Particle Theory 20 February 2021 Learning Objectives Read
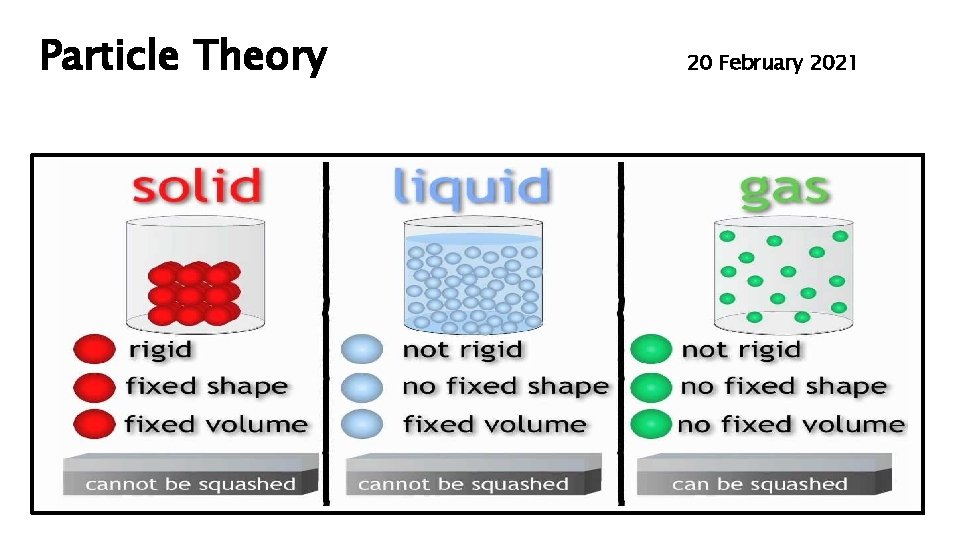
Particle Theory 20 February 2021

Learning Objectives Read pages 297 -300 of your textbook! 1. Describe the particle model 2. Relate the particle model to states of matter and changes of state 3. Explain that moving particles possess kinetic energy 4. Explain that the kinetic energy of a particle increase as its temperature rises 5. Use the particle model to explain various properties of matter including expansion, contraction, pressure and diffusion

Key words… • Particle model • Kinetic energy • Expansion • Contraction • Melting • Freezing sation) ri o p va s a n w o n k o ls a ( n o ti • Evapora • Condensation • Diffusion

Complete page 27 of your booklet using the information on pages 297 -298
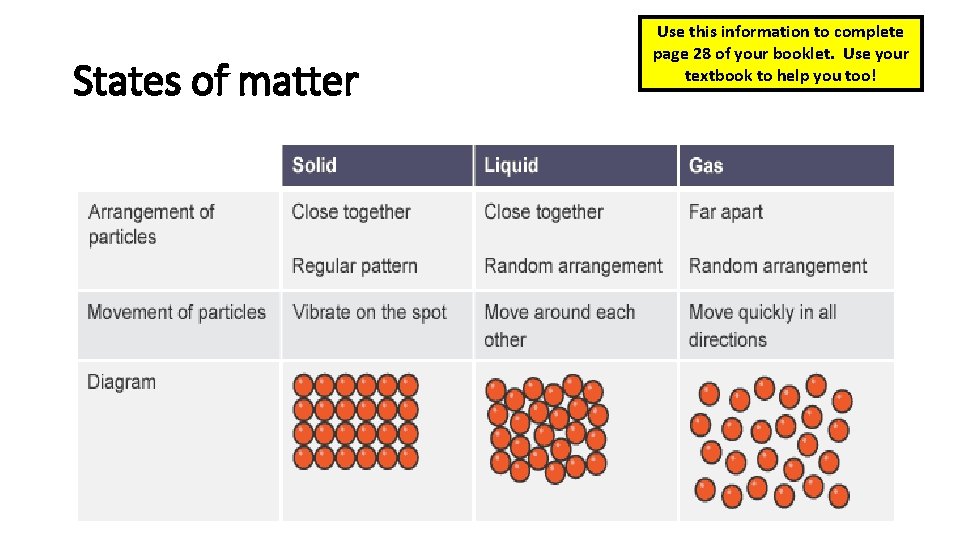
States of matter Use this information to complete page 28 of your booklet. Use your textbook to help you too!

