PARTIAL ORAL VS INTRAVENOUS ANTIBIOTIC TREATMENT OF ENDOCARDITIS

PARTIAL ORAL VS INTRAVENOUS ANTIBIOTIC TREATMENT OF ENDOCARDITIS NEJM AUGUST 28, 2018

INFECTIVE ENDOCARDITIS • Deadly disease associated with high morbidity and mortality. • Has a diverse clinical history and symptoms. • Should be suspected in a variety of different clinical situations. • • Can present as an acute, rapidly progressive infection Or a subacute or chronic illness with low-grade fever and non-specific symptoms that may mislead or confuse initial assessment.

INFECTIVE ENDOCARDITIS • 90% present with fever, associated with chills, poor appetite and weight loss. • 85% have heart murmurs. • 25% of patients have embolic complications at the time of diagnosis. • Imaging, particularly echocardiography, plays a key role in both the diagnosis and management of IE. • Other modalities are also useful: MSCT, MRI, FDG-PET CT.

DUKE CRITERIA

PREDICTORS OF POOR OUTCOME

TREATMENT • Successful treatment relies on microbial eradiation by antimicrobial drugs. • Surgery contributes by removing infected material and draining abscesses. • Host defenses are of little help. • Drug treatment of PVE should last longer (at least 6 weeks) than that of NVE (2 -6 weeks).

OUTPATIENT PARENTERAL ANTIBIOTIC THERAPY

MAIN COMPLICATIONS OF LEFT SIDED ENDOCARDITIS • Heart failure • • Most frequent complication and most common indication for surgery. • Valvular regurgitation may occur as a results of mitral chordal rupture, leaflet perforation, or interference of the vegetation mass with leaflet closure. Observed in 42 -60% of cases of NVE More often present when AV is affected compared to the MV. Mainly caused by new or worsening severe AR or MR, although intracardiac fistulae and valve obstruction could also be implicated.

MAIN COMPLICATIONS OF LEFT SIDED ENDOCARDITIS • Uncontrolled infection • • • More feared complication • Persisting infection: fever and persisting positive cultures after 7 -10 days of antibiotic treatment. Second most frequent cause for surgery Considered to be present when there is persisting infection and when there are signs of locally uncontrolled infection. • May be related to inadequate Abx therapy, resistant organisms, infected lines, locally uncontrolled infection, embolic complications or extracardiac sites of infection, and adverse reaction to antibiotics.

MAIN COMPLICATIONS OF LEFT SIDED ENDOCARDITIS • Perivalvular extension • • • Most frequent cause of uncontrolled infection Associated with a poor prognosis and high likelihood of the need for surgery. Complications include abscess formation, pseudoaneurysm, fistulae, VSD, CHB, ACS. High rates of surgery in this population (87%); hospital mortality remains high (41%). Perivalvular abscess • • More common in aortic IE (10 -40%in NVE), and frequent in PVE (56 -100%). Abscess formation is a dynamic process: • • Starting with root wall thickening. Extending to the development of fistulae.

MAIN COMPLICATIONS OF LEFT SIDED ENDOCARDITIS • Systemic embolization • • Frequent and life threatening Brain and spleen are the most frequent sites of embolism in left sided IE. • • Embolic events occur in 20 -50% of patients. • • PE in right sided IE and pacemaker lead IE. Risk of new events after initiation of antibiotics is only ~6 -21%. Factors associated with increased risk of embolization: • • Size and mobility of vegetations Increasing of decreasing size of the vegetation under antibiotic therapy Particular organisms (S. aureus, S. bovis, Candida spp) Previous emboli and multivalvular IE
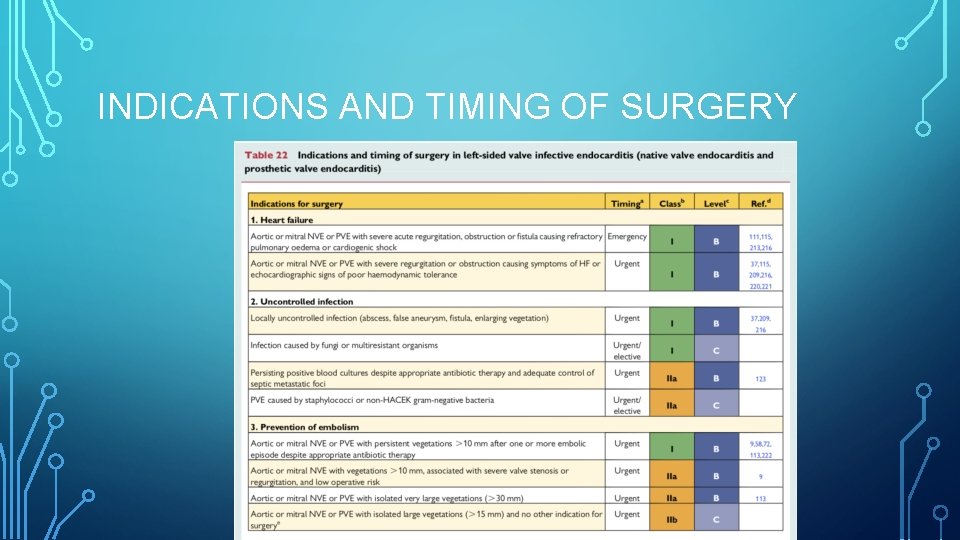
INDICATIONS AND TIMING OF SURGERY

