Particulate trace metals Phoebe Lam Marine Bioinorganic Chemistry

Particulate trace metals Phoebe Lam Marine Bioinorganic Chemistry lecture November 22, 2011

outline • Why are particles important • How do we sample for particulate trace metals (suspended, sinking) • Techniques for analysis • Sample profiles (bulk) • Sample profiles (speciation)

Why are particles important to trace metal (TM) cycling? • • • Source of lithogenic TMs (dust, mobilization of continental margin and benthic sediments) Participate in internal cycling of TMs: release some TMs into solution, provide surfaces for scavenging TMs out of solution; biological uptake and remineralization Are the ultimate sink of dissolved trace metals (vertical particle export and removal to sediments)

Sampling for suspended particles 47 mm 142 mm filter holders 293 mm Gas line to overpressure 47 mm or 25 mm filter holder goes here GO-Flo filtration: 10 L, size fractions hard MULVFS: Multiple Unit Large Volume in-situ Filtration System (ship power): <12, 000 L, 3 flow paths, size fractionated (Jim Bishop) Mc. Lane batteryoperated in-situ pump: <1000 L, size fractionated

Sampling for sinking particles PIT-style surface-tethered sediment trap, adapted for trace metal clean collection (Carl Lamborg) Using 234 Th/238 U disequilibrium and particulate 234 Th: TM ratios (Weinstein and Moran 2005)

The basic analysis: applying crustal ratios to total digests • Total digests using (sub)boiling strong acids with HF to dissolve aluminosilicates Sherrell and Boyle, 1992, after Taylor 1964 GCA
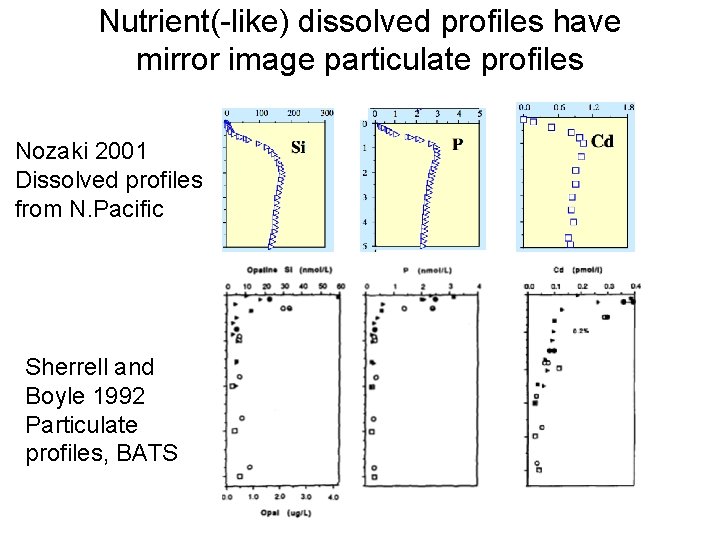
Nutrient(-like) dissolved profiles have mirror image particulate profiles Nozaki 2001 Dissolved profiles from N. Pacific Sherrell and Boyle 1992 Particulate profiles, BATS
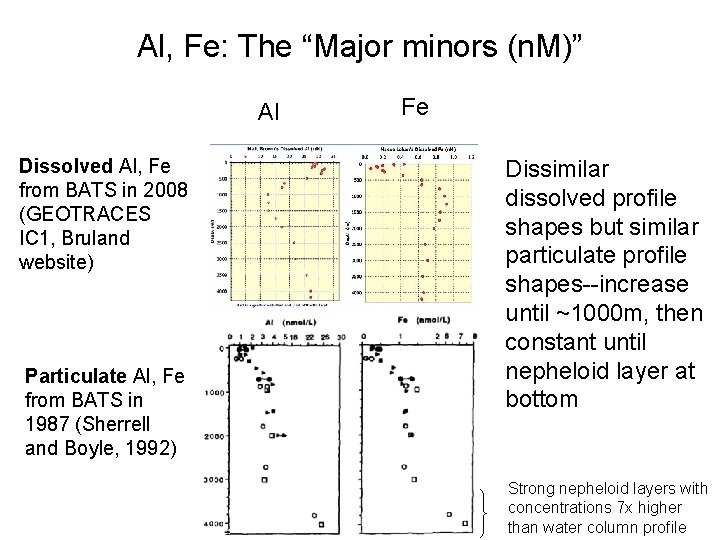
Al, Fe: The “Major minors (n. M)” Al Dissolved Al, Fe from BATS in 2008 (GEOTRACES IC 1, Bruland website) Particulate Al, Fe from BATS in 1987 (Sherrell and Boyle, 1992) Fe Dissimilar dissolved profile shapes but similar particulate profile shapes--increase until ~1000 m, then constant until nepheloid layer at bottom Strong nepheloid layers with concentrations 7 x higher than water column profile
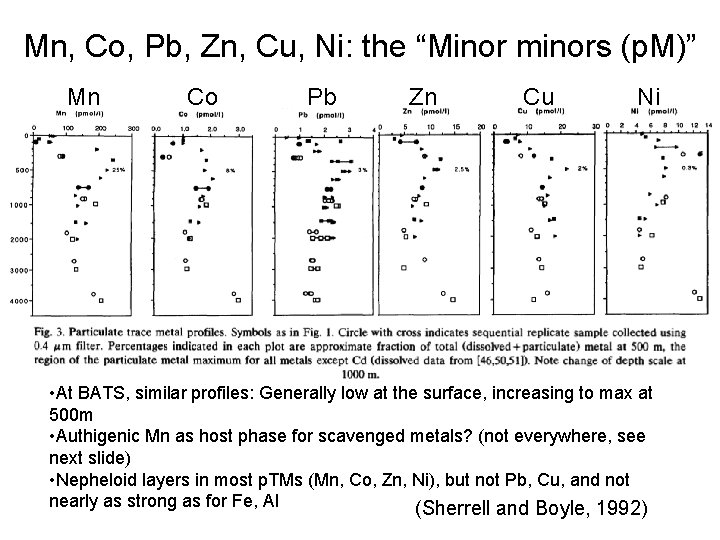
