Part STRUCTURE of MATERIALS and its EFFECTS on






























- Slides: 44

Part Ⅰ STRUCTURE of MATERIALS and its EFFECTS on PROPERTIES of MATERIALS 1

Chapter 2 Atomic Scale Structure: Interatomic Bonding 2

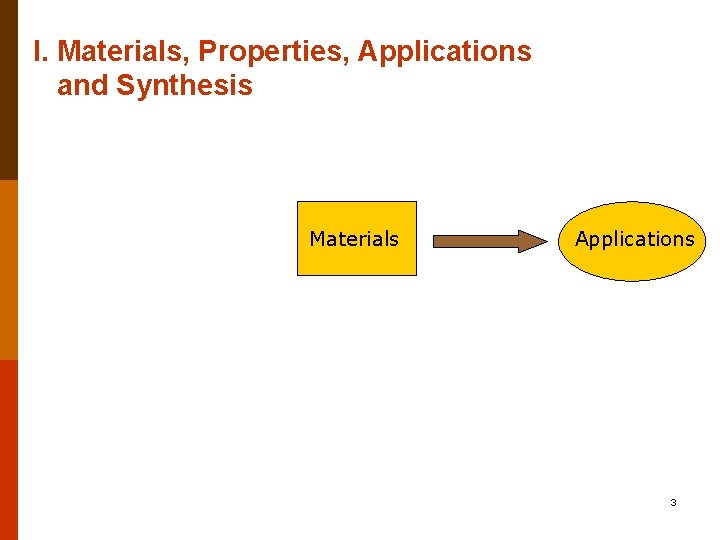
I. Materials, Properties, Applications and Synthesis Materials Applications 3

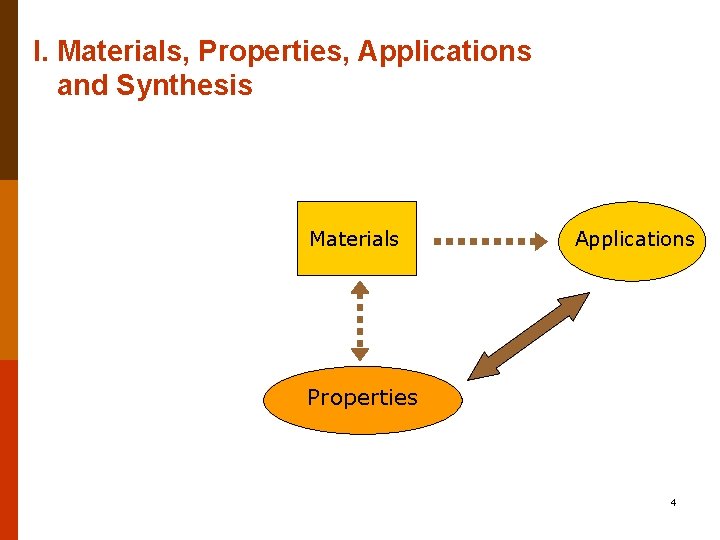
I. Materials, Properties, Applications and Synthesis Materials Applications Properties 4

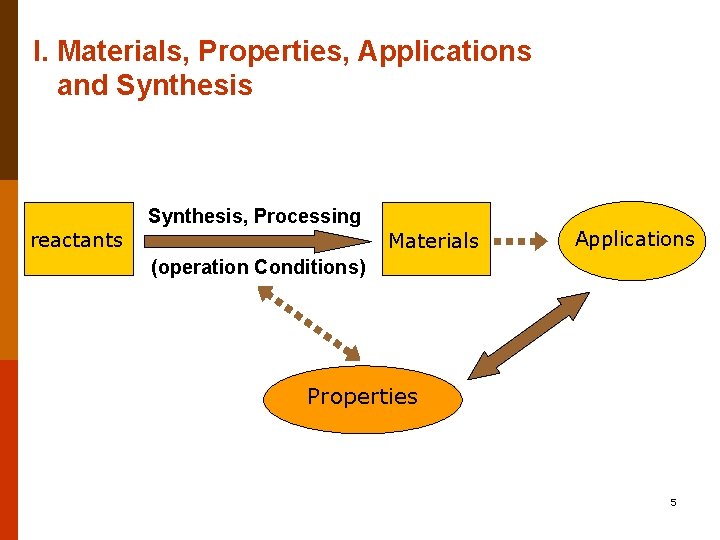
I. Materials, Properties, Applications and Synthesis reactants Synthesis, Processing Materials Applications (operation Conditions) Properties 5