RIGHT SIDED IE • 5 -10% of all IE cases. • Most frequent in IVDUs (esp if coinfected with HIV), though may occur in patients with pacemakers, ICD, CVC, or CHD. • 60 -90% of cases involve staph aureus. • Tricuspid valve is most frequently affected. • In hospital mortality is ~7%.

PROGNOSIS AND TREATMENT OF RIGHT SIDED IE • Predictors of death: • Vegetation length of >20 mm • Fungal aetiology • Treatment: • 2 -week treatment may be sufficient to treat right sided IE, if the following criteria are met: • • • MSSA Good response to treatment Absence of metastatic sites of infection or empyema AND absence of cardiac and extracardiac complications. Absence of associated prosthetic valve or left sided valve infection. <20 mm vegetation Absence of severe immunosuppression.

INDICATIONS FOR SURGICAL TREATMENT OF RIGHT SIDED IE

INTRODUCTION • Patients are typically treated with IV antibiotics for up to 6 weeks, per ESC and AHA guidelines. • In-hospital mortality is reported to range from 15% to more than 45%, depending on the pathogen and on complicating factors. • Approximately half the patients undergo cardiac-valve surgery. • Majority of complications are seen during the initial phase. • For a large proportion of patients, the main reason for staying in hospital after the initial phase is to complete intravenous antibiotic treatment.

INTRODUCTION • Therefore, if oral antibiotic treatment might be safe and efficient, part of the treatment period for patients in stable condition could take place outside hospital, without the need for an intravenous catheter. • Intravenous treatment during long hospital stays may be associated with an increased risk of complications • Shorter length of stay has been associated with better outcomes in studies of other diseases. • Forms the basis for the guidelines recommending outpatient parenteral treatment of endocarditis of patients fulfilling certain criteria.

INTRODUCTION • However, when outpatient parenteral treatment is given, logistical issues are critical. • • Staff and patients need to be educated to ensure adherence to the regimen. Patients need to be adequately monitored for efficacy and adverse effects. • Oral antibiotic therapy may reduce these challenges and may be an appropriate alternative. • However, clinical evidence for the safety and efficacy of oral antibiotic treatment of endocarditis is limited.

HYPOTHESIS • In patients in clinically stable condition who have endocarditis on the left side of the heart, a shift from IV to orally administered antibiotic treatment would result in efficacy and safety that would be similar to those with continued intravenous antibiotic treatment.

PARTIAL ORAL TREATMENT OF ENDOCARDITIS (POET) • Nationwide, investigator-initiated, multicenter, randomized, unblended, noninferiority trial. • Performed at cardiac centers in Denmark.

INCLUSION CRITERIA • Left sided endocarditis based on the Duke criteria • Infected with one of the following organisms: • Streptococci • Enterococcus faecalis • Staphylococcus aureus • Coagulase negative staphylococci • • >18 years old >10 days of appropriate parenteral antibiotic treatment overall, and at least 1 week of appropriate Abx therapy after valve-surgery. • • T<38. 0˚C >2 days • No sign of abscess formation revealed by echo • TTE and TOE performed within 48 hours of randomization. CRP less than 25% of peak value, or <20 mg/d. L and WCC <15 during Abx treatment.

EXCLUSION CRITERIA • BMI >40. • Concomitant infection requiring IV antibiotics. • Inability to give informed consent. • Suspicion of reduced absorption of PO treatment due to abdominal disorder. • Reduced compliance.

