Particle Theory of Matter Are able to move

Particle Theory of Matter
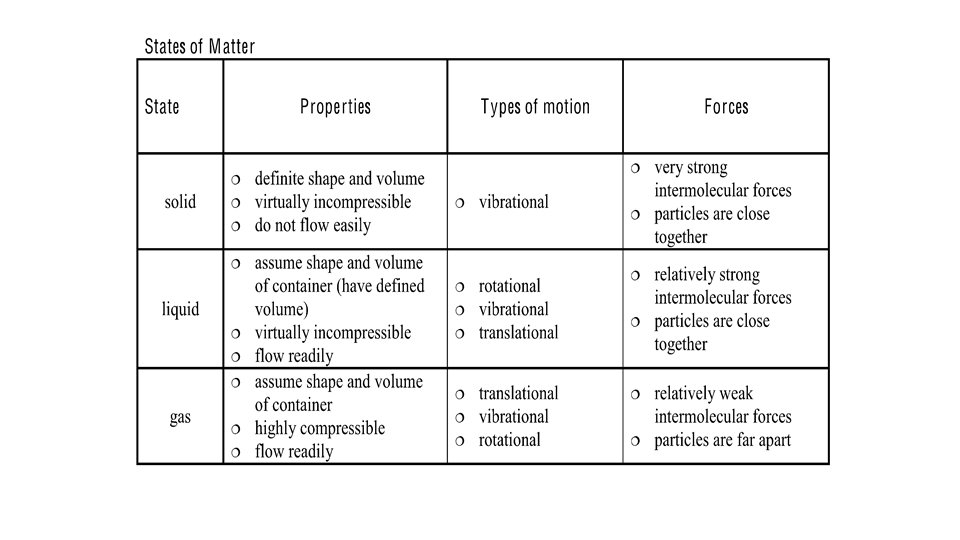

• Are able to move independently • Random motion • Move is straight lines until course is altered by collisions with other particles • Motion is fast • Liquids flow because they are influences by gravity • Gas particles move so fast that gravity does not affect them as much • Compressible • Motion in all directions until all available empty space is filled (expand)


Kinetic Molecular Theory • Theory developed to explain gas behavior. • Theory of moving molecules.

• Assumptions: – Gases consist of a large number of molecules in constant random motion. – Volume of individual molecules negligible compared to volume of container. – Intermolecular forces (forces between gas molecules) negligible. – Energy can be transferred between molecules, but total kinetic energy is constant at constant temperature. – Average kinetic energy of molecules is proportional to temperature.

• Kinetic molecular theory gives us an understanding of pressure and temperature on the molecular level. • Gas molecules have an average kinetic energy. • Each molecule has a different energy.

• When particles are heated: • Particles move faster and have more collisions • Higher kinetic energy helps overcome attractive forces • Particles need more space, making the object expand
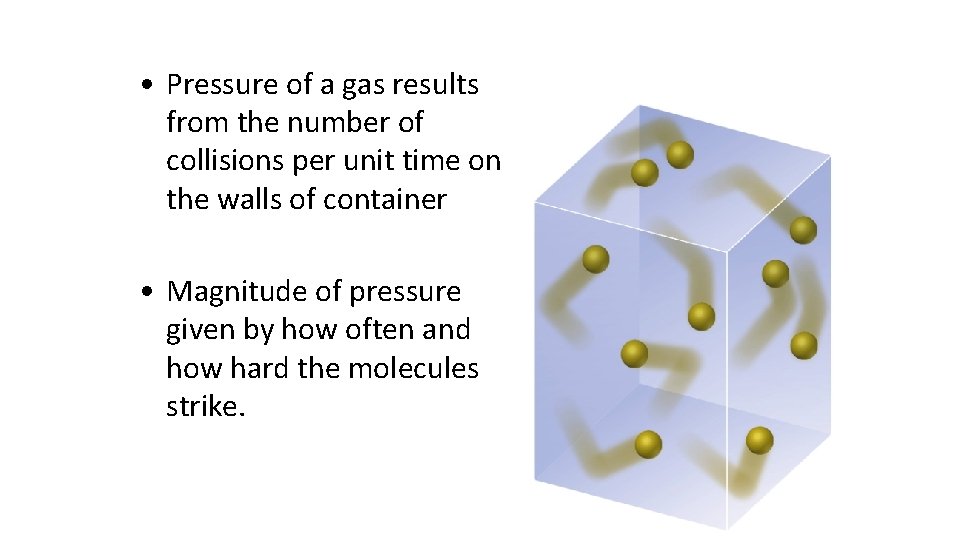
• Pressure of a gas results from the number of collisions per unit time on the walls of container • Magnitude of pressure given by how often and how hard the molecules strike.

• There is a spread of individual energies of gas molecules in any sample of gas. • As the temperature increases, the average kinetic energy of the gas molecules increases.

