PASSIVATION TREATMENTS FOR COPPERNICKEL ALLOYS EXPOSED TO SEAWATER









- Slides: 13

PASSIVATION TREATMENTS FOR COPPER-NICKEL ALLOYS EXPOSED TO SEAWATER Jacob Steiner, Ph. D, jacob. r. steiner@navy. mil, Naval Surface Warfare Center, Carderock Division Elissa Trueman, Ph. D, PE, Naval Surface Warfare Center, Carderock Division Distribution A. Approved for public release. Distribution is unlimited. 1

Overview • Motivation • Basics of passive films • Methodology: film modification – Adsorbed films – Modified outer layer films • • • Experimental details Results Summary Future work Acknowledgements Distribution A. Approved for public release. Distribution is unlimited. 2

Motivation • Current practice requires 21 days in clean seawater for passivation – Clean has a specific meaning including certain biologic oxygen demand, total suspended solids and sulfide concentration amounts • Difficulty due to “dirty” waters in port environments – Port waters often contains sand/silt/debris, biologic organisms – Port waters can be highly variable due to agricultural run off and other pollutants • Length of time may have impact on ships schedules Key Parameters • Decrease number of days required for passivation to 7 days • Examine ability to discharge overboard • Provide a color change to identify passive film (ideal but not essential) Distribution A. Approved for public release. Distribution is unlimited. 3

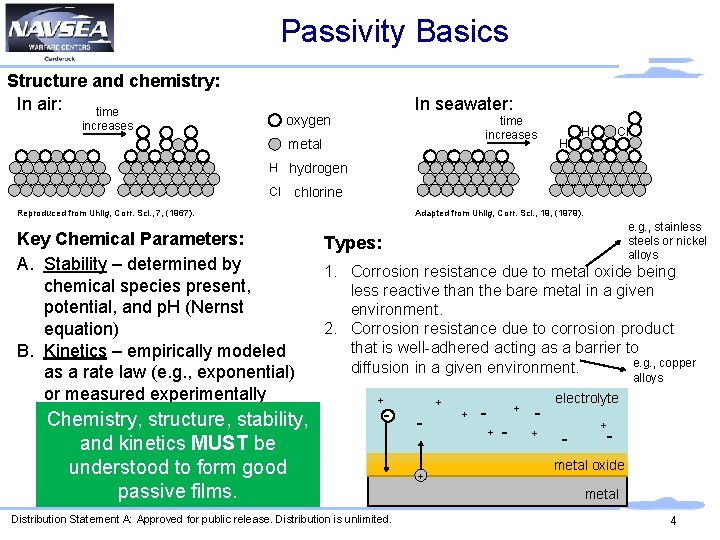
Passivity Basics Structure and chemistry: In air: time In seawater: oxygen increases time increases metal H Cl H H hydrogen Cl chlorine Adapted from Uhlig, Corr. Sci. , 19, (1979). Reproduced from Uhlig, Corr. Sci. , 7, (1967). Key Chemical Parameters: A. Stability – determined by chemical species present, potential, and p. H (Nernst equation) B. Kinetics – empirically modeled as a rate law (e. g. , exponential) or measured experimentally Chemistry, structure, stability, and kinetics MUST be understood to form good passive films. e. g. , stainless steels or nickel alloys Types: 1. Corrosion resistance due to metal oxide being less reactive than the bare metal in a given environment. 2. Corrosion resistance due to corrosion product that is well-adhered acting as a barrier to e. g. , copper diffusion in a given environment. alloys + - Distribution Statement A: Approved for public release. Distribution is unlimited. + + + - + electrolyte - + - metal oxide metal 4

Methodology Adsorbed Films • • • Organic molecule based compounds with electronegative atoms (O, N, S) Possess hydrophilic functional groups or long alkyl chains to hinder diffusion of corrosive species Also commonly referred to as an inhibitor approach Modified Surface Layers • • • Inorganic salt based compounds with substitutive metal cations like Fe or Ni Metal cations replace Cu atoms to form mixed oxide films Mixed oxides can confer Type I passivity or improve Type II passivity of native Cu 2 O Ni. O Cu 2 O Cu Adapted from Chen et al. , Applied Surface Science 456, 2018 Distribution A. Approved for public release. Distribution is unlimited. 5

