Particle in a Box 1 The wavefunctions for
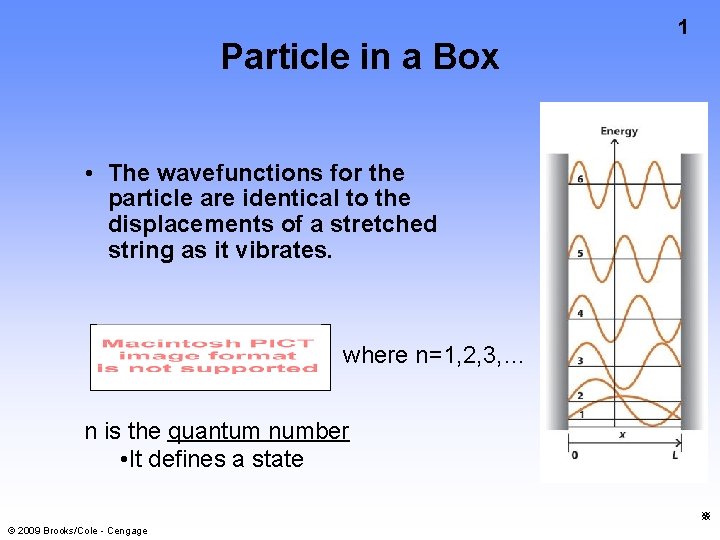
Particle in a Box 1 • The wavefunctions for the particle are identical to the displacements of a stretched string as it vibrates. where n=1, 2, 3, … n is the quantum number • It defines a state © 2009 Brooks/Cole - Cengage

Particle In a Box 2 • Now we know that the allowable energies are : Where n=1, 2, 3, … • This tells us that: 1. The energy levels for heavier particles are less than those of lighter particles. 2. As the length b/w the walls decreases, the ‘distance’ b/w energy levels increases. 3. The energy levels are Quantized. © 2009 Brooks/Cole - Cengage

3 Zero Point Energy • A particle in a container CANNOT have zero energy – A container could be an atom, a box, etc. • The lowest energy (when n=1) is: Zero Point Energy • This is in agreement with the Uncertainty Principle: • ∆p and ∆x are never zero, therefore the particle is always moving © 2009 Brooks/Cole - Cengage

4 Wavefunctions and Probability Densities • • • © 2009 Brooks/Cole - Cengage Examine the 2 lowest energy functions n=1 and n=2 We see from the shading that when n=1, 2 is at a maximum @ the center of the box. When n=2, we see that 2 is at a maximum on either side of the center of the box

5 Wavefunction Summary • The probability density for a particle at a location is proportional to the square of the wavefunction at the point • The wavefunction is found by solving the SchrÖdinger equation for the particle. • When the equation is solved to the appropriate boundary conditions, it is found that the particle can only posses certain discrete energies. • The wavefunctions and their associated energies placed electrons in defined orbitals whose size and shape is determined by the quantum numbers of the particle. © 2009 Brooks/Cole - Cengage
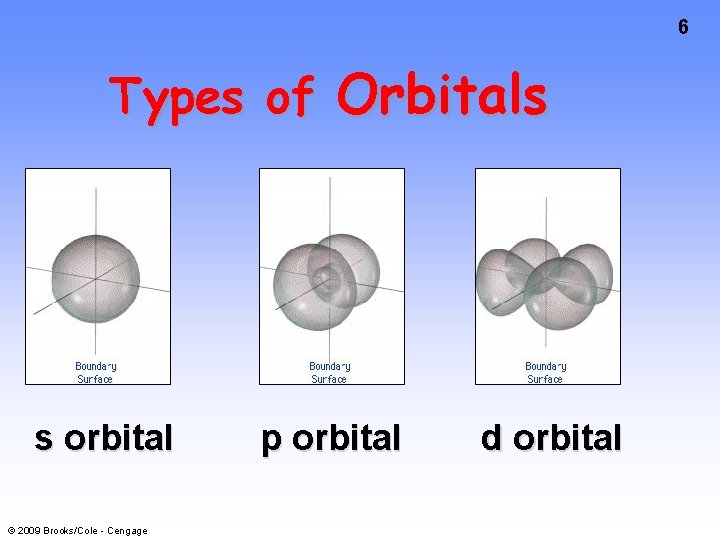
6 Types of Orbitals s orbital © 2009 Brooks/Cole - Cengage p orbital d orbital