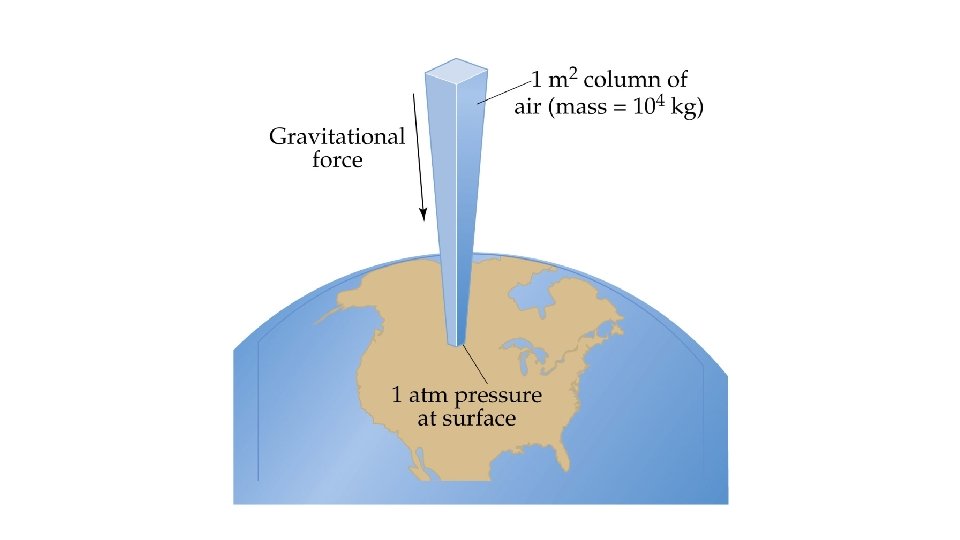
Pressure • Pressure is the force acting on an object per unit area: • Gravity exerts a force on the earth’s atmosphere • Atmospheric pressure is the force per unit area exerted against a surface by the weight of the air above that surface. • A column of air 1 m 2 in cross section exerts a force of 105 N. • The pressure of a 1 m 2 column of air is 100 k. Pa.
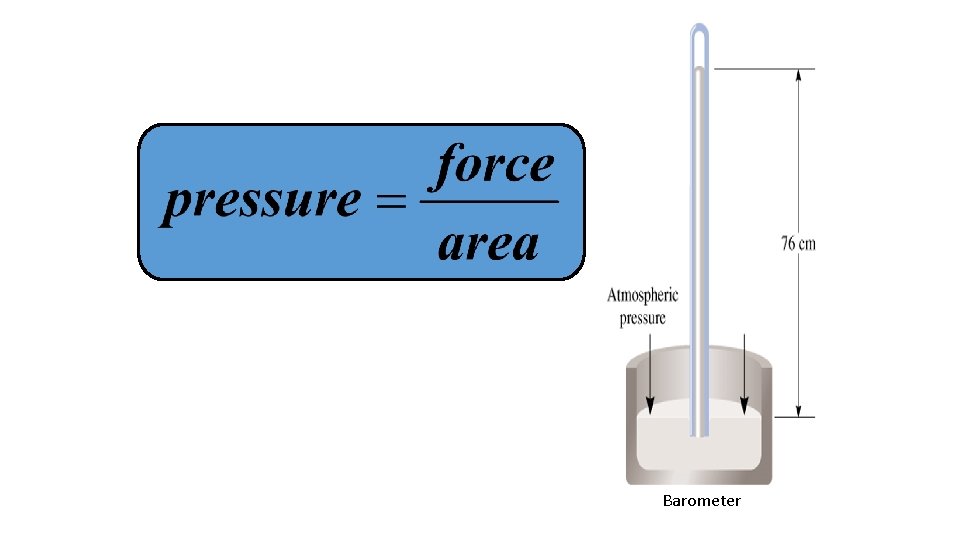
Barometer
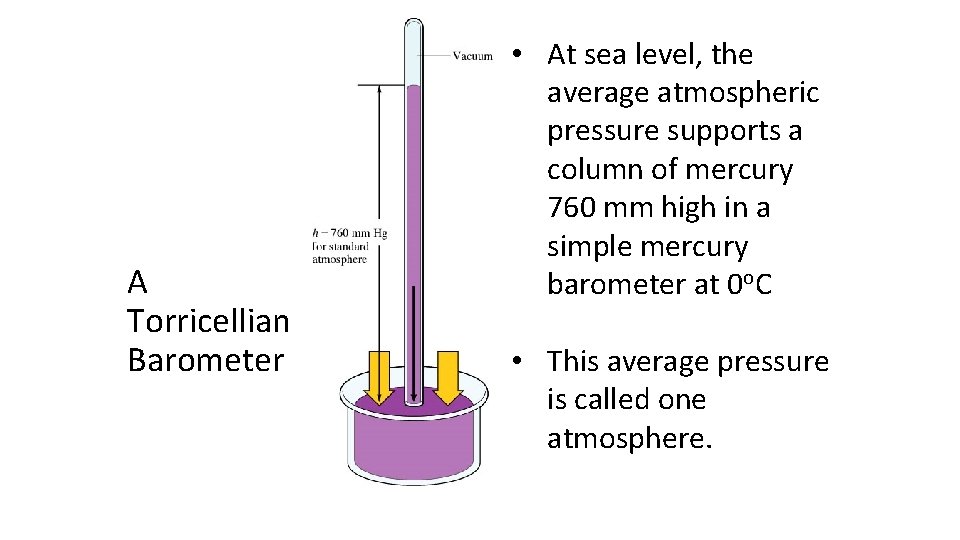
A Torricellian Barometer • At sea level, the average atmospheric pressure supports a column of mercury 760 mm high in a simple mercury barometer at 0 o. C • This average pressure is called one atmosphere.
