Passageway Food Follows Mouth Structure Chemical BreakdownProcess Gland
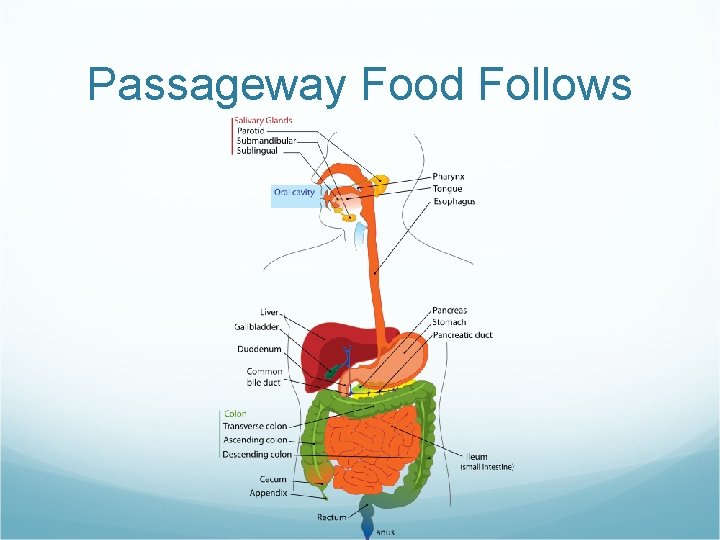
Passageway Food Follows
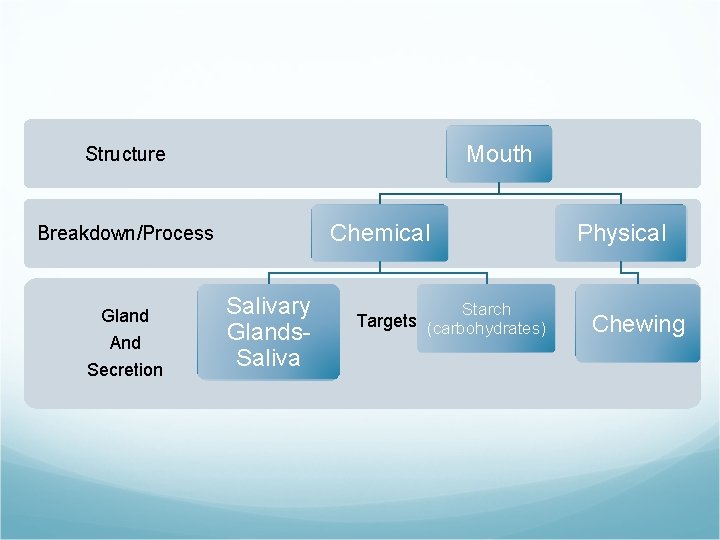
Mouth Structure Chemical Breakdown/Process Gland And Secretion Salivary Glands. Saliva Physical Starch Targets (carbohydrates) Chewing
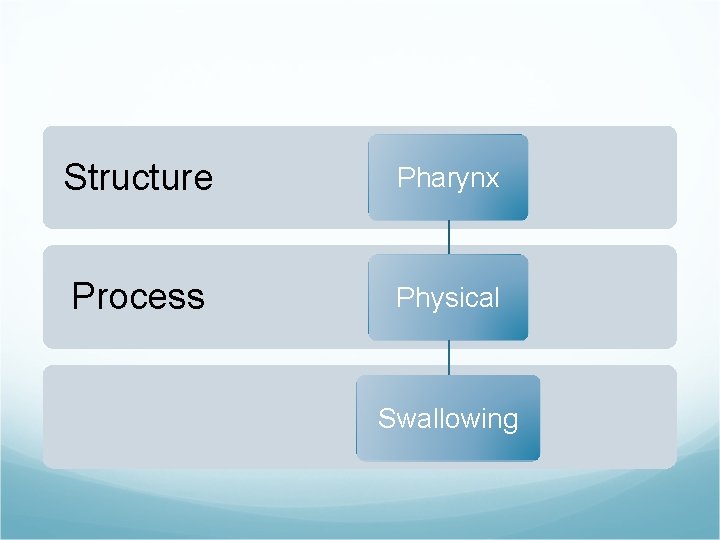
Structure Pharynx Process Physical Swallowing
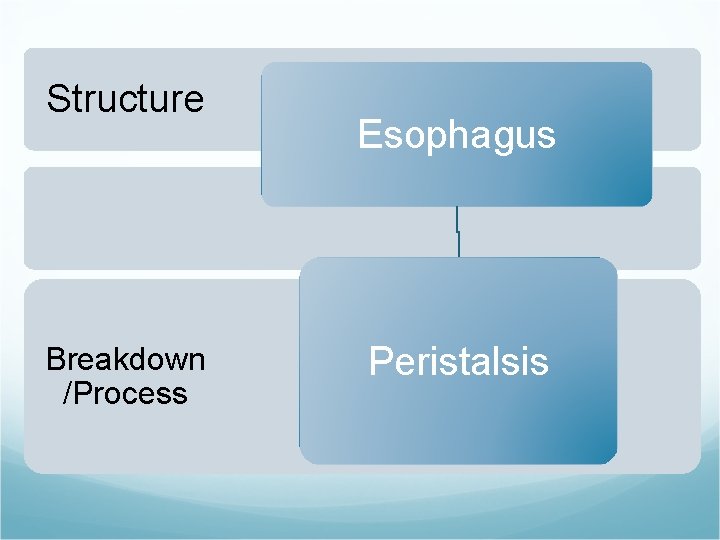
Structure Breakdown /Process Esophagus Peristalsis

