Partial melting 1 Binary and ternary phase diagrams

Partial melting 1. Binary and ternary phase diagrams; melting of the mantle
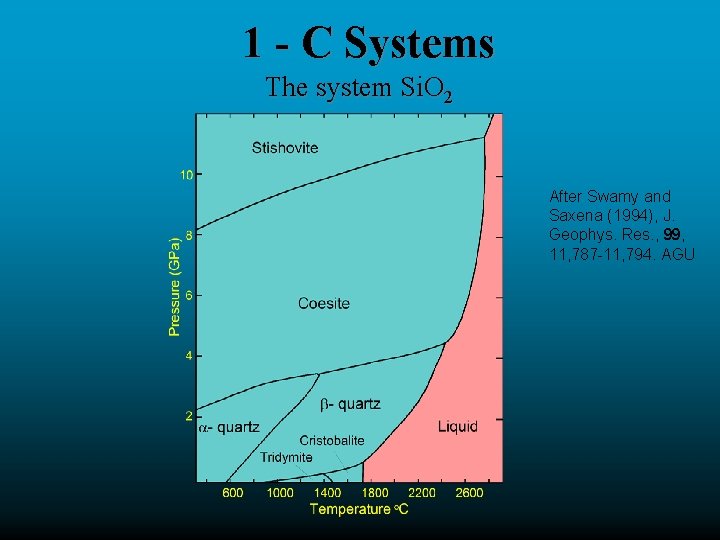
1 - C Systems The system Si. O 2 After Swamy and Saxena (1994), J. Geophys. Res. , 99, 11, 787 -11, 794. AGU
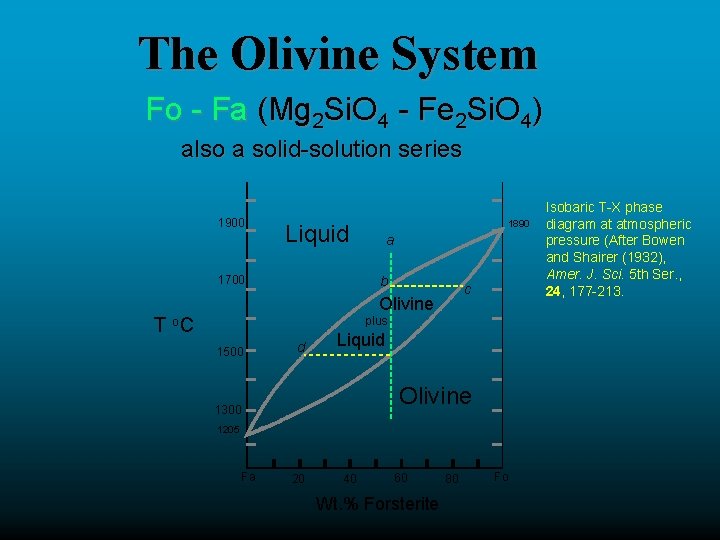
The Olivine System Fo - Fa (Mg 2 Si. O 4 - Fe 2 Si. O 4) also a solid-solution series 1900 1890 Liquid 1700 a b c Olivine T o. C plus 1500 d Liquid Olivine 1300 1205 Fa 20 40 60 Wt. % Forsterite 80 Fo Isobaric T-X phase diagram at atmospheric pressure (After Bowen and Shairer (1932), Amer. J. Sci. 5 th Ser. , 24, 177 -213.

2 -C Eutectic Systems Example: Diopside - Anorthite No solid solution 1600 1553 idus Liquid Liqu 1500 T o. C 1400 Anorthite + Liquid 1392 1300 Diopside + Liquid 1200 1274 Diopside + Anorthite Di 20 40 60 Wt. % Anorthite 80 An Isobaric T-X phase diagram at atmospheric pressure (After Bowen (1915), Amer. J. Sci. 40, 161 -185.