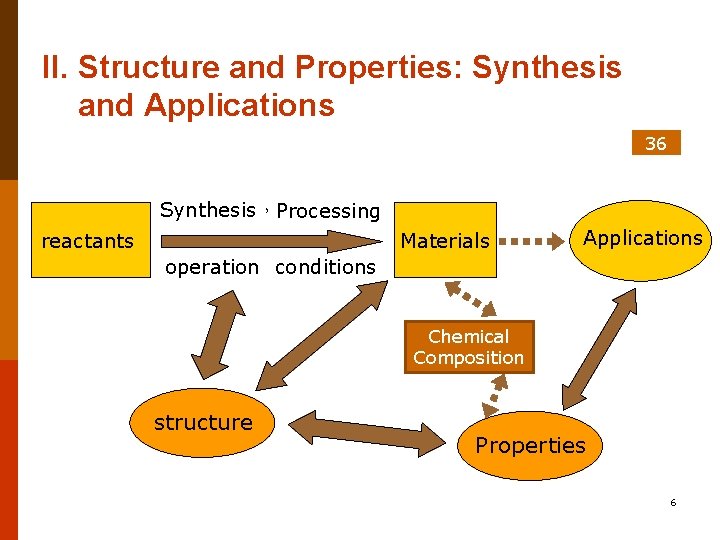
II. Structure and Properties: Synthesis and Applications 36 Synthesis,Processing reactants Materials Applications operation conditions Chemical Composition Structure structure Properties 6

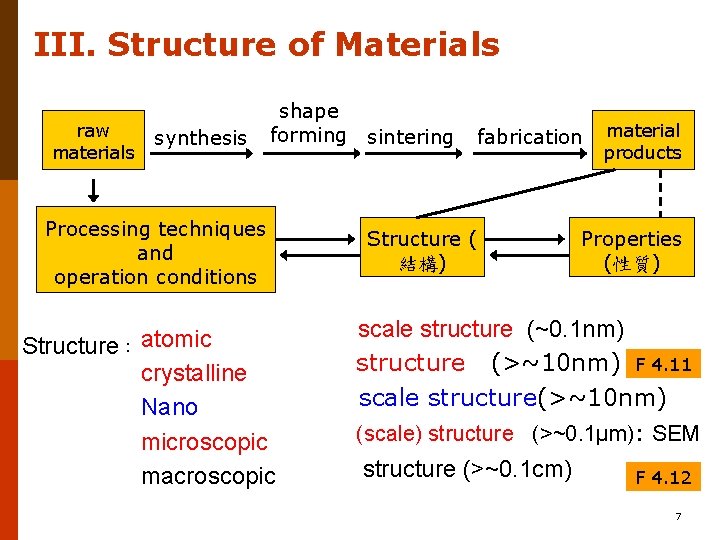
III. Structure of Materials raw materials synthesis shape forming sintering Processing techniques and operation conditions Structure: atomic crystalline Nano microscopic macroscopic fabrication Structure ( 結構) material products Properties (性質) scale structure (~0. 1 nm) structure (>~10 nm) F 4. 11 scale structure(>~10 nm) (scale) structure (>~0. 1µm): SEM structure (>~0. 1 cm) F 4. 12 7

IV. Atomic Scale Structure F 12. 15 (1) the types of atoms present, ·Different types of atoms (different chemical formula) different materials ·same type of atoms (same chemical formula) Different coordination or different crystal structure different materials (2) the types of bonding between the atoms, ·Different types of bonding different materials (3) the way the atoms are packed together: ( coordination and coordination number ) Chap 3 • same type of atoms and same type of bonding Different coordination or different crystal structure different materials ※ Many materials properties are determined by the atomic scale structure alone 8