• • At least 10 days of scheduled antibiotic treatment had to remain. • Patients assigned to PO treatment were treated in the outpatient clinics • Were seen 2 -3 times per week. • Within 1 to 3 days before completion of the assigned Abx treatment, TOE was performed to confirm that the patient had a sufficient treatment response. • All patients were discharged from hospital the day the antibiotic treatment was terminated. • All were seen in outpatient clinic at 1 week, and at 1, 3 and 6 months after completion of antibiotic Patients assigned to receive intravenous treatment remained in the hospital until antibiotic treatment was completed. treatment.

CHOICE OF ANTIBIOTICS • IV abx were administered in accordance with the ESC guidelines, with modifications endorsed by the Danish Society of Cardiology. • Trial investigators developed oral antibiotic treatment regimens. • PO Abx used, in different combinations, were amoxicillin, fusidic acid, rifampicin, linezolid, dicloxacillin, moxifloxacin, and clindamycin. • • Had published data showing moderate to high bioavailability. Regimens were based on pharmacokinetic calculations and expected MICs for each bacterial species.

PHARMACOKINETICS • To ensure that patients received sufficient doses of antibiotics, blood samples for the measurement of plasma levels of orally administered antibiotics were obtained on • day 1 after the administration of a single dose. • At 30 minutes, 1, 2, 4, and 6 hours after administration • And day 5, after the administration of multiple doses. • With the assumption that steady state would have been achieved by this time. • Samples were also obtained from patients in the intravenously treated group on day 1.

TRIAL PROCEDURES • Patients were assigned in a 1: 1 ratio to continued IV administered antibiotic treatment or to a shift to orally administered antibiotic treatment.

OUTCOMES • Primary Outcome: • Composite of all-cause mortality, unplanned cardiac surgery, clinically evident embolic events, or relapse of bacteremia with the primary pathogen from randomization through 6 months after antibiotic treatment was completed. • Adjudicated by a committee consisting of cardiologists and an ID specialist.
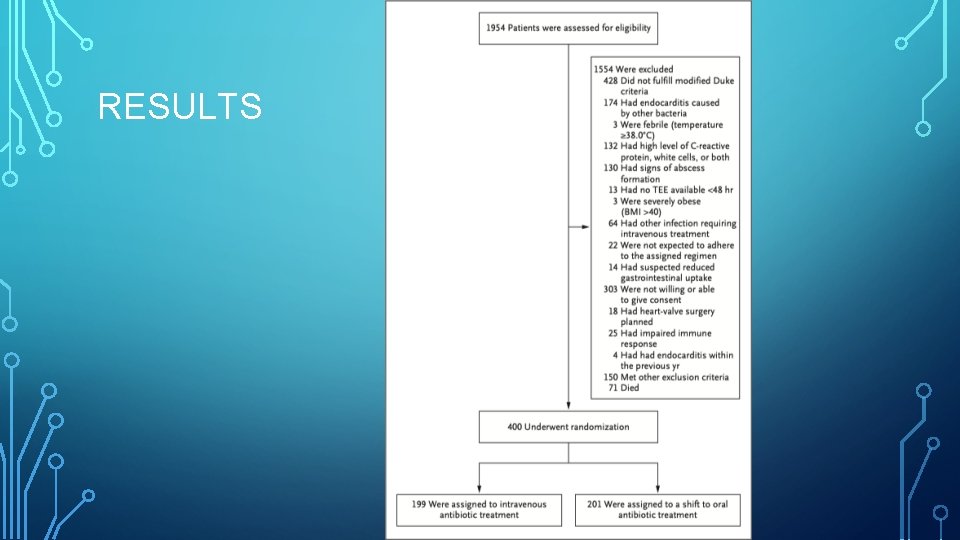
RESULTS
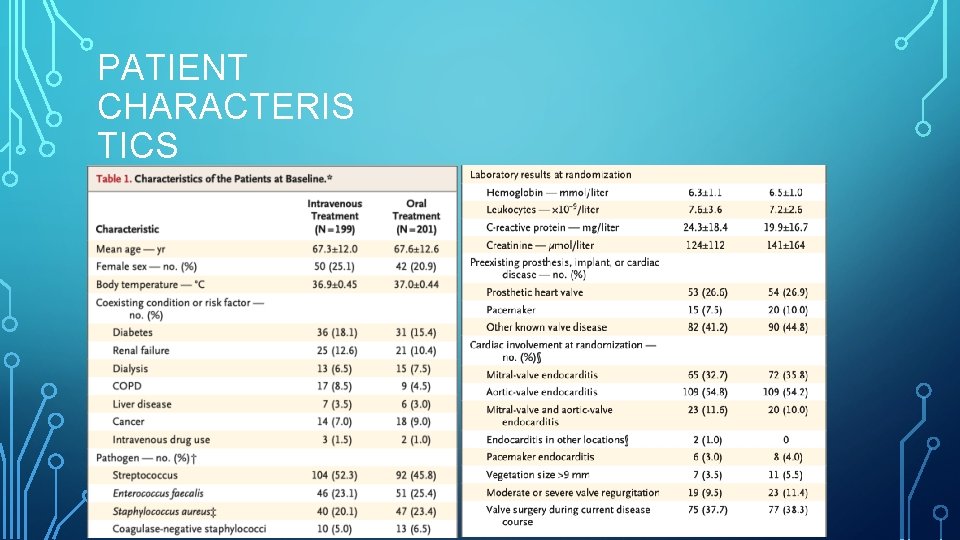
PATIENT CHARACTERIS TICS