Experimental Details • 3/8” diameter x ~1/8” thick annealed bar • Five chemistries in ASTM SW – UNS C 70600, 90/10 Cu. Ni – UNS C 71500, 70/30 Cu. Ni – 600 grit silicon carbide, rinsed in ethanol, rinsed in deionized water, air dried • Exposed for one week – 5 m. M citric acid – 0. 5 m. M nickel hydroxide – 1 m. M potassium iodide + 5 m. M L -leucine – 12. 5 m. M nickel sulfate – 1 m. M propargyl alcohol – Quiescent – Rotating (on orbital shaker) at 100 • Characterization – Optical examination RPM – Scanning electron microscope – 40 m. L of solution • Control chemistry 21 days in ASTM D 1141 artificial seawater (ASTM SW) examination – Electrochemical impedance spectroscopy – Copper ion measurements – 7 days in ASTM D 1141 ASW completed for reference Distribution A. Approved for public release. Distribution is unlimited. 6

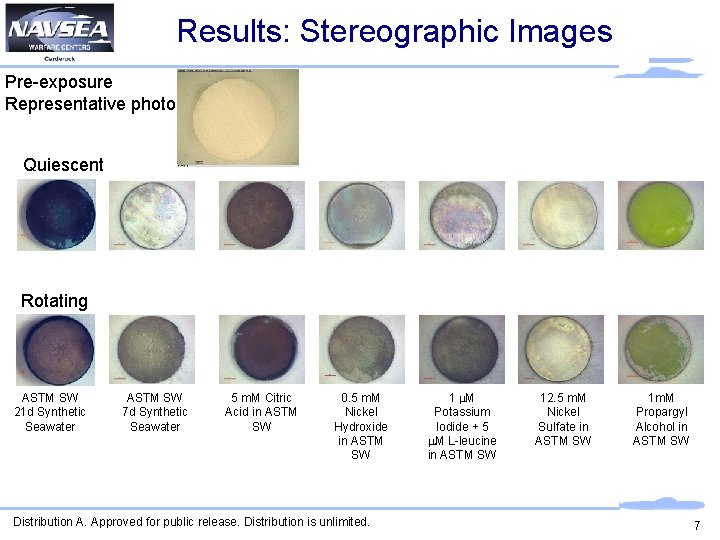
Results: Stereographic Images Pre-exposure Representative photo Quiescent Rotating ASTM SW 21 d Synthetic Seawater ASTM SW 7 d Synthetic Seawater 5 m. M Citric Acid in ASTM SW 0. 5 m. M Nickel Hydroxide in ASTM SW Distribution A. Approved for public release. Distribution is unlimited. 1 m. M Potassium Iodide + 5 m. M L-leucine in ASTM SW 12. 5 m. M Nickel Sulfate in ASTM SW 1 m. M Propargyl Alcohol in ASTM SW 7

Results: SEM Images Quiescent Rotating ASTM SW 7 d Synthetic Seawater 5 m. M Citric Acid in ASTM SW 0. 5 m. M Nickel Hydroxide in ASTM SW Distribution A. Approved for public release. Distribution is unlimited. 1 m. M Potassiu m Iodide + 5 m. M Lleucine in ASTM SW 12. 5 m. M Nickel Sulfate in ASTM SW 1 m. M Propargyl Alcohol in ASTM SW 8

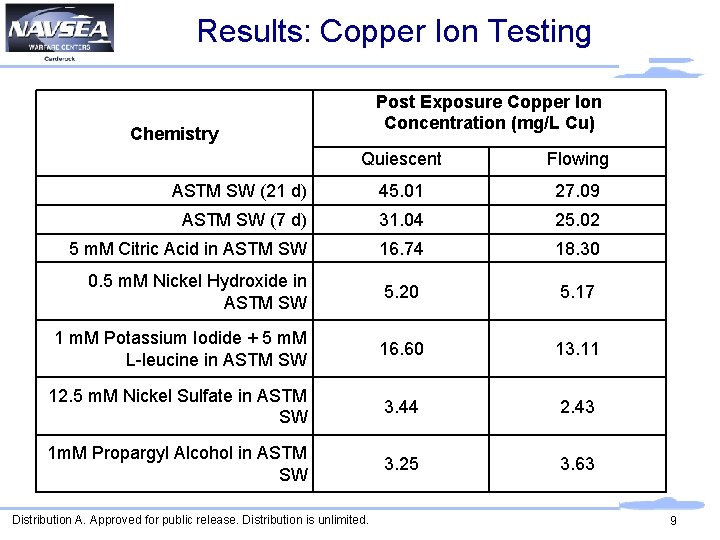
Results: Copper Ion Testing Post Exposure Copper Ion Concentration (mg/L Cu) Chemistry Quiescent Flowing ASTM SW (21 d) 45. 01 27. 09 ASTM SW (7 d) 31. 04 25. 02 5 m. M Citric Acid in ASTM SW 16. 74 18. 30 0. 5 m. M Nickel Hydroxide in ASTM SW 5. 20 5. 17 1 m. M Potassium Iodide + 5 m. M L-leucine in ASTM SW 16. 60 13. 11 12. 5 m. M Nickel Sulfate in ASTM SW 3. 44 2. 43 1 m. M Propargyl Alcohol in ASTM SW 3. 25 3. 63 Distribution A. Approved for public release. Distribution is unlimited. 9