V. Atomic Bonding in Solids A. Atomic properties related to (primary) bonding ◎ the energy of the electrons in the atom: energy level ◎ the ease of adding or removing one or more electrons from the atom to create a charged ion: Electropositive elements /Electronegative elements Electronegativity, EN(or x) , of an element: relative tendency of that element to gain, or attract, an electron (obtained by arbitrarily fixing the value of H at 2. 1) ◎ Spatial distribution and bond angles. F 2. 7 ◎ valence electrons: the electrons that occupy the outmost filled shell. → stable electron configurations: normally, outmost S+P=8(except He) chemical bonding 9

B. Bonding Forces and Energies FN = F A + F R (2 -2) F 2. 8 FN: net force; FA: attractive force; FR: repulsive force at equilibrium: FA+ FR = 0 (2 -3) r = r 0 (≒ 3Å) (equilibrium spacing:bond length) E =∫Fdr r (W = F · Lr , W = E ) ∞ EN = ∫ FN dr = ∫ ∞ r ∞ FA dr +∫ FR dr = EA+ ER (2. 4) (2. 5 -7) F = - d. E / dr E 0: bonding energy 10

C. Types of Bonding l primary (or chemical) bonds l secondary (or van der Waals, or physical) bonds l Mixed bonds C-1. PRIMARY BONDS ◎ Three types of primary bonds:ionic, covalent, and metallic ◎ Factors determining the type of bonding: (1) electronegativity of each atom (2) electronegativity difference between the atoms (Δ EN, or Δx ) 11

C-1 -1. Ionic Bonding ◎ This bond involves electron transfer from the electropositive atom. A high Δ EN between the atoms favors the formation of ionic bonds. F 2. 4 -1 ◎ The attractive bonding forces are coulombic: positive and F 2. 9 negative ions. 12

◎ The bond-force curve and the bond energy curve F 2. 4 -2 ◎ Equilibrium separation distance, r ° , occurs at the value of r for which FN(r) = FA ( r ) + FR ( r ) = 0 FA ( r) = |z 1 z 2| e 2 /4Π ε 0 r 2 FR(r ) = –k/rm r ° : bond length (FA(r ) ≡ Fa ( x ), FR(r ) ≡ Fr ( x ), FN(r )≡ Ft ( x ) ) 13

◎ Ionization potential, e. g. , 5. 14 e. V for Na Na+ +e. F 2. 4 -2 Electron affinity, e. g. , 4. 02 e. V for C 1 + e- C 1 E ( r ) = Ui — [|Z 1 Z 2|e 2/ (4 0 r)] + (B/ rn ) Ui = 5. 14 – 4. 02 = 1. 12 e. V n = m– 1, B = k/n (E(r) = U(r) ) ◎ The relationship between E and r and will be referred to as either the bond-energy curve or the bond-energy well. ◎ The depth of the energy well (at r 0 ) is the strength of the 14 bond, i. e. , bond energy. The equilibrium separation

C-1 -3. Covalent Bonding ◎ Covalent bonds form in compounds composed of electronegative elements, especially those with four or more valence electrons. Since there are no electropositive atoms present, the “ex-tra” electrons required to fill the valence shell of the electronegative atoms must be obtained by sharing electrons. F 2. 10 ◎ r 0 can be found using a similar equation: FA(r ) + FR (r ) = 0 = A' / χ p– B' / χ q F 12. 15 F 12. 17 ◎ Covalent bonds are directional and not flexible. 15

C-1 -3. Metallic Bonding (metals) ◎ Solids composed primarily of electropositive elements containing three or fewer valence electrons are generally held together by metallic bonds. ◎ The valence electrons form a “cloud” or “sea” of electrons that surrounds the ion cores. The electrons are shared, but they are not spatially localized. F 2. 11 ◎ Metallic bonding may be weak or strong; energies range from 68 k. J/ mol (0. 7 e. V/ atom; m. p. , – 390 C ) for mercury to 850 k. J/ mol (. 8 e. V/ atom; m. p. , 34100 C ) for tungsten. 16