TIMING OF RANDOMIZATION AND LOS IN HOSPITAL • Median time from diagnosis of endocarditis of the left side of the heart to randomization: • • 17 days in both groups. After randomization, patients were treated for a median of: • • 19 days in the IV group 17 days in the PO group. • In the PO Abx group, 160 patients (80%) were partially or completely treated as outpatients. • Median length of stay after randomization was • • 19 days in the IV group 3 days in the PO group.

ANTIBIOTIC TREATMENT • S. aureus • Dicloxacillin and rifampicin: 15 (33%) • Amoxicillin and rifampicin: 13 (29%) • E. faecalis • Amoxicillin and moxifloxacin: 24 (47%) • Amoxicillin and linezolid: 13 (25%) • Streptococci • Amoxicillin and rifampicin: 47 (52%) • Amoxicillin and moxifloxacin: 12 (13%) • Coagulase negative • Fusidic acid and linezolid: 5 (38%) • Rifampicin and linezolid: 4 (31%)

ANTIBIOTIC TREATMENT • 4 patients crossed over from the orally treated group to the IV group. • • • 1 because of nausea 1 because of a new incident of bacteremia with a different pathogen 2 because of patient preference • No crossover from the IV group to the PO group. • From time of randomization until completion of Abx therapy • • 43 patients (22%) in the IV group switched to a different IV Abx regimen 24 patients (12%) in the PO treated group were switched to a different PO regimen.

PRIMARY OUTCOME • All patients were followed for 6 months after the antibiotic treatment was completed or until death. • • No patients lost to follow up. Primary composite outcome occurred in: • • • 42 patients (10. 5%) in total. 24 patients (12. 1%) out of 199 patients in the IV treated group. 18 patients (9%) out of 197 patients in the PO treated group.

PRIMARY OUTCOME • Between group difference was 3. 1% in favour of PO treatment • Criterion for non-inferiority was therefore met. • In a sensitivity analysis in which the 4 patients who crossed over from PO to IV were considered to have treatment failure, the criterion for non-inferiority was still met. • Primary outcome occurred in 24 out of 199 (12. 1%) in the IV group and 22 out of 201 (10. 9%) in the PO group.
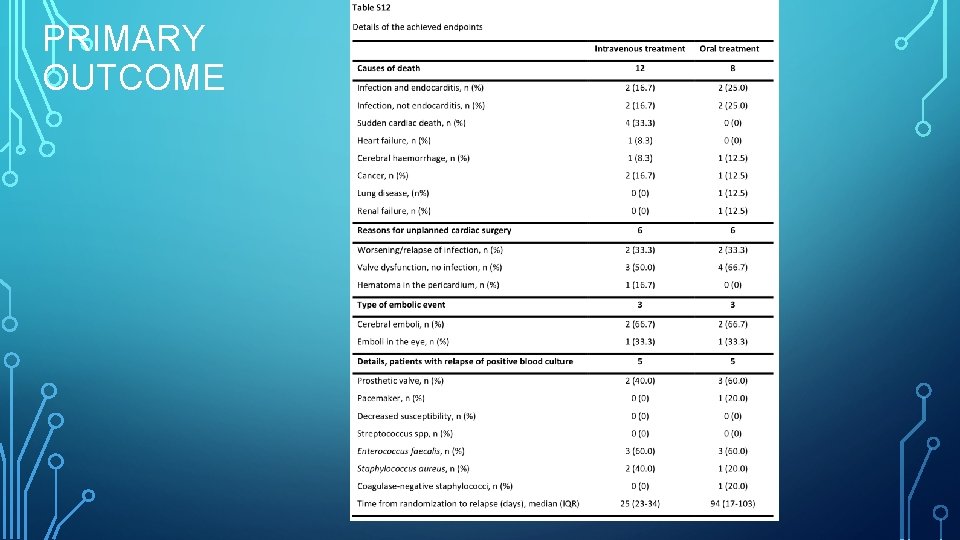
PRIMARY OUTCOME