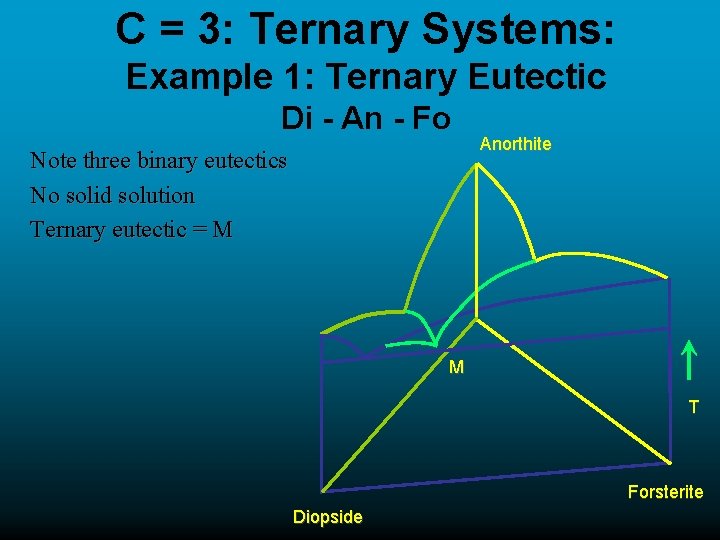
C = 3: Ternary Systems: Example 1: Ternary Eutectic Di - An - Fo Note three binary eutectics No solid solution Ternary eutectic = M Anorthite M T Forsterite Diopside
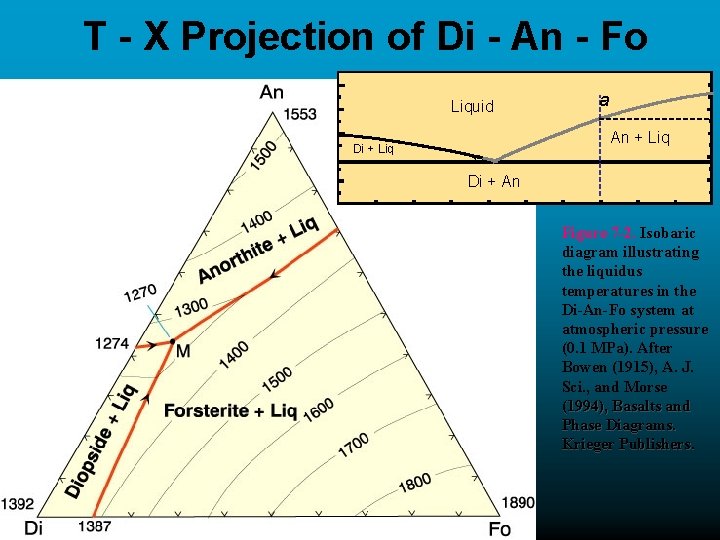
T - X Projection of Di - An - Fo Liquid a An + Liq Di + An Figure 7 -2. Isobaric diagram illustrating the liquidus temperatures in the Di-An-Fo system at atmospheric pressure (0. 1 MPa). After Bowen (1915), A. J. Sci. , and Morse (1994), Basalts and Phase Diagrams. Krieger Publishers. A
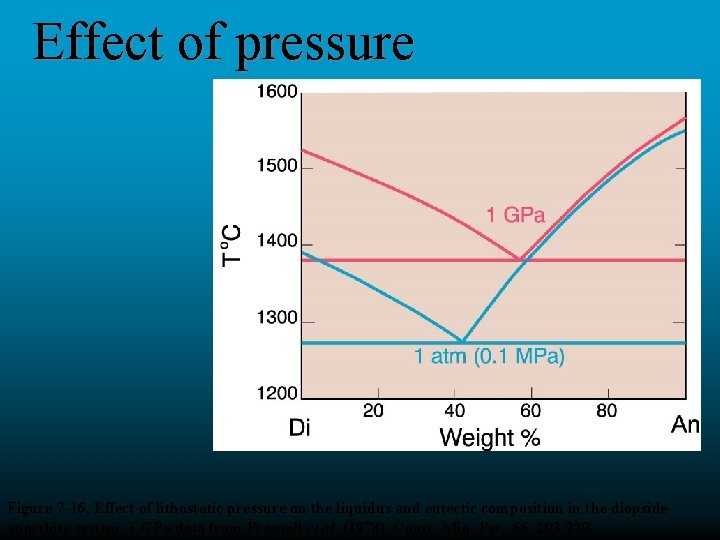
Effect of pressure Figure 7 -16. Effect of lithostatic pressure on the liquidus and eutectic composition in the diopsideanorthite system. 1 GPa data from Presnall et al. (1978). Contr. Min. Pet. , 66, 203 -220.