Orbitals 7 • No more than 2 e- assigned to an orbital • Orbitals grouped in s, p, d (and f) subshells s orbitals d orbitals © 2009 Brooks/Cole - Cengage p orbitals

8 s orbitals p orbitals d orbitals s orbitals p orbitals No. orbs. 1 3 5 No. e- 2 6 10 © 2009 Brooks/Cole - Cengage d orbitals

9 Subshells & Shells • Subshells grouped in shells. • Each shell has a number called the PRINCIPAL QUANTUM NUMBER, n • The principal quantum number of the shell is the number of the period or row of the periodic table where that shell begins. © 2009 Brooks/Cole - Cengage

10 Subshells & Shells n=1 n=2 n=3 n=4 © 2009 Brooks/Cole - Cengage

QUANTUM NUMBERS The shape, size, and energy of each orbital is a function of 3 quantum numbers: n (major; Principal) l shell Determines the energy and size of the orbital – The bigger the number, the higher the energy and the larger the orbital radius (angular or Azimuthal) subshell l=n– 1 Determines the shape of the orbital ml (magnetic) designates an orbital within a subshell ml = l, l - 1, …, -l © 2009 Brooks/Cole - Cengage 11
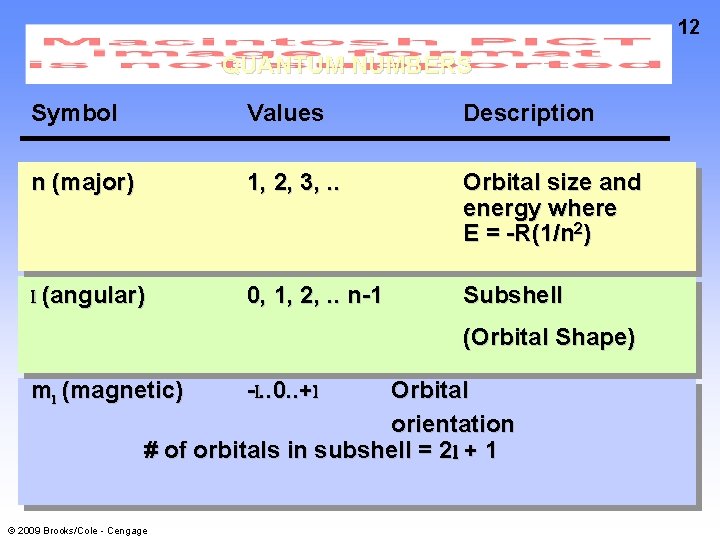
12 QUANTUM NUMBERS Symbol Values Description n (major) 1, 2, 3, . . Orbital size and energy where E = -R(1/n 2) l (angular) 0, 1, 2, . . n-1 Subshell (Orbital Shape) ml (magnetic) Orbital orientation # of orbitals in subshell = 2 l + 1 © 2009 Brooks/Cole - Cengage -l. . 0. . +l