Kinetic Particle Theory – what is it? • The kinetic particle theory explains the properties of the different states of matter. • The particles in solids, liquids and gases have different amounts of energy. • They are arranged differently and move in different ways. https: //www. youtube. com/watch? v=1 zr. Bp. VFv. Rb. M#action=share
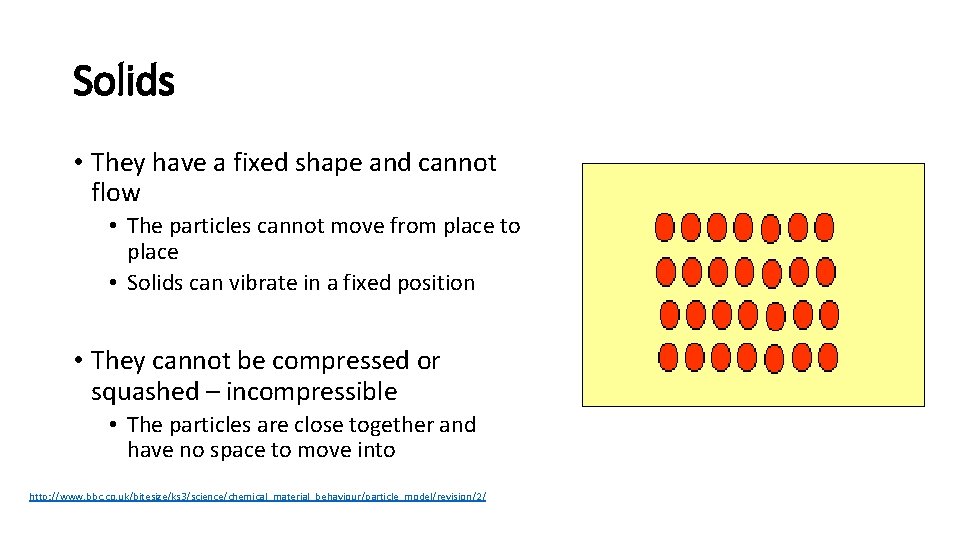
Solids • They have a fixed shape and cannot flow • The particles cannot move from place to place • Solids can vibrate in a fixed position • They cannot be compressed or squashed – incompressible • The particles are close together and have no space to move into http: //www. bbc. co. uk/bitesize/ks 3/science/chemical_material_behaviour/particle_model/revision/2/
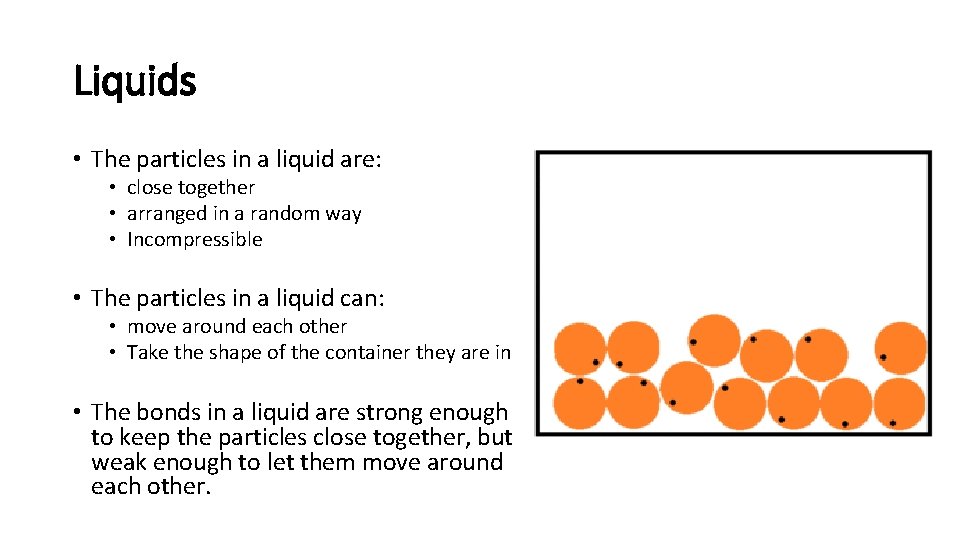
Liquids • The particles in a liquid are: • close together • arranged in a random way • Incompressible • The particles in a liquid can: • move around each other • Take the shape of the container they are in • The bonds in a liquid are strong enough to keep the particles close together, but weak enough to let them move around each other.