Mn, Co, Pb, Zn, Cu, Ni: the “Minor minors (p. M)” Mn Co Pb Zn Cu Ni • At BATS, similar profiles: Generally low at the surface, increasing to max at 500 m • Authigenic Mn as host phase for scavenged metals? (not everywhere, see next slide) • Nepheloid layers in most p. TMs (Mn, Co, Zn, Ni), but not Pb, Cu, and not nearly as strong as for Fe, Al (Sherrell and Boyle, 1992)
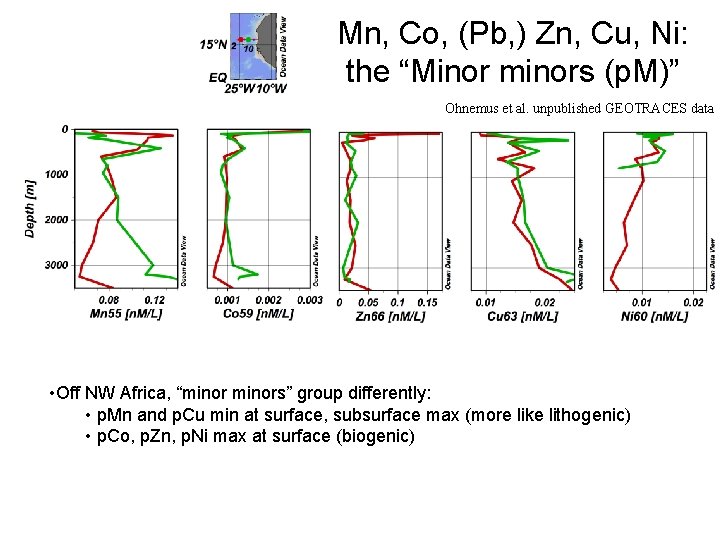
Mn, Co, (Pb, ) Zn, Cu, Ni: the “Minor minors (p. M)” Ohnemus et al. unpublished GEOTRACES data • Off NW Africa, “minors” group differently: • p. Mn and p. Cu min at surface, subsurface max (more like lithogenic) • p. Co, p. Zn, p. Ni max at surface (biogenic)
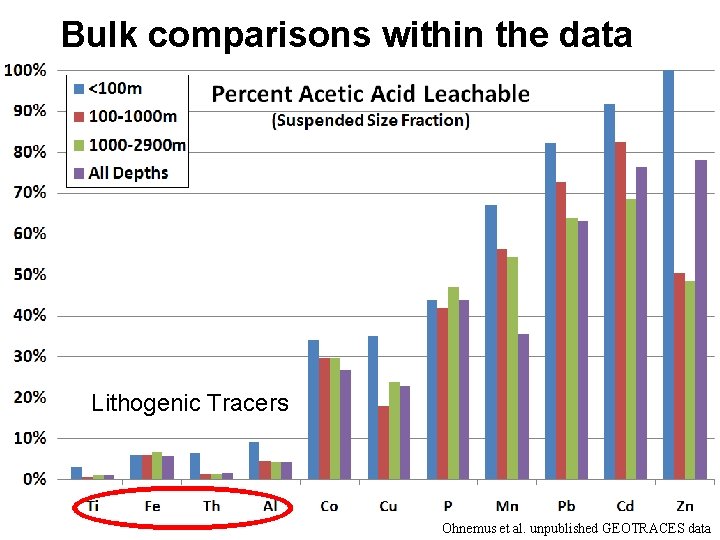
Bulk comparisons within the data Lithogenic Tracers Ohnemus et al. unpublished GEOTRACES data

Lithogenic contribution to p. TMs % particulate Al: <10% Fe: ~50% Mn: <25% Co: <10% Zn: <5% Cu: <5% Ni: <5% Cd: <5% Pb: <5% • Lithogenics are strong sources for Al, Fe everywhere, moderate for Mn and Co, not at all for Zn, Cu, Ni (? ), Cd, Pb • Fe has the highest %particulate (Sherrell and Boyle, 1992)
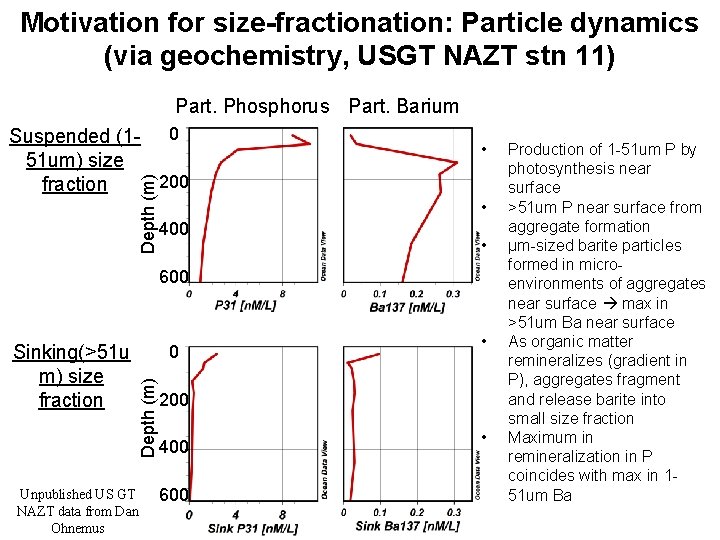