C-1 -4. Mixed bonding • Mixed bonding between primary bonds • Mixed bonding between primary and secondary bonds C-1 -4 -1. Mixed bonding between primary bonds (a bond with characteristics of two different primary bonds) (a) Ceramics: Many ceramics contain primary bonds with mixed ionic/covalent characteristics ◎ Classification of the bonds – qualitative Classification: A bond is considered predominantly ionic when Δx > 1. 7 and predominantly covalent if Δx <1. 7 17

- Quantitative classification: The percent ionic character of a bond between elements A and B ( A being the most electronegative ) may be approximated by the expression % ionic character = ( 1 -e –(0. 25)(XA-XB)2 ) 100 where x. A and x. B are the electro negativities for the respective elements. T 12. 1 e. g. , Si. O 2 : Si atom, EN (XB ) = 1. 9 ; O atom, EN (XA )= 3. 44, ΔEN(Δ X)= 1. 54, the Si-O bond is ~45% ionic and ~55% covalent. 18

(b) Elements in same row of periodic table In the Group IA metals, moving from left to right in the periodic table, bonds begin to take on some covalent characteristics. F 2. 7 The increase in covalent characteristic is part of the reason the density of metals generally decreases as one moves to the right in the periodic table in a given row. (c) Intermetallic, e. g. , Al. Li, Ni 3 Al, Al 3 V, Al. Sb, Cu. Zn, Ti 3 Al, and Mg 2 S, exhibit either mixed metallic/covalent or mixed metallic/ionic bond characteristics. They are more brittle and stiffer and lighter than metals because of possessing ceramic properties. These materials often have good high-temperature resistance and a high strength-to weight ratio. (As a result, Ni 3 Al and Ti 3 Al are finding usage in the aerospace 19 industry) intermetallic

C-1 -4 -2 Mixed bonding between primary and secondary bonds ◎ Another type of “mixed” bonding occurs in materials having both primary and secondary bonds. ◎ Many materials properties are determined by the secondary bonding: - melting point (sublimation temp, glass transition temp) - mechanical strength - ductility (Bonds to be broken for melting or softening or sublimation: secondary bonds. ) 52 36 F 2. 9 -1 F 13. 19 F 3. 14 54 77 20

C-2. SECONDARY BONDS (or van der Waals, or physical bonds) ◎ bonding energies are typically on the order of 10 Kj/mol ( 0. 1 ev/ atom). ◎ Secondary bonds are fundamentally different from primary bonds in that they involve neither electron transfer nor electron sharing. Instead, attractive forces are produced when the center of positive charge is different from the location of the center of negative charge. The resulting electric dipole can be either temporary, induced, or permanent and can occur in atoms or molecules F 2. 27 F 2. 13 F 2. 14 F 2. 12 21

◎ Two major types of secondary bonds • dipoles (or van der Waals bonding) ← ordinary dipoles • hydrogen bonding ← strong dipoles (A) Bonding by dipoles (a) Temporary dipole (or van der Waals bonding, <4 KJ/mol) Fluctuating induced (temporary) dipole bonds ·Temporary dipole is responsible for the condensation of noble gases at low temperatures ·The strength of the van der Waals bond generally increases as the number of atoms in the compound increases. This phenomenon explains why the melting temperatures and also b. p. of the hydrocarbons with chemical formulas Cn. H 2 n+2 22 increase as n increases. F 2. 13

(b) Permanent dipoles: formed between polar molecules, generally stronger than van der Waals bonds. (c) Polar molecule — induced dipole bonds. F 2. 13 F 2. 14 (B) Hydrogen bond (8 -40 KJ/mol) ·occurs whenever a hydrogen atom can be shared between two strongly electronegative atoms such as N, O, F, or C 1. ·Hydrogen bonds hold the wood fibers in a sheet of paper together. F 2. 15 ◎ Secondary bonds control properties such as melting point and elastic modulus in solids where primary bonds do not form a three-dimensional (3 -D) network. 23