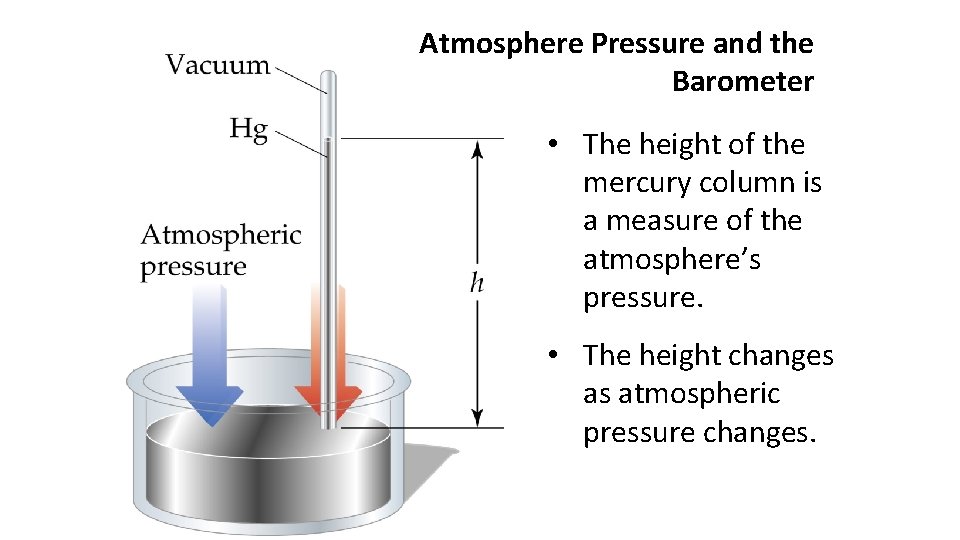
Atmosphere Pressure and the Barometer • The height of the mercury column is a measure of the atmosphere’s pressure. • The height changes as atmospheric pressure changes.
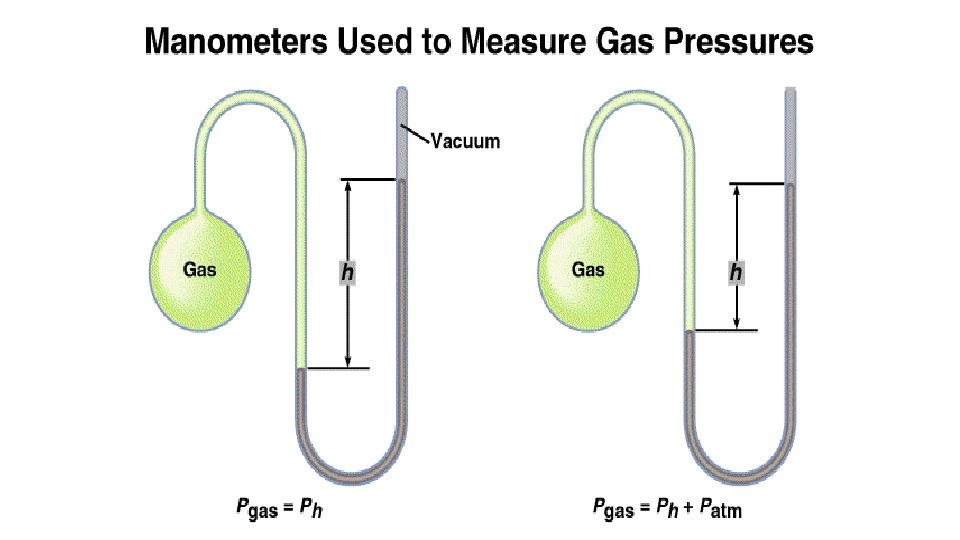
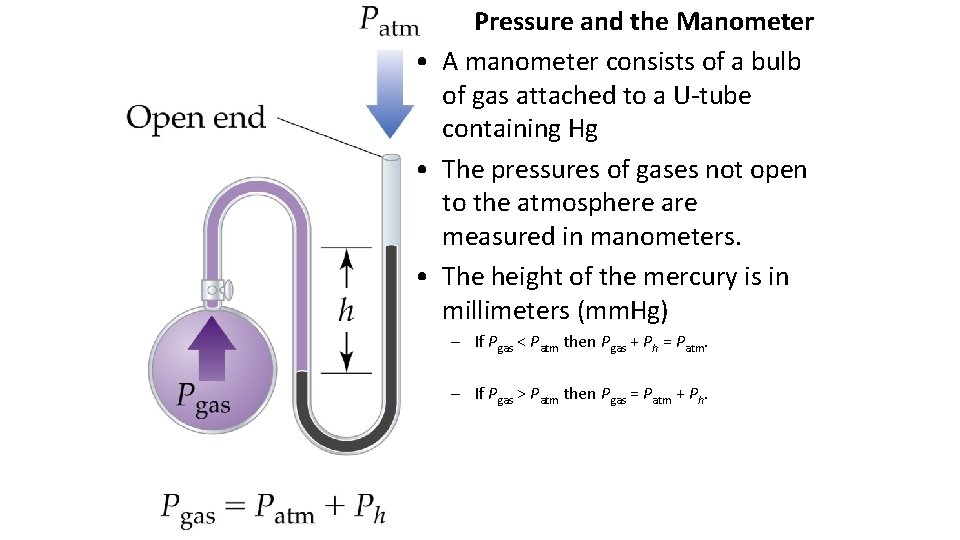
Pressure and the Manometer • A manometer consists of a bulb of gas attached to a U-tube containing Hg • The pressures of gases not open to the atmosphere are measured in manometers. • The height of the mercury is in millimeters (mm. Hg) – If Pgas < Patm then Pgas + Ph = Patm. – If Pgas > Patm then Pgas = Patm + Ph.