Structure Stomach Breakdown Gland And Secretion Chemical Gastric glands. Gastric Juice Targets Proteins Physical Churning
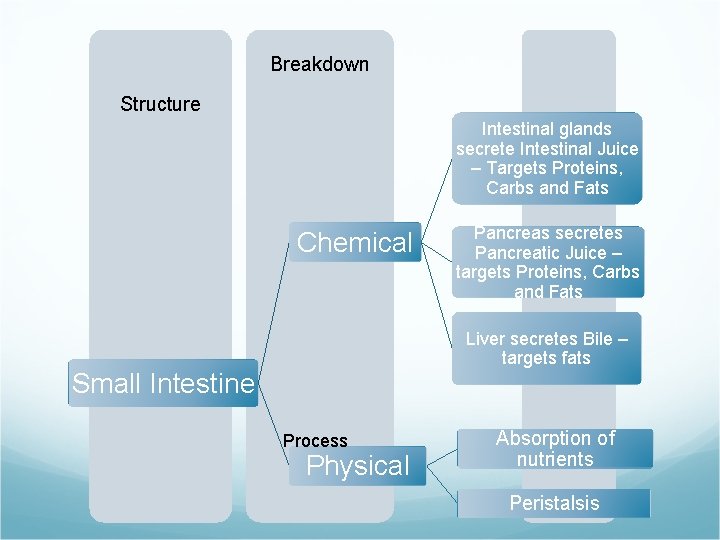
Breakdown Structure Intestinal glands secrete Intestinal Juice – Targets Proteins, Carbs and Fats Chemical Pancreas secretes Pancreatic Juice – targets Proteins, Carbs and Fats Liver secretes Bile – targets fats Small Intestine Process Physical Absorption of nutrients Peristalsis

Structure Large Intestine Process Physical Absorption of water Peristalsis

Nutrition Function of different nutrients Proteins – Heal and Grow Carbohydrates – Provide Energy Fats – Store Energy Water Vitamins Regulate Metabolism Minerals

Nutrition Amount of energy from each nutrient Protein – 17 k. J/g Carbohydrates – 17 k. J/g Fats – 37 k. J/g The nutrient that gives you the most energy tells us the function of the food (if you get the most energy from protein, then the food is used to heal and grow)

Nutrition Examples of foods that provide each nutrient Proteins – Dairy products (milk, cheese), meat, fish, eggs Carbohydrates – Sugar, breads, cereals, pasta, potatoes, rice Fats – Dairy products (except skimmed), butter, fried food Water – Juice, soup, water Vitamins – Fruits, vegetables, dairy products Minerals – Dairy products, seafood, fruits, vegetables

Nutrition How many grams do you need each day? Carbohydrates: 500 g Fat: 75 g Protein: 2 g per Kg of your mass

Properties Cobalt Chloride paper – turns pinking beige in the presence of WATER Litmus paper Turns RED in the presence of an ACID Turns BLUE in the presence of a BASE Go over Liquid Identification table