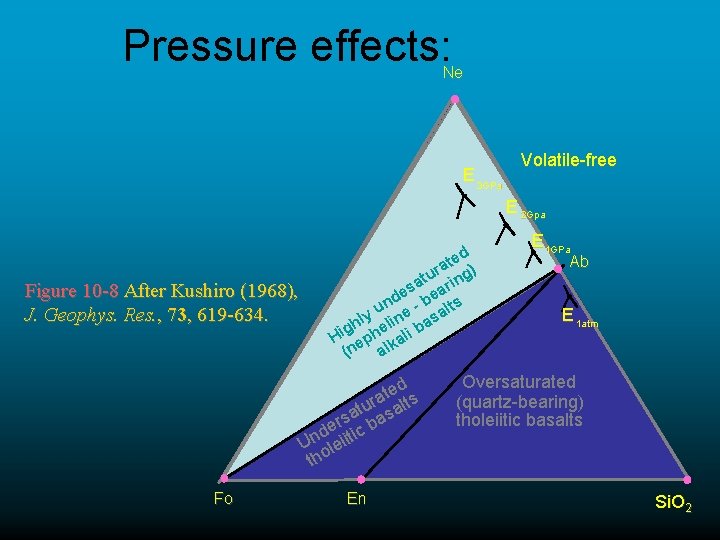
Pressure effects: Ne E 3 GPa Volatile-free E 2 Gpa Figure 10 -8 After Kushiro (1968), J. Geophys. Res. , 73, 619 -634. d e t ra g) u t sa earin e nd - b lts u ly line asa h b e g Hi eph kali (n al d e t ra alts u t rsa bas e d ic Un leiit tho Fo En E 1 GPa Ab E 1 atm Oversaturated (quartz-bearing) tholeiitic basalts Si. O 2
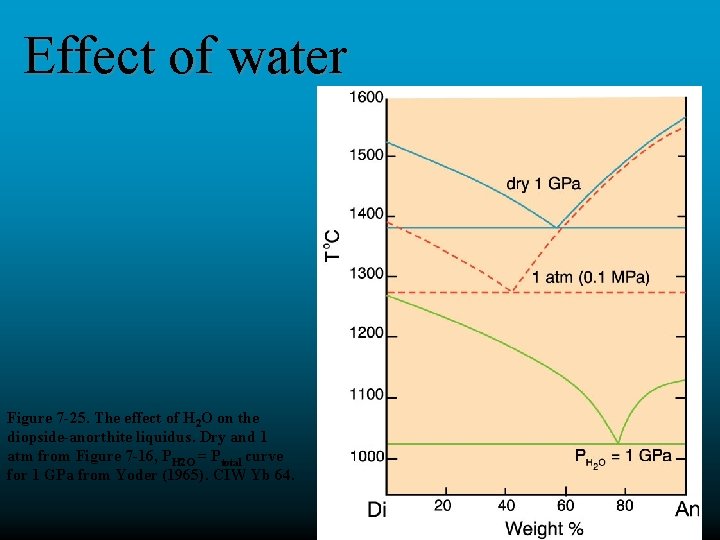
Effect of water Figure 7 -25. The effect of H 2 O on the diopside-anorthite liquidus. Dry and 1 atm from Figure 7 -16, PH 2 O = Ptotal curve for 1 GPa from Yoder (1965). CIW Yb 64.
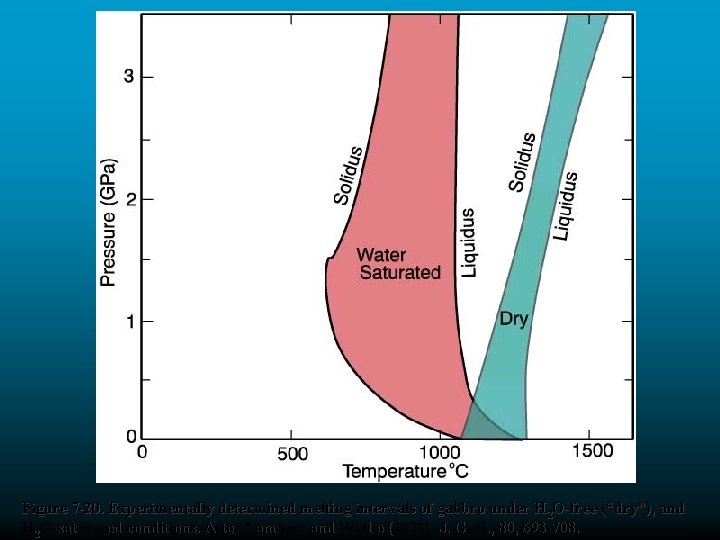
Figure 7 -20. Experimentally determined melting intervals of gabbro under H 2 O-free (“dry”), and H 2 O-saturated conditions. After Lambert and Wyllie (1972). J. Geol. , 80, 693 -708.