SAFETY • In 7 patients in the PO group • Plasma concentrations of one of the two Abx was not at the most effective level. • • Rifampicin in 3, moxifloxacin in 2, linezolid in 1, and dicloxacillin in 1. The primary outcome did not occur in any of these patients. • Adverse effects were reported in 22 patients (6%) after randomization. • • 12 patients (6%) in the IV group. 10 patients (5%) in the PO group.

SAFETY • Adverse effects: • • • Allergy (50%) Bone marrow suppression (27%) GI effects (14%)

DISCUSSION • A shift from initial IV abx to PO abx was non-inferior when compared to continuous IV treatment • In patients who had • • Endocarditis of the left side of the heart. Caused by strep, E. faecalis, S. aureus, or coag-neg staph. Clinically stable. Adequate response to initial treatment.

DISCUSSION • The patients were shifted from IV to PO on about day 17, the midpoint of the treatment period. • Results seemed consistent across prespecified subgroups, including • • Type of valve affected (native vs prosthetic) Type of treatment (surgery vs conservative) • Primary outcome was similar across the four different bacterial types.

DISCUSSION • Rationale for this trial was that in patients with normal GI function, the uptake of orally administered Abx may allow sufficient plasma concentrations of antibiotics to achieve bacterial killing. • All oral regimens included two antibiotics from different drug classes and with different antibacterial effects and metabolisation processes • In order to avoid the risk of subtherapeutic antibiotic levels due to potentially reduced GI uptake as well as the risks of variations in pharmacokinetics of the orally administered Abx.

• Recommendations for the duration of antibiotic therapy and for in-hospital IV administration in patients with endocarditis are based mainly on observational studies. • Longer hospital stays may be a psychological and physical burden. • • Shorter stays are associated with better outcomes in studies in other diseases, and may reduce costs. Oral Abx therapy also minimizes the challenges associated with outpatient parenteral treatment • • • Logistics Monitoring Risks of complications associated with IV catheters (bleeding, local and systemic infection, venous thrombosis, etc).

• Several observational studies and a systematic review have addressed the safety and efficacy of a shift from IV to PO therapy in the treatment of endocarditis. • Generally, partial oral treatment has been shown to have an acceptable cure rate in selected cases of endocarditis on the right side of the heart. • Literature on endocarditis on the left side of the heart is sparse.

LIMITATIONS • Only patients with endocarditis of the left side of the heart were enrolled. • However, patients with simultaneous infection of a cardiovascular implantable electronic device or endocarditis of the right side of the heart were not excluded. • Only patients with endocarditis caused by certain bacteria species were eligible. • • Results may not apply to patients who have endocarditis caused by other organisms. Only 5 IVDU patients were referred Only 22% of the patients enrolled had S. aureus No patients had MRSA or other antibiotic-resistant phenotypes

LIMITATIONS • Potential referral bias: • Elderly patients who were fragile and have serious coexisting conditions may not have been referred to one of the participating centers. • Criteria for inclusion in this trial was strict. • Clinicians should use these criteria in the decision to shift a patient form IV to PO therapy. • These criteria could also be used in areas with higher rates of antibiotic resistance, as they are based on antibiotic treatment guided by state-of-the-art susceptibility testing.

• Discharge of patients who were receiving oral treatment to outpatient treatment was not mandatory. • Was decided according to the patient’s preference and the discretion of the treating physician. • Therefore duration of outpatient treatment may have been underestimated. • Only 20% of the screened population underwent randomization.

CONCLUSION • In patients who had endocarditis on the left side of the heart caused by streptococcus, E. faecalis, S. aureus, or coagulase negative staphylococci, and who were in a stable condition, a shift from intravenously administered to orally administered antibiotic treatment was non-inferior to continued intravenous antibiotic treatment.
- Slides: 46