13 © 2009 Brooks/Cole - Cengage
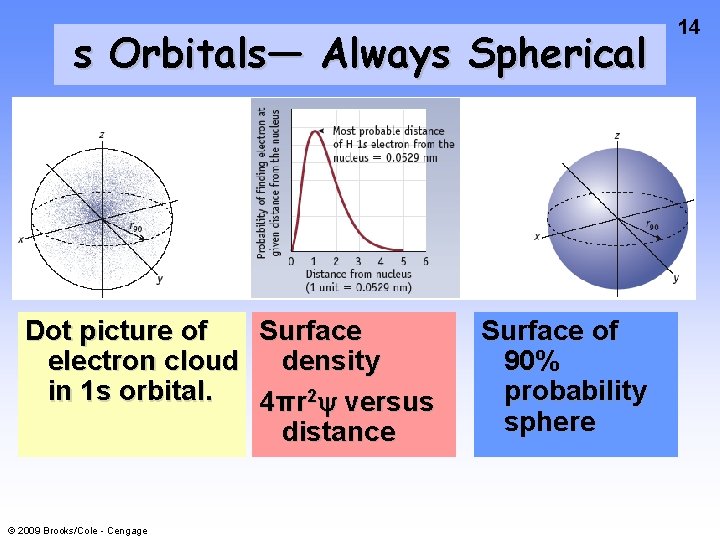
s Orbitals— Always Spherical Dot picture of Surface electron cloud density in 1 s orbital. 4πr 2 y versus distance © 2009 Brooks/Cole - Cengage Surface of 90% probability sphere 14

p Orbitals 15 When n = 2, then l = 0 and 1 Therefore, in n = 2 shell there are 2 types of orbitals — 2 subshells For l = 0 ml = 0 this is a s subshell For l = 1 ml = -1, 0, +1 this is a p subshell with 3 orbitals © 2009 Brooks/Cole - Cengage When s = 1, there is a PLANAR NODE thru the nucleus.
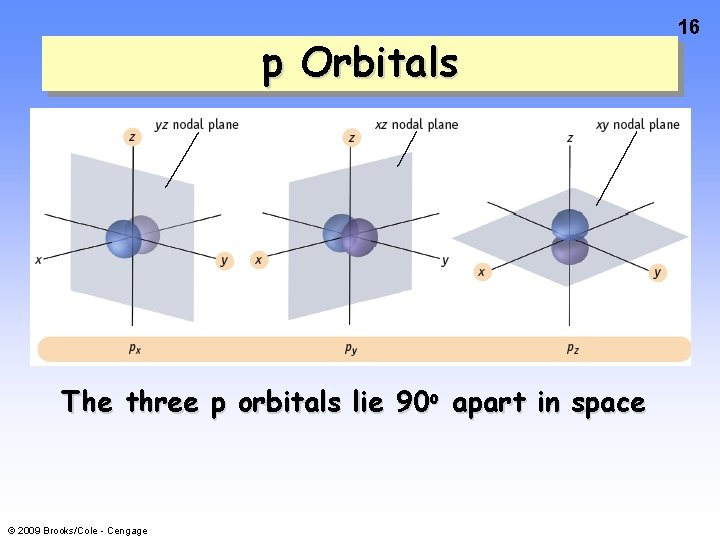
p Orbitals The three p orbitals lie 90 o apart in space © 2009 Brooks/Cole - Cengage 16

d Orbitals When n = 3, what are the values of l? l = 0, 1, 2 and so there are 3 subshells in the shell. For l = 0, ml = 0 s subshell with single orbital For l = 1, ml = -1, 0, +1 p subshell with 3 orbitals For l = 2, ml = -2, -1, 0, +1, +2 d subshell with 5 orbitals © 2009 Brooks/Cole - Cengage 17

d Orbitals s orbitals have no planar node (l = 0) and so are spherical. p orbitals have l = 1, and have 1 planar node, and so are “dumbbell” shaped. This means d orbitals (with l = 2) have 2 planar nodes © 2009 Brooks/Cole - Cengage 18

f Orbitals When n = 4, s = 0, 1, 2, 3 so there are 4 subshells in the shell. For l = 0, ml = 0 s subshell with single orbital For l = 1, ml = -1, 0, +1 p subshell with 3 orbitals For l = 2, ml = -2, -1, 0, +1, +2 d subshell with 5 orbitals For l = 3, ml = -3, -2, -1, 0, +1, +2, +3 f subshell with 7 orbitals © 2009 Brooks/Cole - Cengage 19