Motivation for size-fractionation: Particle dynamics (via geochemistry, USGT NAZT stn 11) Part. Phosphorus Part. Barium Depth (m) Suspended (1 - 0 51 um) size 200 fraction • • 400 • 600 0 Depth (m) Sinking(>51 u m) size fraction Unpublished US GT NAZT data from Dan Ohnemus • 200 400 600 • Production of 1 -51 um P by photosynthesis near surface >51 um P near surface from aggregate formation μm-sized barite particles formed in microenvironments of aggregates near surface max in >51 um Ba near surface As organic matter remineralizes (gradient in P), aggregates fragment and release barite into small size fraction Maximum in remineralization in P coincides with max in 151 um Ba
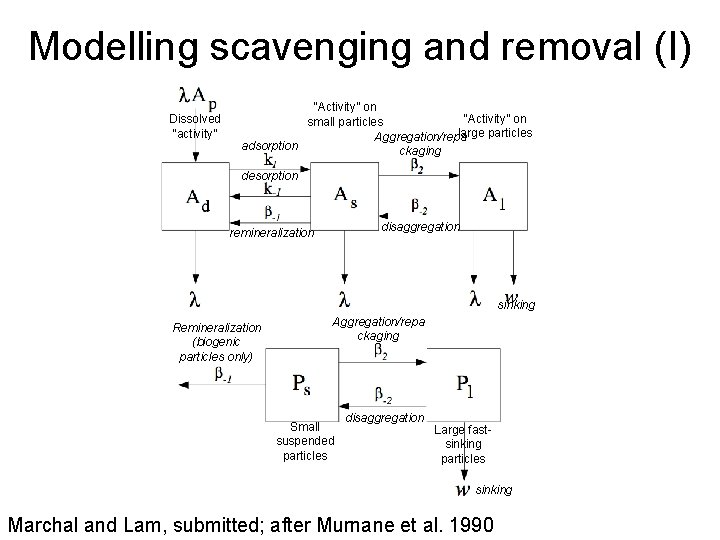
Modelling scavenging and removal (I) Dissolved “activity” “Activity” on small particles large particles Aggregation/repa adsorption ckaging desorption disaggregation remineralization sinking Remineralization (biogenic particles only) Aggregation/repa ckaging Small suspended particles disaggregation Large fastsinking particles sinking Marchal and Lam, submitted; after Murnane et al. 1990
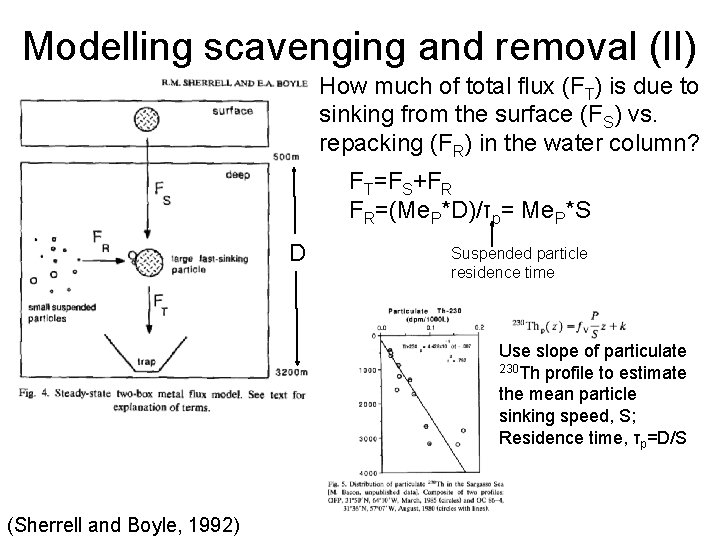