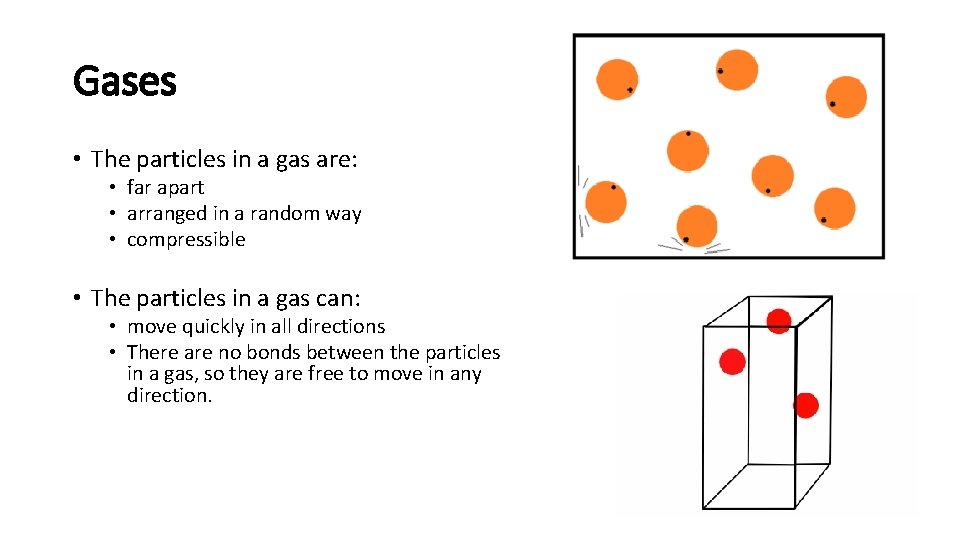
Gases • The particles in a gas are: • far apart • arranged in a random way • compressible • The particles in a gas can: • move quickly in all directions • There are no bonds between the particles in a gas, so they are free to move in any direction.
Compress or not to compress? • Read the paragraphs on solids, liquids and gases on page 298 of your textbook. State Solid • Which states of matter are compressible and incompressible? Can you explain your answer? • Complete the table opposite to complete your notes for this section. Liquid Gas Can it be compressed? Reason
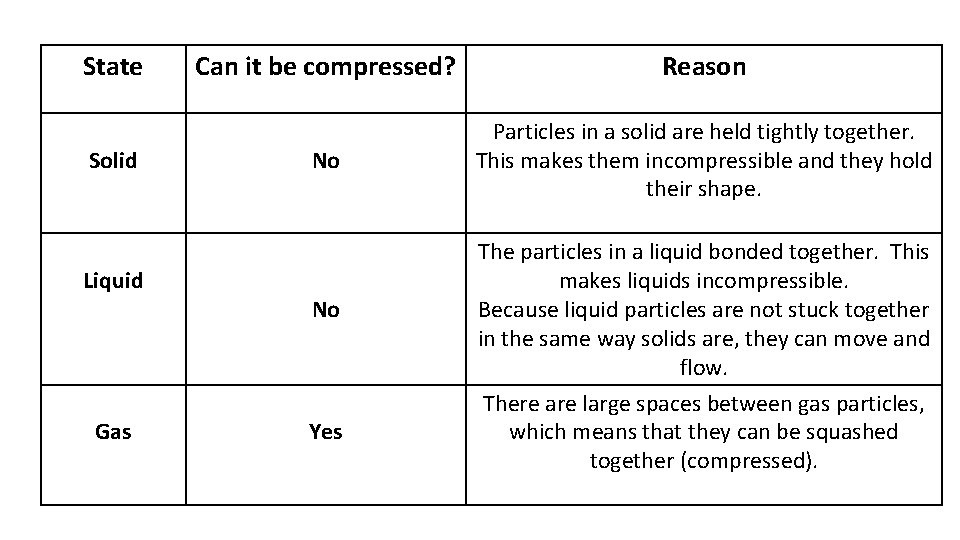
State Solid Liquid Gas Can it be compressed? Reason No Particles in a solid are held tightly together. This makes them incompressible and they hold their shape. No Yes The particles in a liquid bonded together. This makes liquids incompressible. Because liquid particles are not stuck together in the same way solids are, they can move and flow. There are large spaces between gas particles, which means that they can be squashed together (compressed).