Arrangement of Electrons in Atoms Each orbital can be assigned no more than 2 electrons! This is tied to the existence of a 4 th quantum number, the electron spin quantum number, ms. © 2009 Brooks/Cole - Cengage 20

21 Electron Spin Quantum Number, ms Can be proved experimentally that electron has an intrinsic property referred to as “spin. ” Two spin directions are given by ms where ms = +1/2 and -1/2. © 2009 Brooks/Cole - Cengage

Electron Spin and Magnetism • Diamagnetic: NOT attracted to a magnetic field • Paramagnetic: substance is attracted to a magnetic field. • Substances with unpaired electrons are paramagnetic. © 2009 Brooks/Cole - Cengage 22

23 QUANTUM NUMBERS Now there are four! n l shell subshell ml orbital in subshell ms electron spin © 2009 Brooks/Cole - Cengage 1, 2, 3, 4, . . . 0, 1, 2, . . . n - 1 - l. . . 0. . . + l +1/2 and -1/2

24 Pauli Exclusion Principle No two electrons in the same atom can have the same set of 4 quantum numbers. That is, each electron has a unique address. © 2009 Brooks/Cole - Cengage

25 Hund’s Rule • You must add electrons to unoccupied orbitals of a subshell before doubly occupying any of them. • Very critical in determining the filling order for electron shells. © 2009 Brooks/Cole - Cengage

Arrangement of Electrons in Atoms Electrons in atoms are arranged as SHELLS (n) SUBSHELLS (l) ORBITALS (ml) © 2009 Brooks/Cole - Cengage 26

27 © 2009 Brooks/Cole - Cengage

Electrons in Atoms When n = 1, then l = 0 this shell has a single orbital (1 s) to which 2 ecan be assigned. When n = 2, then l = 0, 1 and ml = -1, 0, +1 2 s orbital 2 e- three 2 p orbitals 6 e- TOTAL = 8 e- © 2009 Brooks/Cole - Cengage 28

Electrons in Atoms When n = 3, then l = 0, 1, 2 and ml = -2, -1, 0, +1, +2 3 s orbital three 3 p orbitals five 3 d orbitals TOTAL = © 2009 Brooks/Cole - Cengage 2 e 6 e 10 e 18 e- 29

Electrons in Atoms When n = 4, then l = 0, 1, 2, 3 and ml = -3, -2, -1, 0, +1, +2, +3 4 s orbital 2 e- three 4 p orbitals five 4 d orbitals seven 4 f orbitals TOTAL = 6 e 10 e 14 e 32 e- © 2009 Brooks/Cole - Cengage 30

31 © 2009 Brooks/Cole - Cengage

32 Assigning Electrons to Atoms • Electrons generally assigned to orbitals of successively higher energy. • For H atoms, E = - C(1/n 2). E depends only on n. • For many-electron atoms, energy depends on both n and l. © 2009 Brooks/Cole - Cengage

33 Electron Filling Order Print this chart out and use it at home! © 2009 Brooks/Cole - Cengage

Electron Filling Order © 2009 Brooks/Cole - Cengage 34

Effective Nuclear Charge, Zeff • Zeff is the nuclear charge experienced by the outermost electrons. • Explains why E(2 s) < E(2 p) • Zeff increases across a period owing to incomplete shielding by inner electrons. • Estimate Zeff = [ Z - (no. inner electrons) ] • Charge felt by 2 s e- in Li Zeff = 3 - 2 = 1 • Be Zeff = 4 - 2 = 2 • B Zeff = 5 - 2 = 3 and so on! © 2009 Brooks/Cole - Cengage 35