Units of Pressure • atmospheres (symbol = atm) • millimeters of mercury (symbol = mm Hg). This unit is also called a torr. • Pascals (symbol = Pa). A Pa is N/m 2. More commonly used is kilo. Pascals (symbol = k. Pa). This is the SI unit for pressure.

Conversion Between Units 1 atm = 760 mm Hg = 101. 325 k. Pa = 101325 Pa

• Example #1 - Convert 0. 875 atm to mm. Hg 1 atm = 760 mm. Hg 0. 875 atm ? ? = 665 mm. Hg • Example #2 - Convert 745. 0 mm. Hg to atm. 1 atm = 760 mm. Hg ? 745. 0 mm. Hg ? = 0. 9802 atm

• Example #3 - Convert 0. 955 atm to k. Pa 1 atm = 101. 325 k. Pa 0. 955 atm ? ? = 96. 8 k. Pa • Example #4 - Convert 98. 35 k. Pa to atm 1 atm = 101. 325 k. Pa ? 98. 35 k. Pa ? = 1. 030 atm

• Example #5 - Convert 740. 0 mm. Hg to k. Pa 760 mm. Hg = 101. 325 k. Pa 740 mm. Hg ? ? = 98. 6 (99) k. Pa • Example #6 - Convert 99. 25 k. Pa to mm. Hg 760 mm. Hg = 101. 325 k. Pa ? 99. 25 k. Pa ? = 744. 4 mm. Hg

Convert the following pressures: 1. 658. 2 mm Hg to k. Pa 2. 1. 85 atm to torr 3. 337. 3 k. Pa to atm 4. 6. 6 x 10 -2 torr to atm 5. 147. 2 k. Pa to torr

Convert the following pressures: 1. 658. 2 mm Hg to k. Pa 87. 75 k. Pa 2. 1. 85 atm to torr 1. 41 x 103 torr 3. 337. 3 k. Pa to atm 3. 329 atm 4. 6. 6 x 10 -2 torr to atm 8. 7 x 10 -5 atm 5. 147. 2 k. Pa to torr 1104 torr

The mercury in a manometer is 46 mm higher on the open end than on the gas bulb end. If atmospheric pressure is 102. 2 k. Pa, what is the pressure of the gas in the bulb?

On a certain day the barometer in a laboratory indicates that the atmospheric pressure is 764. 7 torr. A sample of gas is placed in a vessel attached to an open-end mercury manometer. A meter stick is used to measure the height of the mercury above the bottom of the manometer. The level of mercury in the open end arm of the manometer has a measured height of 136. 4 mm. In the arm that is in contact with the gas has a height of 103. 8 mm. What is the pressure of the gas (a) in atm and (b) in k. Pa?
- Slides: 26