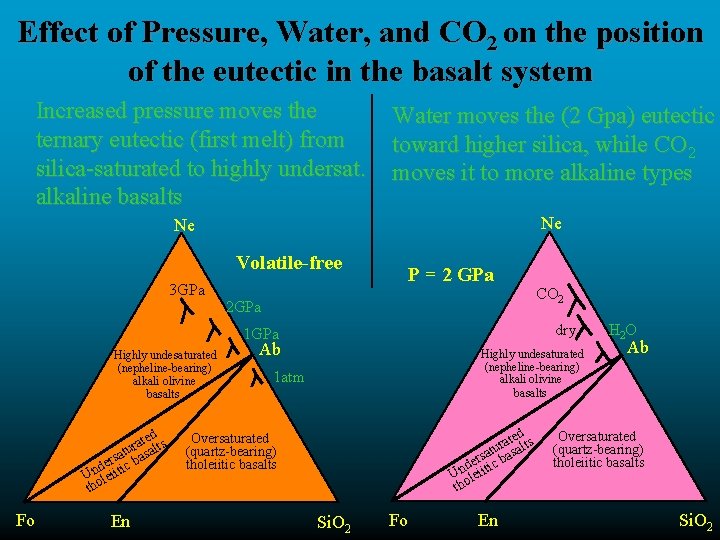
Effect of Pressure, Water, and CO 2 on the position of the eutectic in the basalt system Increased pressure moves the ternary eutectic (first melt) from silica-saturated to highly undersat. alkaline basalts Water moves the (2 Gpa) eutectic toward higher silica, while CO 2 moves it to more alkaline types Ne Ne Volatile-free 3 GPa P = 2 GPa 2 GPa dry 1 GPa Highly undesaturated (nepheline-bearing) alkali olivine basalts d ate lts r u a at s erstic ba d Unoleii th Fo En CO 2 Ab Highly undesaturated (nepheline-bearing) alkali olivine basalts 1 atm ted ts a r atu basal s r de ic Unoleiit th Oversaturated (quartz-bearing) tholeiitic basalts Si. O 2 Fo En H 2 O Ab Oversaturated (quartz-bearing) tholeiitic basalts Si. O 2
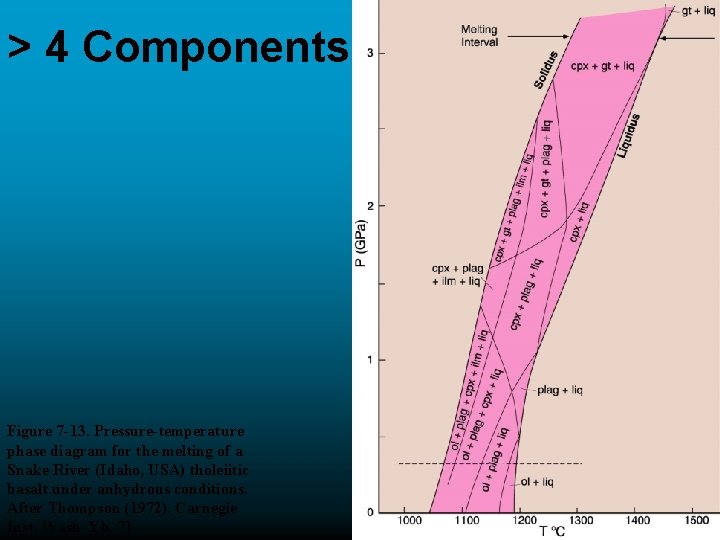
> 4 Components Figure 7 -13. Pressure-temperature phase diagram for the melting of a Snake River (Idaho, USA) tholeiitic basalt under anhydrous conditions. After Thompson (1972). Carnegie Inst. Wash Yb. 71
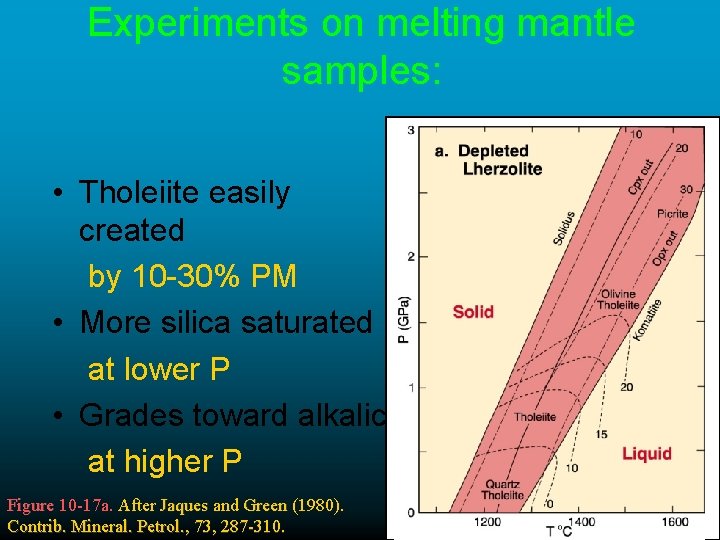
Experiments on melting mantle samples: • Tholeiite easily created by 10 -30% PM • More silica saturated at lower P • Grades toward alkalic at higher P Figure 10 -17 a. After Jaques and Green (1980). Contrib. Mineral. Petrol. , 73, 287 -310.
