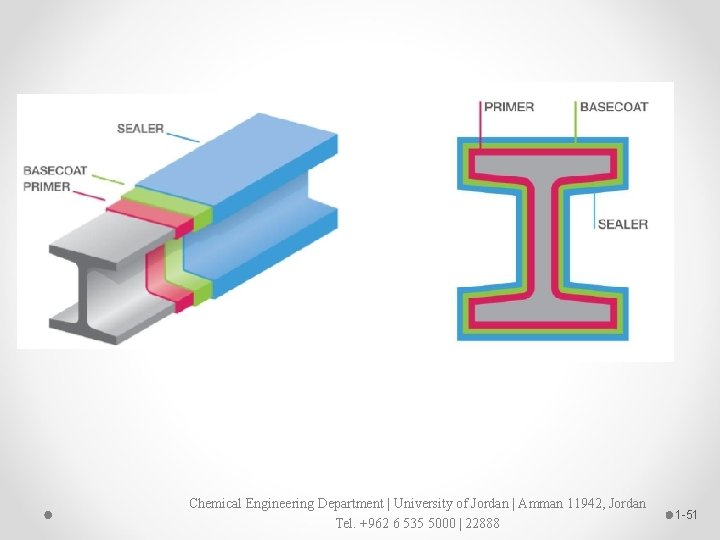
Fire protection coating system Chemical Engineering Department | University of Jordan | Amman 11942, Jordan Tel. +962 6 535 5000 | 22888 1 -47

Chemical Engineering Department | University of Jordan | Amman 11942, Jordan Tel. +962 6 535 5000 | 22888 1 -48

Chemical Engineering Department | University of Jordan | Amman 11942, Jordan Tel. +962 6 535 5000 | 22888 1 -49

Chemical Engineering Department | University of Jordan | Amman 11942, Jordan Tel. +962 6 535 5000 | 22888 1 -50

Chemical Engineering Department | University of Jordan | Amman 11942, Jordan Tel. +962 6 535 5000 | 22888 1 -51

Chemical Engineering Department | University of Jordan | Amman 11942, Jordan Tel. +962 6 535 5000 | 22888 1 -52

Chemical Engineering Department | University of Jordan | Amman 11942, Jordan Tel. +962 6 535 5000 | 22888 1 -53

Factors for consideration when specifying intumescent coating system Chemical Engineering Department | University of Jordan | Amman 11942, Jordan Tel. +962 6 535 5000 | 22888 1 -54

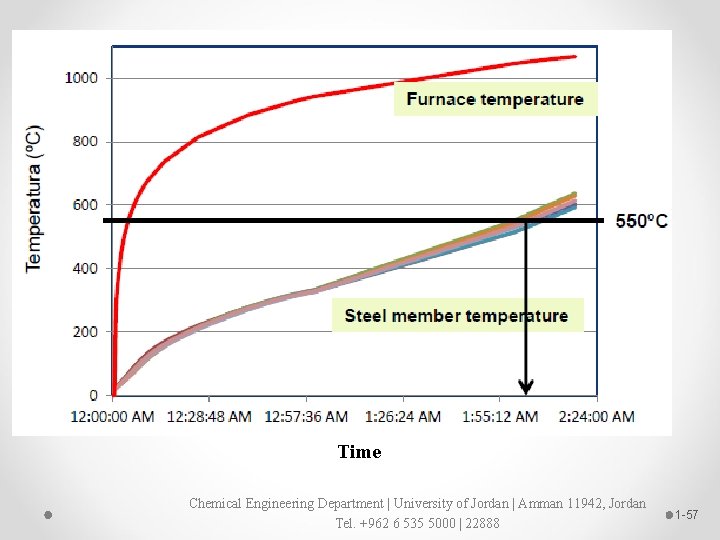
Test methods Ø Fire resistance level (FRL) is the nominal grading period in minutes that is determined by subjecting a specimen to the standard time temperature curve regime to specify: o Structure adequacy o Integrity o Insulation Ø The minimum FRL’s requirements for fire protection vary depending on (1) the building classification, (2) fire compartment size and (3) type of construction (rise in storeys) Ø These three variables contribute and therefore reflect fire risk in a building, determine the type of construction required (A, B or C). Chemical Engineering Department | University of Jordan | Amman 11942, Jordan Tel. +962 6 535 5000 | 22888 1 -55

Ø “Test methods for determining the contribution to the fire resistance of structural members” describe how intumescent coatings are tested with cellulosic fire exposure. Ø Thermocouples are used to measure furnace temperature and core steel temperature. Other test standards include restoration afterwards. Ø Poor durability can also lead to corrosion of the substrate, compromising structural integrity. Ø To ensure durability of intumescent coatings the key ingredients — ammonium polyphosphate, melamine and pentaerythritol — are all sensitive to moisture and must be formulated carefully. Chemical Engineering Department | University of Jordan | Amman 11942, Jordan Tel. +962 6 535 5000 | 22888 1 -56

Time Chemical Engineering Department | University of Jordan | Amman 11942, Jordan Tel. +962 6 535 5000 | 22888 1 -57

New R&D Laboratory for Fire Protection Chemical Engineering Department | University of Jordan | Amman 11942, Jordan Tel. +962 6 535 5000 | 22888 1 -58

Internal testing External testing Chemical Engineering Department | University of Jordan | Amman 11942, Jordan Tel. +962 6 535 5000 | 22888 1 -59

Intumescent coating system exposed to radiant heat Chemical Engineering Department | University of Jordan | Amman 11942, Jordan Tel. +962 6 535 5000 | 22888 1 -60

Chemical Engineering Department | University of Jordan | Amman 11942, Jordan Tel. +962 6 535 5000 | 22888 1 -61