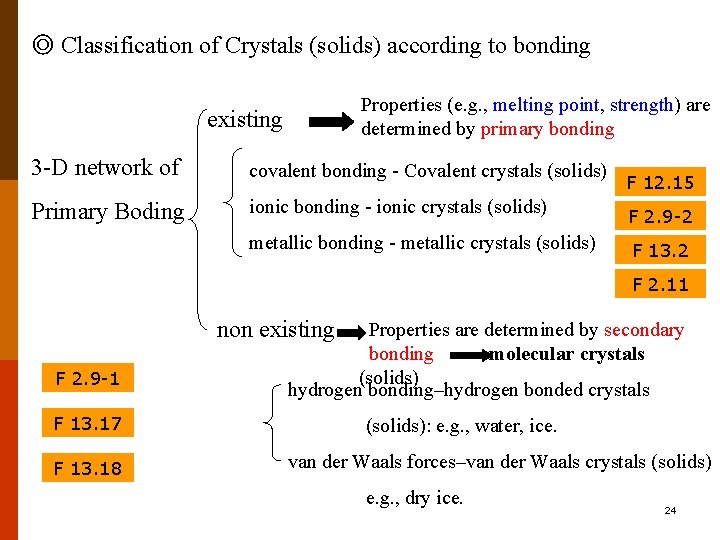
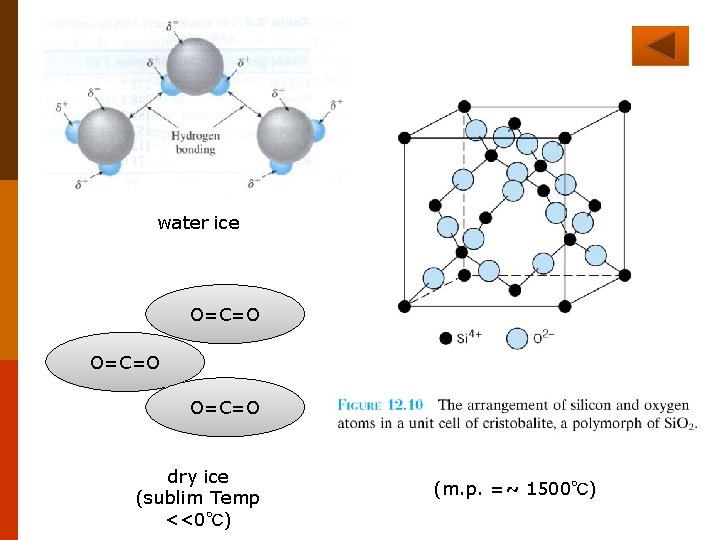
◎ Classification of Crystals (solids) according to bonding Properties (e. g. , melting point, strength) are determined by primary bonding existing 3 -D network of covalent bonding - Covalent crystals (solids) Primary Boding ionic bonding - ionic crystals (solids) F 2. 9 -2 metallic bonding - metallic crystals (solids) F 13. 2 F 12. 15 F 2. 11 non existing F 2. 9 -1 F 13. 17 F 13. 18 Properties are determined by secondary bonding molecular crystals (solids) hydrogen bonding–hydrogen bonded crystals (solids): e. g. , water, ice. van der Waals forces–van der Waals crystals (solids) e. g. , dry ice. 24

VI. Influence of Bonding on Material Properties A. Ductility and Brittleness Some of the mechanical and electrical properties of solids are a function of bond type. A-1. Metallic Bonded Materials ◎ Atoms in the metal can slip and slide past one another with regard to applied force and thus absorb the impact without breaking. This phenomenon is called ductile. (A large extent of deformation can occur. ) ◎ Examples:most metals. F 2. 4 -5 25

A-2. Ionic Bonded Materials ◎ In an ionic solid, each ion is surrounded by oppositely charged ions. Ionic slip may lead to like charges moving adjacent positions, causing Coulombic repulsion. This makes slipping much more difficult to achieve and the material responds by breaking. Such behavior is known as brittle. (Little deformation can occur. ) ◎ Examples:most ceramics. F 2. 4 -5 A-3. Covalent Bonded Materials ◎ Covalent bonds are directional bonding, thus are not flexible. Such materials are therefore brittle. (Little deformation can occur. ) ◎ Example:diamond. 36 26