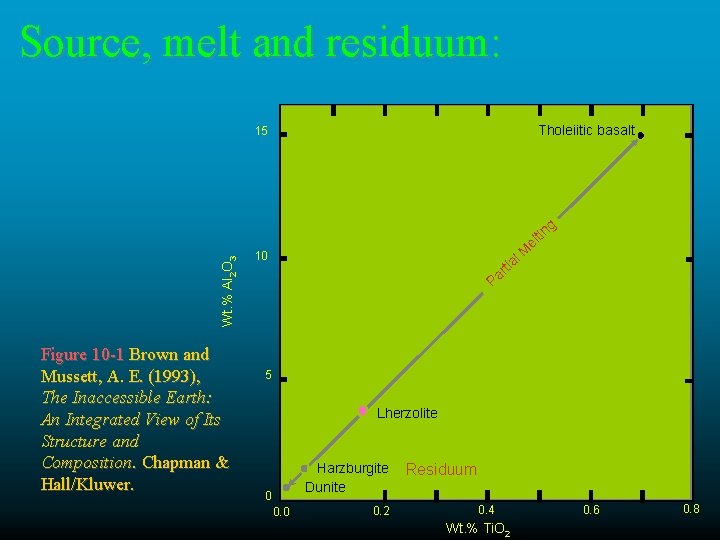
Source, melt and residuum: Tholeiitic basalt 15 Wt. % Al 2 O 3 g Figure 10 -1 Brown and Mussett, A. E. (1993), The Inaccessible Earth: An Integrated View of Its Structure and Composition. Chapman & Hall/Kluwer. l. M tr ia Pa 10 tin el 5 Lherzolite Harzburgite Dunite 0 0. 2 Residuum 0. 4 Wt. % Ti. O 2 0. 6 0. 8
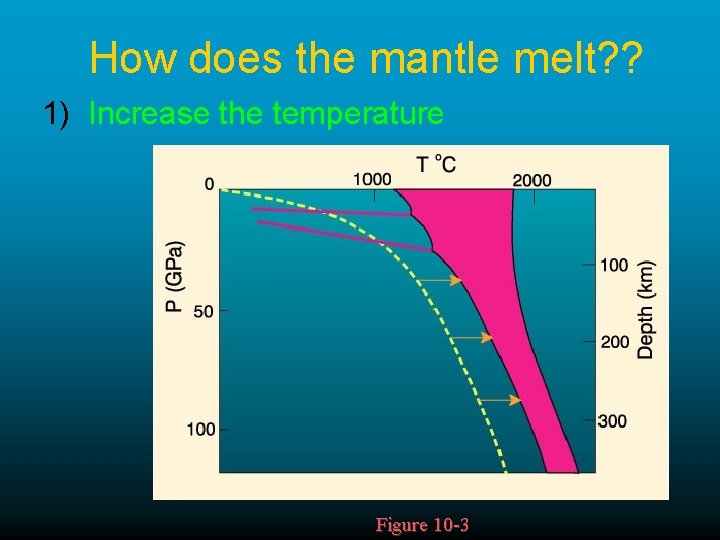
How does the mantle melt? ? 1) Increase the temperature Figure 10 -3
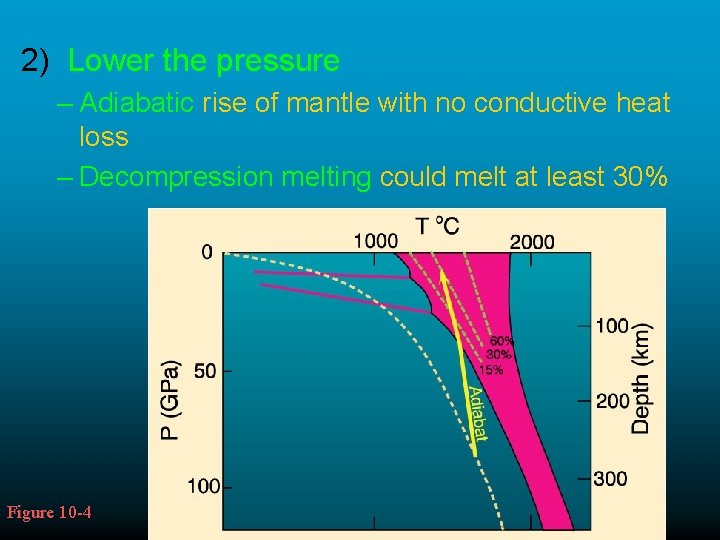
