Particles solids liquids and gases What are three








- Slides: 11

Particles, solids liquids and gases

What are three states of matter? At any given temperature, all substances exist in one of the three states of matter. solid liquid gas

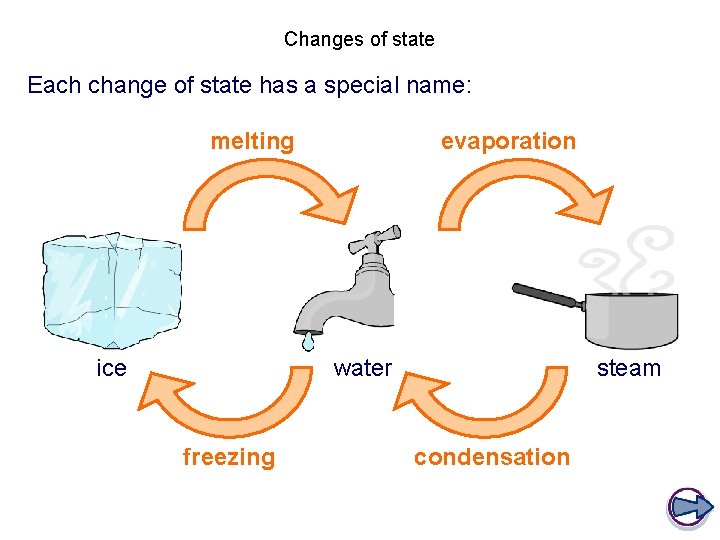
Changes of state Each change of state has a special name: melting ice evaporation water freezing steam condensation

Particles The differences between solids, liquids and gases can be explained by looking at the particles. l All substances are made up of particles. l The particles are attracted to each other. Some particles are attracted strongly to each other, and others weakly. l The particles move around. They are described as having kinetic energy. l The kinetic energy of the particles increases with temperature.

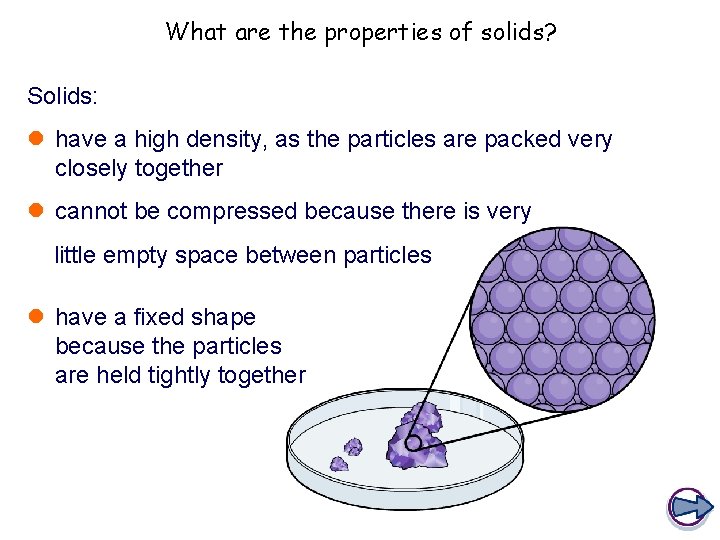
What are the properties of solids? Solids: l have a high density, as the particles are packed very closely together l cannot be compressed because there is very little empty space between particles l have a fixed shape because the particles are held tightly together

What are the properties of liquids? Liquids: l have a fairly high density because the particles are close together l cannot be compressed because there is very little empty space between particles l take up the shape of its container because the particles can move l can diffuse because the particles are able to change places.

What are the properties of gases? Gases: l have a low density because the particles are spaced far apart l can be compressed because there is space between particles l have no fixed shape because the particles move about rapidly in all directions l can diffuse because the particles are able to move in all directions.

Melting point and boiling point The temperature at which a substance changes from a solid to a liquid is called its melting point (or freezing point). What is the melting point of pure water? The temperature at which a substance changes from a liquid to a gas is called its boiling point. What is the boiling point of pure water? What state is water at 25 °C, 300 °C and 100 °C?

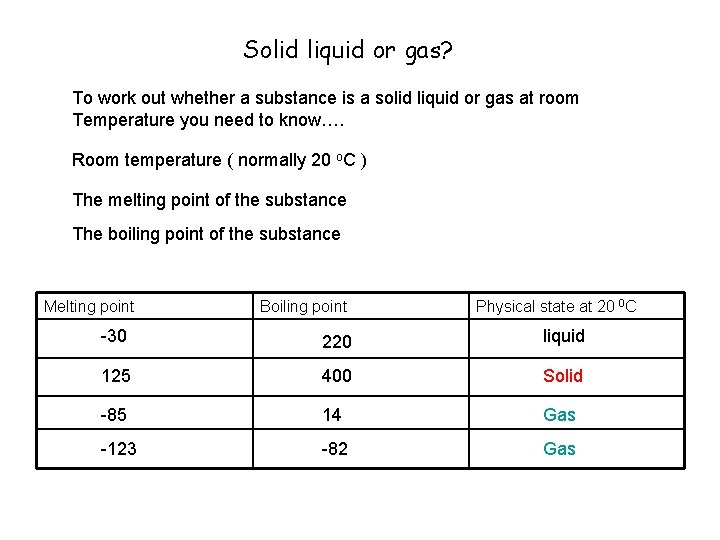
Solid liquid or gas? To work out whether a substance is a solid liquid or gas at room Temperature you need to know…. Room temperature ( normally 20 o. C ) The melting point of the substance The boiling point of the substance Melting point Boiling point Physical state at 20 0 C -30 220 liquid 125 400 Solid -85 14 Gas -123 -82 Gas

Solids, liquids and gases: which one?

Expansion When substances are heated, they get bigger… they Expand. . When substances cool down, they get smaller… the Contract We cannot stop expansion. . But we can let it happen without things going wrong!! Power lines Bridges Railway lines