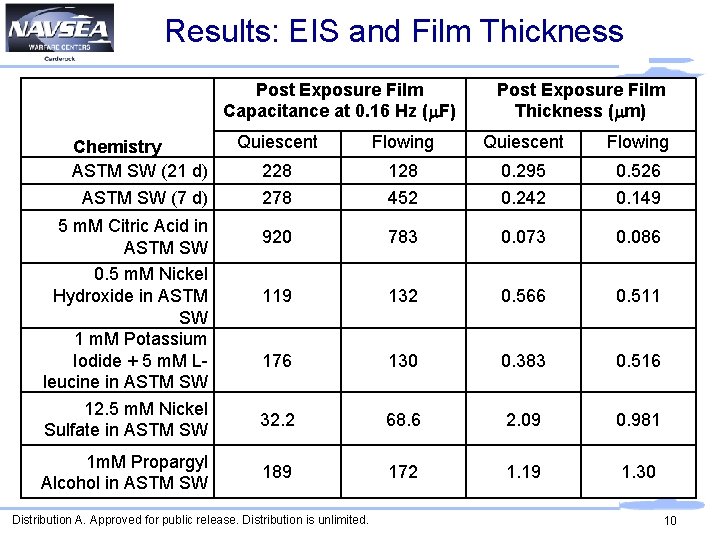
Results: EIS and Film Thickness Post Exposure Film Capacitance at 0. 16 Hz (m. F) Chemistry ASTM SW (21 d) ASTM SW (7 d) 5 m. M Citric Acid in ASTM SW 0. 5 m. M Nickel Hydroxide in ASTM SW 1 m. M Potassium Iodide + 5 m. M Lleucine in ASTM SW 12. 5 m. M Nickel Sulfate in ASTM SW 1 m. M Propargyl Alcohol in ASTM SW Post Exposure Film Thickness (mm) Quiescent Flowing 228 278 128 452 0. 295 0. 242 0. 526 0. 149 920 783 0. 073 0. 086 119 132 0. 566 0. 511 176 130 0. 383 0. 516 32. 2 68. 6 2. 09 0. 981 189 172 1. 19 1. 30 Distribution A. Approved for public release. Distribution is unlimited. 10

Summary • Surface films formed within seven days when exposed to the various chemistries show equivalent or improved performance when measured by copper ion concentration after exposure and film thickness – except for films formed when exposed to citric acid. • The lowest copper ion concentration and thickest films were formed in solutions with nickel sulfate and propargyl alcohol – The propargyl alcohol also provides a distinct color change from the alloy, making application easy to identify • However, propargyl alcohol is not environmentally friendly. Of the environmentally friendly (able to be discharged overboard) chemical additives examined, only the potassium iodide plus L-leucine performed adequately Distribution A. Approved for public release. Distribution is unlimited. 11

Future Work • Further work is needed to down select the chemistries that performed well when compared to 21 day exposures in ASTM SW – Compatibility with 70/30 Cu. Ni (UNS C 71500) – Investigation into barrier properties of the films formed using EIS – Films composition will be accomplished via x-ray photoelectron spectroscopy and other surface characterization techniques in order to verify the protection mechanisms (modified outer layer film or molecular barrier) – The final technologies selected will need to take into account the durability of the films in flowing seawater and resistance to sulfide poisoning Distribution A. Approved for public release. Distribution is unlimited. 12

Acknowledgements THANKS to: • The Cross Platform System Development Program (Naval Sea Systems Command, NAVSEA, SEA 05 T), Ms. Danesha Gross for funding • Ms. Erin Miller, Mr. Tony Nguyen, and Mr. Tom Armstrong, NAVSEA 05 Z 25, for technical discussion. Disclaimer: Any opinions, findings, conclusions or recommendations expressed in this document are those of the authors and do not necessarily reflect the views of the NAVSEA or Naval Surface Warfare Center, Carderock Division. This document has been approved as Distribution A: Approved for public release; distribution unlimited. Distribution A. Approved for public release. Distribution is unlimited. 13