2) Lower the pressure – Adiabatic rise of mantle with no conductive heat loss – Decompression melting could melt at least 30% Figure 10 -4
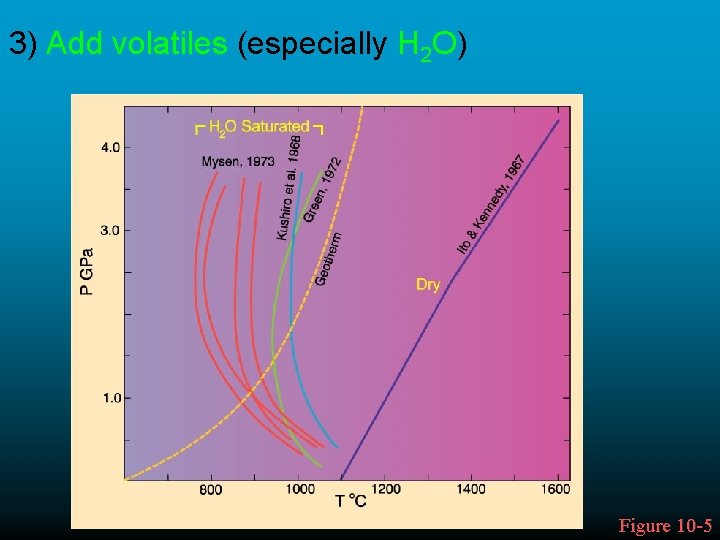
3) Add volatiles (especially H 2 O) Figure 10 -5




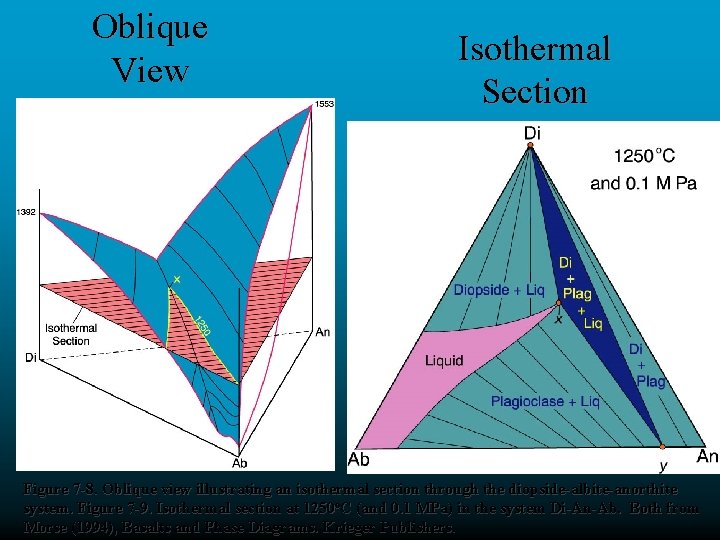
Oblique View Isothermal Section Figure 7 -8. Oblique view illustrating an isothermal section through the diopside-albite-anorthite system. Figure 7 -9. Isothermal section at 1250 o. C (and 0. 1 MPa) in the system Di-An-Ab. Both from Morse (1994), Basalts and Phase Diagrams. Krieger Publishers.
- Slides: 22