Parts of an atom Label the diagram Atoms
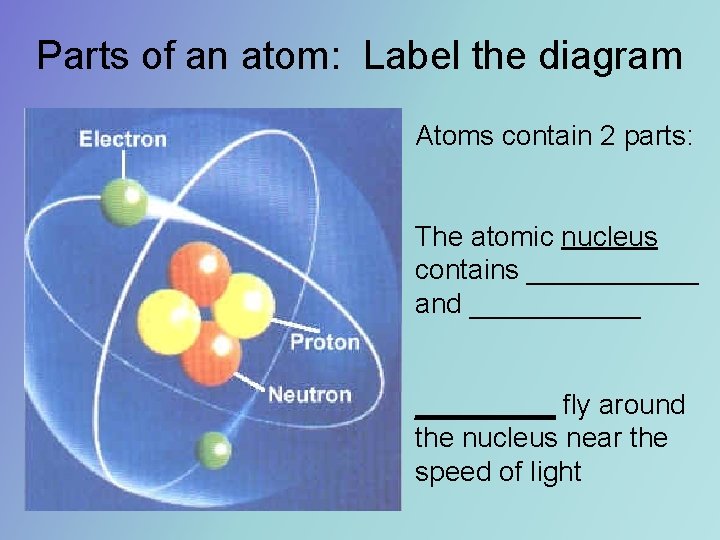
Parts of an atom: Label the diagram Atoms contain 2 parts: The atomic nucleus contains ______ and ______ fly around the nucleus near the speed of light

What Atoms are made of • Atoms are made of 3 parts. • Protons are found in the atomic nucleus. They have a _____electrical charge. • Neutrons are also found in the atomic nucleus. They have _______. • Electrons are found outside of the nucleus. They have a ______charge.
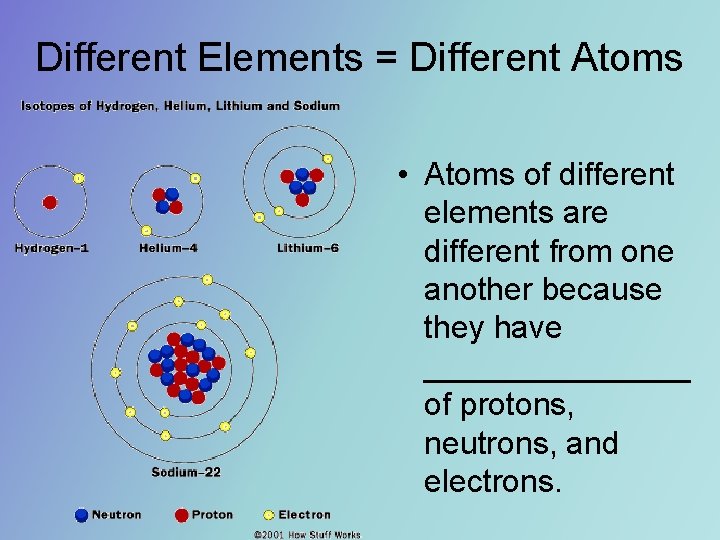
Different Elements = Different Atoms • Atoms of different elements are different from one another because they have ________ of protons, neutrons, and electrons.

Atomic Number • The atomic number is the _________________ in an atom’s nucleus. • In a neutral atom (charge = 0), there are _____ numbers of positive protons and negative electrons. • If atoms ______electrons they become negative, and if they ____electrons, they become positive.

What causes atomic bonds? • When atoms _________________ to one another, that is called a chemical reaction. • Why do chemical reactions happen?

It’s all about Potential Energy • Atoms bond to get a lower energy configuration. Arrows indicate increasing potential energy

But Why? ELECTRONS DETERMINE HOW ATOMS BOND • Atoms that have a full outer energy level are very stable. This is the driving force of bonding reactions. • Atoms bond together in a way that fills their outermost electron energy level. (2 or 8) • This is the lowest energy configuration possible.
Types of Bonds Type of bond What happens Example molecule

Ionic Bonds • An atom loses 1 or more valence electrons, giving it a positive charge. • Another atom gains the electron(s) and becomes negatively charged • + & - ions attract. + e- e. Na e- - e- Cl ee- e- e-
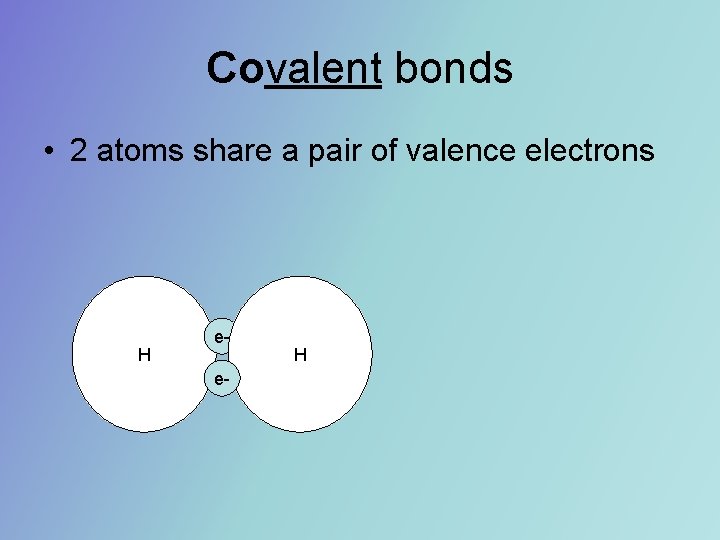
Covalent bonds • 2 atoms share a pair of valence electrons H ee- H

TWIST: Polar Covalent Bonds • In a polar covalent bond, a pair of electrons is shared between two atoms… unevenly • One part of the molecule now has a small – charge, and the other has a small + charge Polar covalent Electron Sharing 100% Non-polar covalent 100% Ionic

Recap • Atoms are made of positive protons, neutrons with no charge, and negative electrons. • The number of protons and electrons determines what charge an atom will have. • Chemical reactions occur when atoms change how they are bonded together. • Atomic bonds can be ionic, covalent, or polar covalent. – Ionic: electron(s) transferred – Covalent: pairs of electrons are shared – Polar covalent: pairs of electrons are shared unevenly.
- Slides: 12