Modelling scavenging and removal (II) How much of total flux (FT) is due to sinking from the surface (FS) vs. repacking (FR) in the water column? FT=FS+FR FR=(Me. P*D)/τp= Me. P*S D Suspended particle residence time Use slope of particulate 230 Th profile to estimate the mean particle sinking speed, S; Residence time, τp=D/S (Sherrell and Boyle, 1992)

Modelling scavenging and removal (III) -repackaging flux (FR) provides ~30% of total flux out of surface (except Cd: 80%, Zn: 10%); i. e. Most of total flux due to flux out of surface (FS) (Sherrell and Boyle, 1992)
Pools of particulate trace metals Biological Surface adsorbed Authigenic particles Lithogenic particles
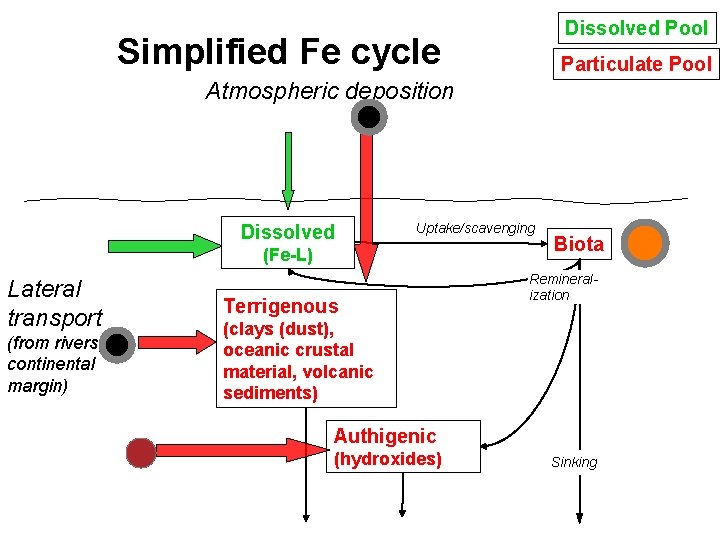
Dissolved Pool Simplified Fe cycle Particulate Pool Atmospheric deposition Dissolved Uptake/scavenging (Fe-L) Lateral transport (from rivers, continental margin) Terrigenous Biota Remineralization (clays (dust), oceanic crustal material, volcanic sediments) Authigenic (hydroxides) Sinking