B. Electrical Conductivity ◎ Electrical conductivity of materials depends (1) the type of charge carrier in the materials (electrons or ions), (2) the spatial density of charge carriers (3) the charge carrier mobility. → determined by type of bonding ◎ Metallic bonded materials: metals F 2. 11 39 F 2. 10 36 F 13. 17 F 13. 18 (freely moving electrons: free electrons) The combination of high mobility and high concentration of charge carriers leads to high electrical conductivities for most metals. ◎ Ionic and covalent bonded materials: electrical insulators (no free electrons) 27

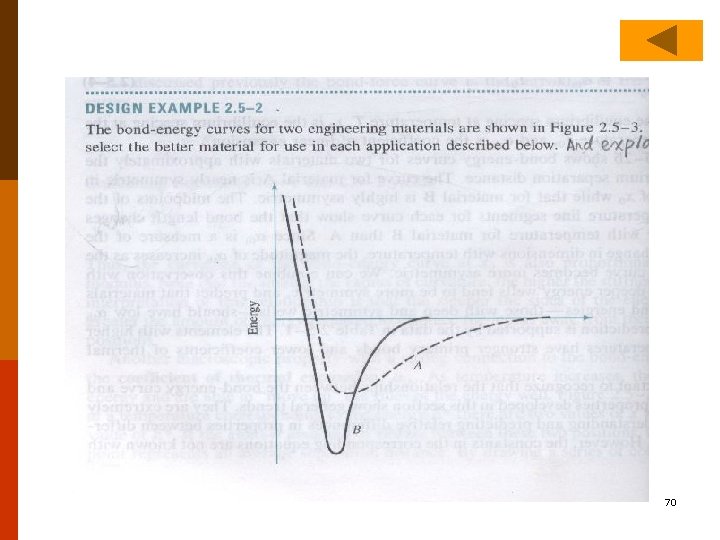
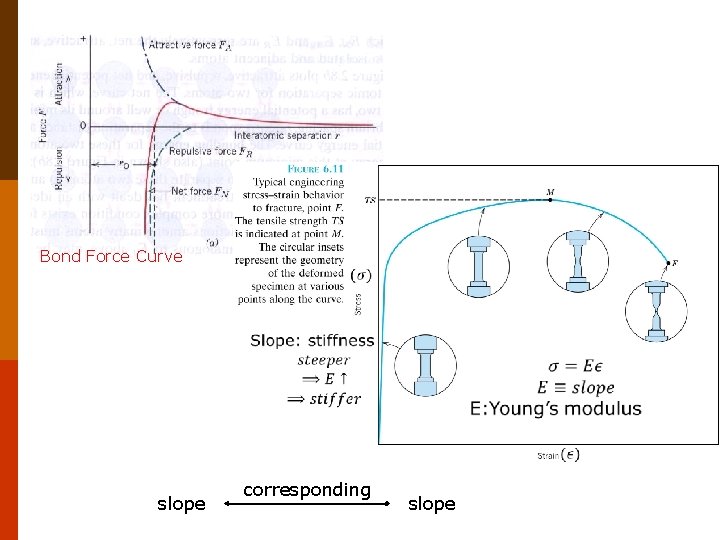
C. Stiffness and Elastic Modulus ◎ The slope of the bond force curve, ( F/ χ ), at χ。 is a measure of the force required to displace atoms from their equilibrium positions. Usually F or F a. E (χ≡r, ≡ r) (2. 5 -1) Where a is a geometric factor and E is a materials property known As Young’s modulus is a measure of the resistance of the material to relative atomic separation (stiffness). The steeper the slope (I. e. , the higher the value of E), the greater the force required to move the atoms from their equilibrium positions. 78 Materials with large values of E are stiff. #70 #71 #72 #73 #74 F 2. 8 F 2. 5 -1 F 6. 7 28

D. Bond Energy and Bond Length ◎The bond-energy curve Several important macroscopic materials properties directly from the bond-energy curve, e. g. , bond energy, average bond length, elastic modulus and coefficient of thermal expansion. ◎ Bond Energy and Bond Length The depth of the energy well (at r ) is the strength of the bond, 0 i. e. , bond energy. The equilibrium separation distance, r ° , corresponds to the bond length. F 2. 8 29