36 Effective Nuclear Charge Electron cloud for 1 s electrons © 2009 Brooks/Cole - Cengage Zeff is the nuclear charge experienced by the outermost electrons. This will help explain some of the periodic properties we’ll examine later this chapter.
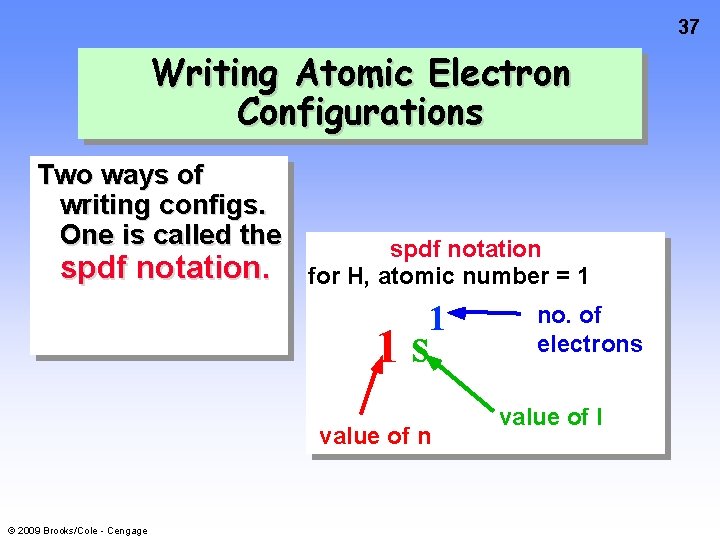
37 Writing Atomic Electron Configurations Two ways of writing configs. One is called the spdf notation for H, atomic number = 1 1 1 s value of n © 2009 Brooks/Cole - Cengage no. of electrons value of l

Writing Atomic Electron Configurations Two ways of writing configs. Other is called the orbital box notation. One electron has n = 1, s = 0, ms = + 1/2 Other electron has n = 1, s = 0, ms = - 1/2 © 2009 Brooks/Cole - Cengage 38

39 © 2009 Brooks/Cole - Cengage

Electron Configurations and the Periodic Table 40 See Active Figure 7. 4 © 2009 Brooks/Cole - Cengage

Lithium Group 1 A Atomic number = 3 1 s 22 s 1 f 3 total electrons © 2009 Brooks/Cole - Cengage 41

42 Beryllium Group 2 A Atomic number = 4 1 s 22 s 2 f 4 total electrons © 2009 Brooks/Cole - Cengage

Boron Group 3 A Atomic number = 5 1 s 2 2 p 1 f 5 total electrons © 2009 Brooks/Cole - Cengage 43

Carbon Group 4 A Atomic number = 6 1 s 2 2 p 2 f 6 total electrons Here we see for the first time HUND’S RULE. When placing electrons in a set of orbitals having the same energy, we place them singly as long as possible. © 2009 Brooks/Cole - Cengage 44

45 Nitrogen Group 5 A Atomic number = 7 1 s 2 2 p 3 f 7 total electrons © 2009 Brooks/Cole - Cengage

46 Oxygen Group 6 A Atomic number = 8 1 s 2 2 p 4 f 8 total electrons © 2009 Brooks/Cole - Cengage

47 Fluorine Group 7 A Atomic number = 9 1 s 2 2 p 5 f 9 total electrons © 2009 Brooks/Cole - Cengage

48 Neon Group 8 A Atomic number = 10 1 s 2 2 p 6 f 10 total electrons Note that we have reached the end of the 2 nd period, and the 2 nd shell is full! © 2009 Brooks/Cole - Cengage

49 Electron Configurations of p -Block Elements PLAY MOVIE © 2009 Brooks/Cole - Cengage

Sodium Group 1 A Atomic number = 11 1 s 2 2 p 6 3 s 1 or “neon core” + 3 s 1 [Ne] 3 s 1 (uses rare gas notation) Note that we have begun a new period. All Group 1 A elements have [core]ns 1 configurations. © 2009 Brooks/Cole - Cengage 50

Aluminum Group 3 A Atomic number = 13 1 s 2 2 p 6 3 s 2 3 p 1 [Ne] 3 s 2 3 p 1 All Group 3 A elements have [core] ns 2 np 1 configurations where n is the period number. © 2009 Brooks/Cole - Cengage 51