How to distinguish between different pools? ? Leaching methods (not exhaustive!): “biogenic”: weak acid+mild reductant+heat (Berger et al. 2007); total-lithogenic (Frew et al. 2006) “surface adsorbed”: oxalate wash (Tovar-Sanchez et al. 2004) “authigenic”: mild reductant+acid (eg. Poulton and Canfield 2005) “lithogenic”: strong acid digest (w/ HF) and crustal Al: TM ratio (eg. Sherrell and Boyle 1992; Frew et al. 2006) Biological Surface adsorbed Authigenic particles Lithogenic particles
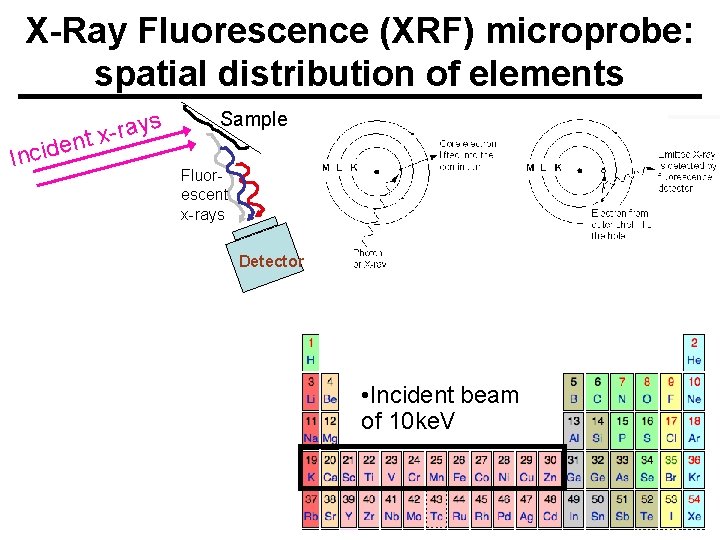
X-Ray Fluorescence (XRF) microprobe: spatial distribution of elements n e d i c In ys a r x t Sample Fluorescent x-rays Detector Wikipedia • Incident beam of 10 ke. V
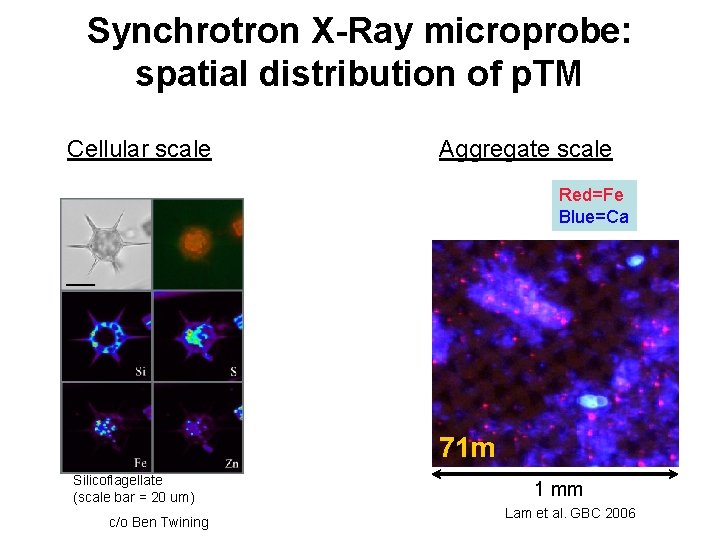
Synchrotron X-Ray microprobe: spatial distribution of p. TM Cellular scale Aggregate scale Red=Fe Blue=Ca 71 m Silicoflagellate (scale bar = 20 um) c/o Ben Twining 1 mm Lam et al. GBC 2006
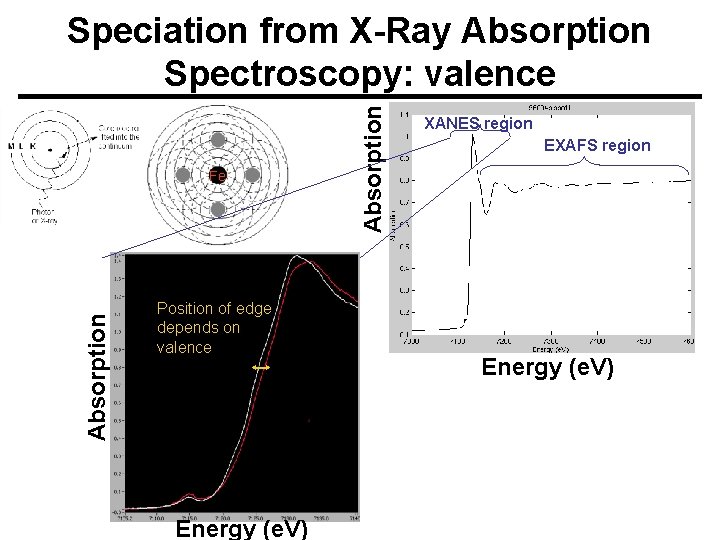
Absorption Fe Position of edge depends on valence Energy (e. V) Absorption Speciation from X-Ray Absorption Spectroscopy: valence XANES region EXAFS region Energy (e. V)
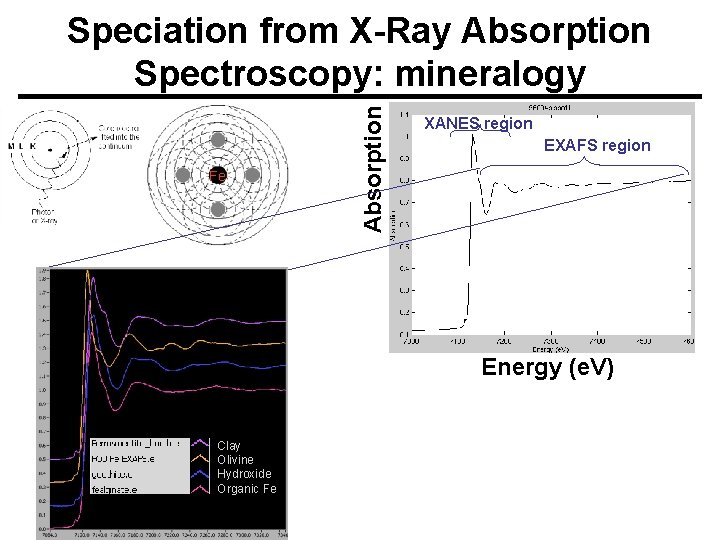
Fe Absorption Speciation from X-Ray Absorption Spectroscopy: mineralogy XANES region EXAFS region Energy (e. V) Clay Olivine Hydroxide Organic Fe