◎ Elastic (young’s) Modulus As discussed previously U F and 2 U 2 F (2. 5 -2) U a (2. 5 -3) 30

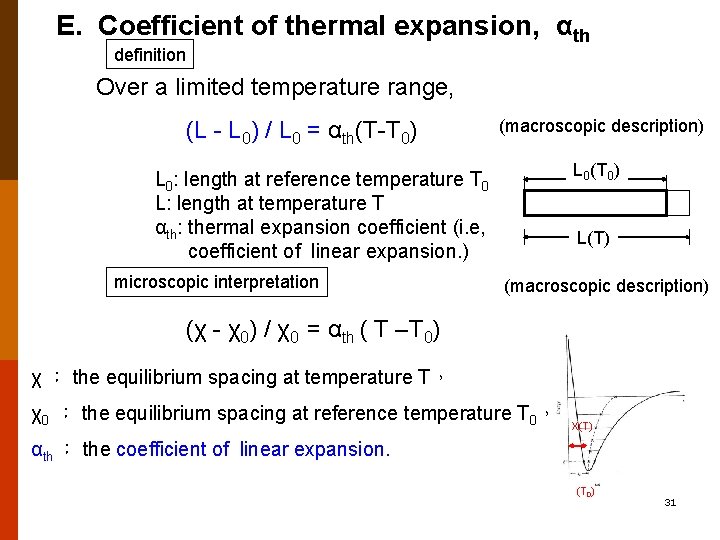
E. Coefficient of thermal expansion, αth definition Over a limited temperature range, (L - L 0) / L 0 = αth(T-T 0) (macroscopic description) L 0(T 0) L 0: length at reference temperature T 0 L: length at temperature T αth: thermal expansion coefficient (i. e, coefficient of linear expansion. ) microscopic interpretation L(T) (macroscopic description) (χ - χ0) / χ0 = αth ( T –T 0) χ : the equilibrium spacing at temperature T, χ0 : the equilibrium spacing at reference temperature T 0, X(T) αth : the coefficient of linear expansion. (T 0) 31

◎ As temperature increases, the atoms gain energy and are able to “ move up “ the sides of the energy well , i. e. , the average atomic-separation distance increases as the temperature increases. ◎ The magnitude of α th increases as the bond-energy curve becomes more asymmetric. Deeper energy wells tend to be more symmetric, materials with high bond energies- those with deep and symmetric wells-- should have low α th values. Thermal expansion coefficient αth L↑ T↑ (macroscopic property) Atomic scale structure (interatomic bonding) (bond energy curve) Bond length↑ potential energy↑ T↑ (microscopic property) F 2. 5 -3 F 2. 5 -1 F 2. 5 -2 32 F 13. 17

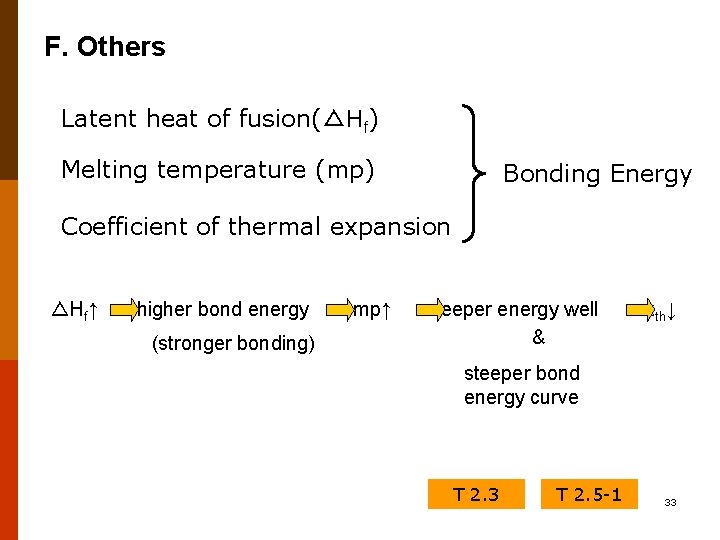
F. Others Latent heat of fusion(△Hf) Melting temperature (mp) Bonding Energy Coefficient of thermal expansion △Hf↑ higher bond energy (stronger bonding) mp↑ deeper energy well & α th↓ steeper bond energy curve T 2. 3 T 2. 5 -1 33