Phosphorus Group 5 A Atomic number = 15 1 s 2 2 p 6 3 s 2 3 p 3 [Ne] 3 s 2 3 p 3 All Group 5 A elements have [core ] ns 2 np 3 configurations where n is the period number. © 2009 Brooks/Cole - Cengage 52 Yellow P Red P

53 Calcium Group 2 A Atomic number = 20 1 s 2 2 p 6 3 s 2 3 p 6 4 s 2 [Ar] 4 s 2 All Group 2 A elements have [core]ns 2 configurations where n is the period number. © 2009 Brooks/Cole - Cengage

Electron Configurations and the Periodic Table © 2009 Brooks/Cole - Cengage 54
![55 Transition Metals All 4 th period elements have the configuration [argon] nsx (n 55 Transition Metals All 4 th period elements have the configuration [argon] nsx (n](http://slidetodoc.com/presentation_image_h/bf5682d07593669d8652a954b91423b9/image-55.jpg)
55 Transition Metals All 4 th period elements have the configuration [argon] nsx (n - 1)dy and so are d-block elements. Chromium © 2009 Brooks/Cole - Cengage Iron Copper

Transition Element Configurations 3 d orbitals used for Sc-Zn © 2009 Brooks/Cole - Cengage 56

57 © 2009 Brooks/Cole - Cengage
![Lanthanides and Actinides All these elements have the configuration [core] nsx (n - 1)dy Lanthanides and Actinides All these elements have the configuration [core] nsx (n - 1)dy](http://slidetodoc.com/presentation_image_h/bf5682d07593669d8652a954b91423b9/image-58.jpg)
Lanthanides and Actinides All these elements have the configuration [core] nsx (n - 1)dy (n - 2)fz and so are f-block elements. Cerium [Xe] 6 s 2 5 d 1 4 f 1 Uranium [Rn] 7 s 2 6 d 1 5 f 3 © 2009 Brooks/Cole - Cengage 58

Lanthanide Element Configurations 4 f orbitals used for Ce - Lu and 5 f for Th - Lr © 2009 Brooks/Cole - Cengage 59
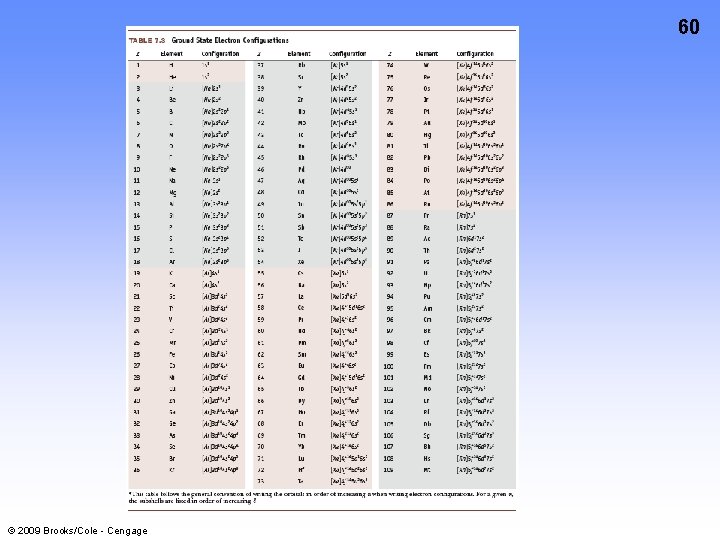
60 © 2009 Brooks/Cole - Cengage

Ion Configurations To form cations from elements remove 1 or more e- from subshell of highest n [or highest (n + l)]. P [Ne] 3 s 2 3 p 3 - 3 e- f P 3+ [Ne] 3 s 2 3 p 0 © 2009 Brooks/Cole - Cengage 61

Ion Configurations For transition metals, remove ns electrons and then (n - 1) electrons. Fe [Ar] 4 s 2 3 d 6 loses 2 electrons Fe 2+ [Ar] 4 s 0 3 d 6 To form cations, always remove electrons of highest n value first! © 2009 Brooks/Cole - Cengage 62
- Slides: 62