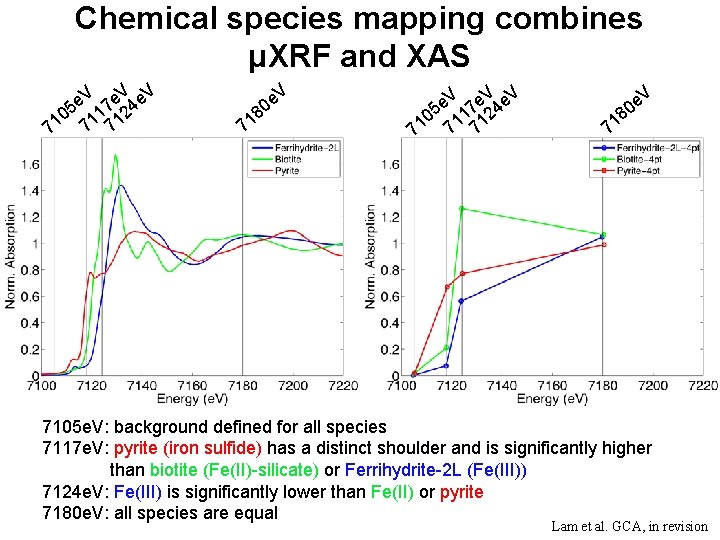
Chemical species mapping combines μXRF and XAS V e 5 17 24 0 71 71 71 8 1 7 V e 0 V e 5 17 24 0 71 71 71 e. V 0 8 71 7105 e. V: background defined for all species 7117 e. V: pyrite (iron sulfide) has a distinct shoulder and is significantly higher than biotite (Fe(II)-silicate) or Ferrihydrite-2 L (Fe(III)) 7124 e. V: Fe(III) is significantly lower than Fe(II) or pyrite 7180 e. V: all species are equal Lam et al. GCA, in revision
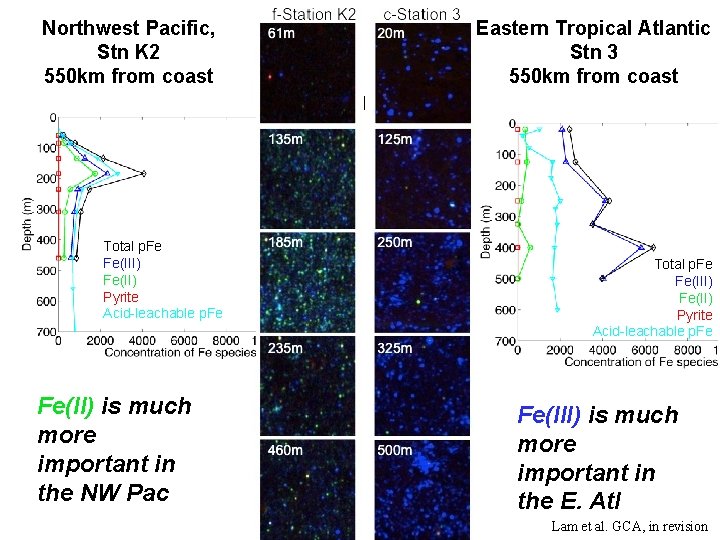
Northwest Pacific, Stn K 2 550 km from coast Total p. Fe Fe(III) Fe(II) Pyrite Acid-leachable p. Fe Fe(II) is much more important in the NW Pac Eastern Tropical Atlantic Stn 3 550 km from coast Total p. Fe Fe(III) Fe(II) Pyrite Acid-leachable p. Fe Fe(III) is much more important in the E. Atl Lam et al. GCA, in revision
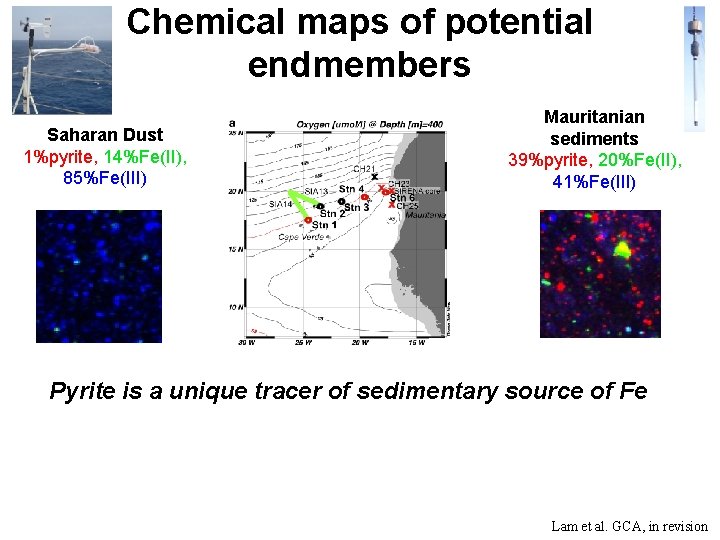
Chemical maps of potential endmembers Saharan Dust 1%pyrite, 14%Fe(II), 85%Fe(III) Mauritanian sediments 39%pyrite, 20%Fe(II), 41%Fe(III) Pyrite is a unique tracer of sedimentary source of Fe Lam et al. GCA, in revision

References
- Slides: 27