Properties Characteristic Properties Non-Characteristic Properties Chemical property Physical property

Density (a characteristic property) Density = mass / volume Density of distilled water = 1 g/m. L

Organization of Matter Difference between molecule, compound, atom and element Molecule – group of 2 or more atoms bonded together Ex. HCl, O 2, H 2 Compound – group of 2 or more DIFFERENT atoms bonded together Ex. Na. OH, NH 3 Atom(s) – smallest particle of matter Ex. O, 4 H, 3 Al Element – contains only one type of atom Ex. Fe, O, H

Organization of Matter Drawing Molecules Ex. NH 3 N H H H
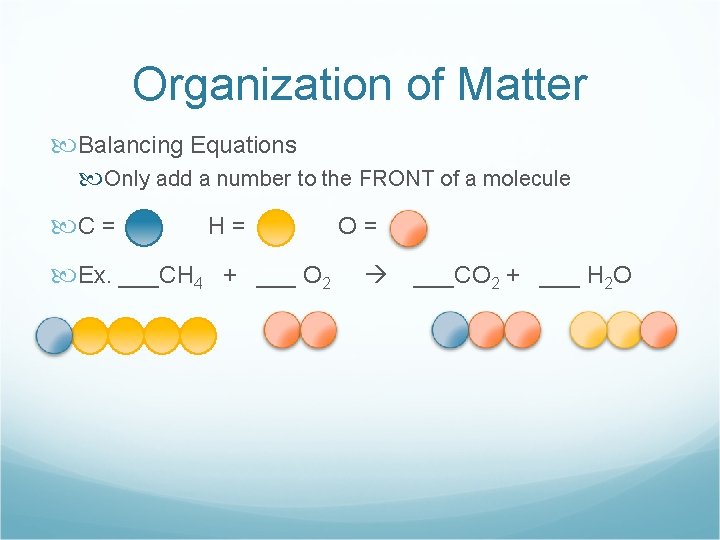
Organization of Matter Balancing Equations Only add a number to the FRONT of a molecule C = H= Ex. ___CH 4 + ___ O 2 O= ___CO 2 + ___ H 2 O
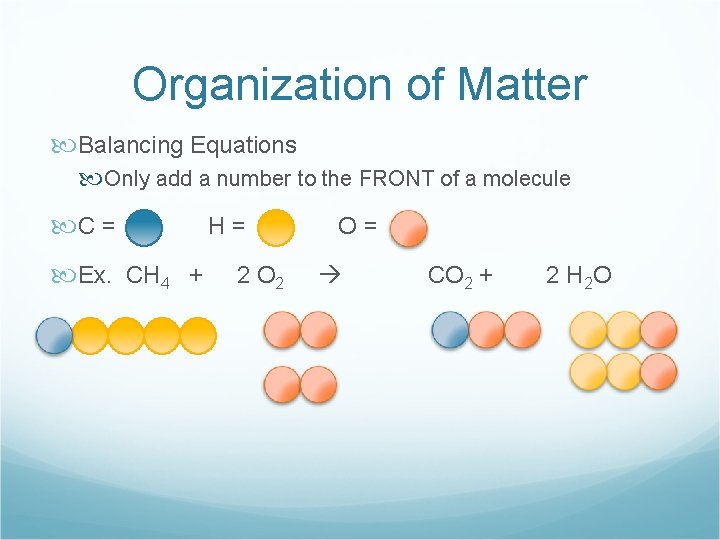
Organization of Matter Balancing Equations Only add a number to the FRONT of a molecule C = Ex. CH 4 + H= 2 O= CO 2 + 2 H 2 O

Types of Reactions Synthesis – Formation of a complex molecule from atoms or simpler molecules (BUILD) Decomposition – Transformation of complex molecules into simpler molecules or atoms (BREAK DOWN) Oxidation – Reaction involving OXYGEN

Respiration Pathway (structure + process) Ex: In the nasal passages, mucous warms and moistens air and hairs filter the air Diffusion – passage of molecules through a semipermeable membrane from high concentration to low concentration

Blood types – what does this mean? Who can donate and receive from whom Agglutination Blood components

Circulation Systemic/Pulmonary Circulation (Pathway blood follows) Label the diagram of the heart Heart problems (blockage, high blood pressure)
- Slides: 22