Particulate Model of Matter Textbook Chp 7 What

Particulate Model of Matter Textbook Chp 7

What is a model?

What is a model? • Models are man-made things, built to try and replicate things which already exist in real life



What makes a good model? • Which is a better model aeroplane?

What makes a good model? • When comparing between two models, the better model is the one which is closer to real life • In science there also models, which man has come up with, trying to replicate how nature works in real life • However, just like cardboard models, there also better and poorer models • There are some models you will learn in secondary school, but in JC you will learn the better models, making your previously learnt models obsolete

THE PARTICULATE MODEL

The Particulate Model • These things are true in real life: • Matter exists in solid, liquid and gaseous states • Solids, liquids and gases have different properties • By heating & cooling matter, you can convert from one state to another

The Particulate Model • As scientists, we are interested in coming up with explanations • Thus, we create models to attempt to explain what happens in real life • Thus, the particulate model (or Kinetic Model) was proposed in order to explain the differences in the previous table
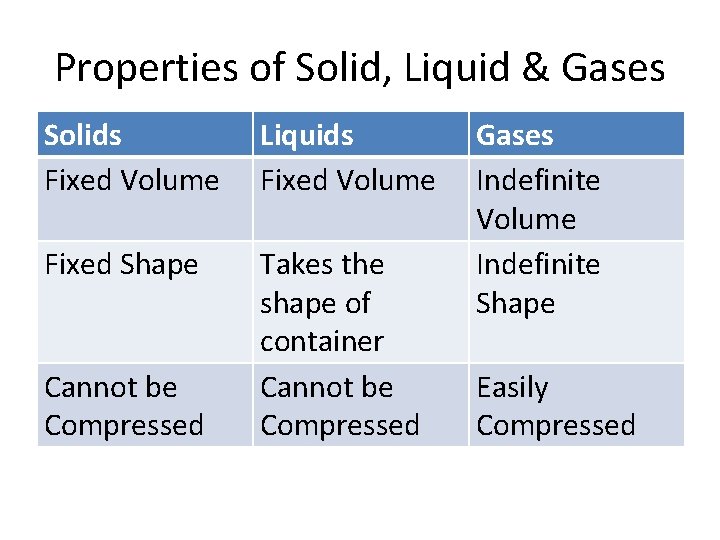
Properties of Solid, Liquid & Gases Solids Fixed Volume Liquids Fixed Volume Fixed Shape Takes the shape of container Cannot be Compressed Gases Indefinite Volume Indefinite Shape Easily Compressed

The Particulate Model • Proposal 1: All matter (solid, liquid or gas) exists as tiny particles (e. g. tiny balls) • Proposal 2: There are forces of attraction in between these tiny particles • Proposal 3: The temperature of an object is related to how fast these tiny particles are moving

Particulate Model - Solid • Forces between particles are very strong • Particles are closely packed to each other • As a result they are not free to move about very much • They can only vibrate about fixed positions • As a result, they are arranged in very regular and orderly arrangements • If a solid gets heated up, they vibrate more vigorously

Particulate Model - Liquid • Liquids have moderate force between particles • They still stick to each other, but are free enough to move around • As a result, their arrangement is irregular and random • But they are still quite close to each other (which is why it is hard to compress liquid)

Particulate Model - Gas • Gases have very weak forces between the particles • Also, particles are very far apart from each other • As a result, they are free to move about in high speeds • However, despite being far apart, there are still so many particles that they collide into each other regularly

Diffusion • When someone sprays perfume at one corner of the room, can you smell it at the other end of the room? • This phenomenon is called diffusion • Diffusion is the spreading of a substance from an area of higher concentration to an area of lower concentration • Video: bromine diffusion in air and in vacuum: http: //www. youtube. com/watch? v=Aa 03 WKRB 2 z 0

HEATING CURVES
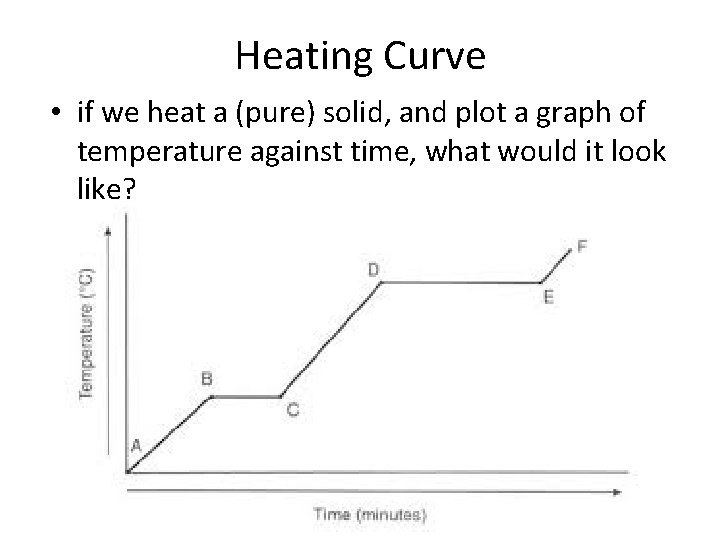
Heating Curve • if we heat a (pure) solid, and plot a graph of temperature against time, what would it look like?

Heating Curve • Notice whenever there is change of state (e. g. melting or boiling) the temperature is constant • When you are heating an object, you are supplying energy to the object • Remember Proposal 3: The temperature of an object is related to how fast these tiny particles are moving • Normally (when not changing state), the energy supplied to an object is increasing the speed of the particles moving (and hence increasing temperature)

Heating Curve • However, at the melting point (or boiling point) of a substance, the energy goes into overcoming the forces of attraction between the particles, instead of increasing the speed of the particles • In chemistry, we say that the energy is used to break the bonds between the particles

Expansion when heating • Previously we mentioned that when we heat up the object, the particles move faster • In solids or liquids, when particles move faster, they will naturally move further apart from each other • This results in the expansion of the object (i. e. they become bigger when heated up)

Boiling vs Evaporation • 2 differences between boiling and evaporation: • Boiling occurs only at the boiling point but evaporation occurs at any temperature • Evaporation only happens at the surface of the liquid, but when boiling occurs, the whole liquid is being converted into gas

Summary • • • What is a model, what makes a good model Properties of solid, liquid, gas Particulate model for solid, liquid, gas Heating Curve (melting, boiling) Expansion when heating Boiling vs Evaporation

Did You Know? (not in syllabus) • There actually FOUR states of matter, not three. What’s the fourth state? (hint: what state of matter is fire? ) • There are substances which are not exactly in any one state of matter, e. g. hairspray, gel, shaving foam. These are a special kind of mixtures called “colloids”

For Fun • Bill Nye the Science Guy: • http: //www. youtube. com/watch? v=Gxwj 24 m REy. A
- Slides: 25