Task • Read pages 298 -300 of your textbook • Complete pages 29 – 30 of your booklet

States of matter video – a 20 minute extravaganza!! https: //www. youtube. com/watch? v=p 7 m. OLbp. Ec. A 4#action=share

Changing States • When a solid is heated it melts to become a liquid. • When a liquid is heated it boils and becomes a gas i. e. Boiling. • If a liquid becomes a gas without bubbling we call it evaporation.
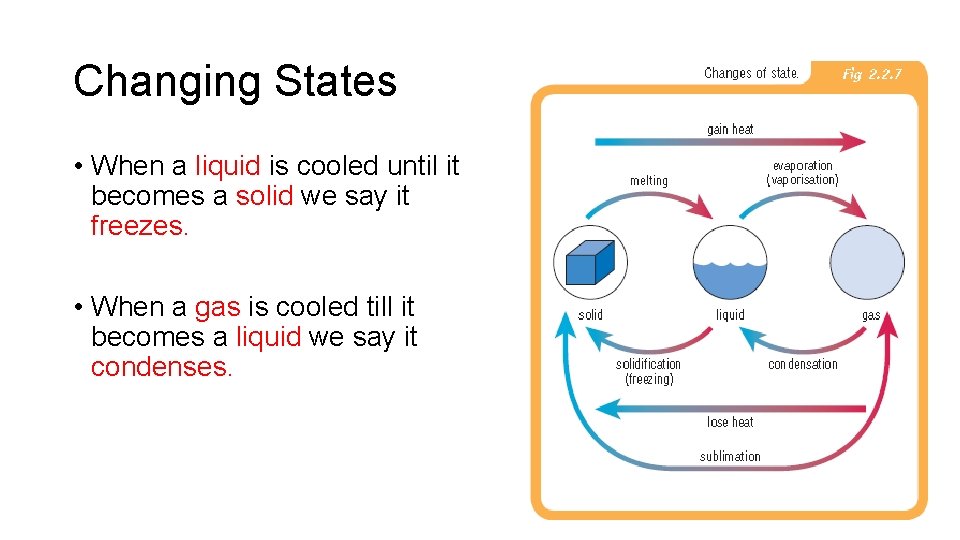
Changing States • When a liquid is cooled until it becomes a solid we say it freezes. • When a gas is cooled till it becomes a liquid we say it condenses.
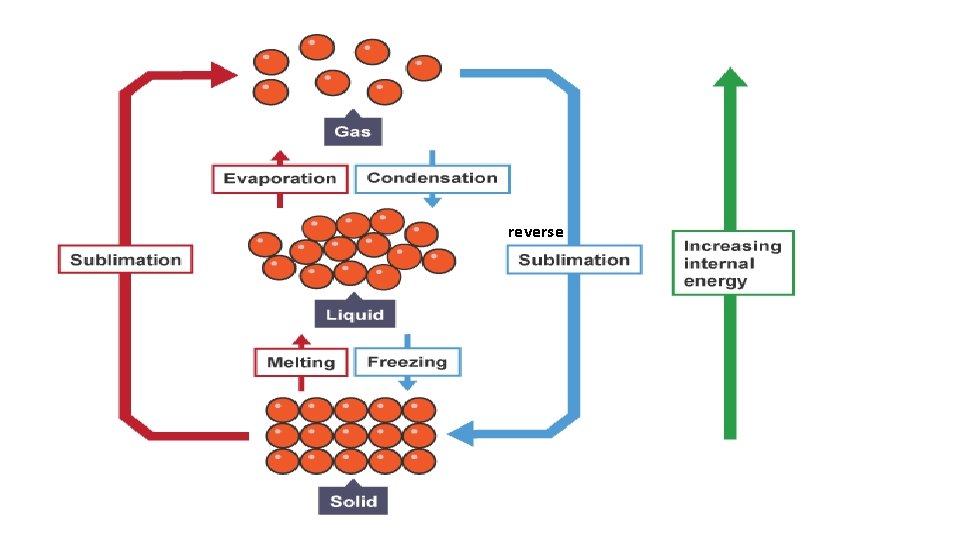
reverse

Changing States-The Particles Explain in terms of the energy of atoms why: 1. 2. 3. 4. Solids melt Liquids evaporate Gases condense Liquids freeze Use pages 25 -26 of your booklet to help you with the answers • Give an example of each. Answering these questions will help you with your assignment!!!

https: //www. youtube. com/watch? v=o. S 8 j. Hfbcme 8#action=share

Task Use pages 299 -300 of your textbook to help you too! 1. Using this link, http: //www. bbc. co. uk/education/guides/zc 9 q 7 ty/revision/3 complete the sections of the table on expansion and contraction on page 32 of your booklet. 2. Using this link, http: //www. bbc. co. uk/bitesize/ks 3/science/chemical_material_behaviour/beh aviour_of_matter/revision/3/ complete the section of the table on pressure. 3. Using this link, http: //www. bbc. co. uk/bitesize/ks 3/science/chemical_material_behaviour/beh aviour_of_matter/revision/4/ complete the section of the table on diffusion.

Help with your assignment • All the notes, videos and links from this lesson will help you to understand about particle theory. • Questions 2 -12 on page 303 of your textbook will also be really helpful. Think about what the answers to these questions are and how you can use these to help you build up your assignment.
- Slides: 20