Thermal Shock Resistant Materials: (Thermal Shock Resistance) low thermal expansion coefficient material p symmetric energy well:strong bonding p low density:low CN F 12. 15 F 2. 5 -2 F 12. 10 strong covalent bonded materials T 2. 5 -1 Thermal shock : very quick change of themperature Thermal shock resistant material T 4. 3 -1 F 4. 4 uneven thermal expansion (or contraction) within the material A material, which does not crack under a thermal shock low thermal expansion coefficient material cracking strong covalent bonded materials 34

◎ The relationships between the bond-energy curve and macroscopic properties developed in this section show general trends. The constants in the corresponding equations are not known with sufficient accuracy to facilitate calculation of the absolute values of bond length , bond energy, modulus of elasticity, and coefficient of thermal expansion. The values of these properties for engineering materials are usually directly measured in the laboratory. 35

70

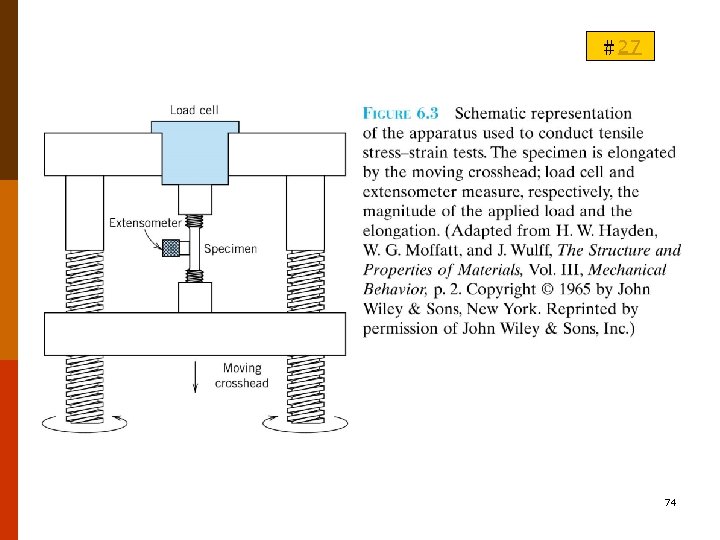
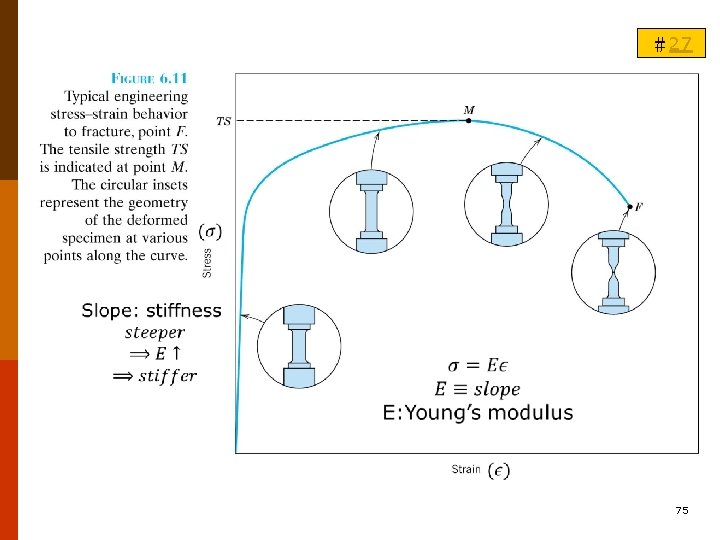
B. ELASTIC DEFORMATION (彈性變形) #27 B-1. Stress-Strain Behavior (A) Tension ◎ For most metals at relatively low levels, stress and strain are proportional to each other (6. 5) F 6. 11 Hooke’s law; the constant of proportionality E (GPa or psi): Modulus of elasticity or Young’s modulus. For most typical metals, E=45 GPa (6. 5 x 106 psi) for Mg to 407 MPa (59 x 106 psi) for W. T 6. 1 71

#27 ◎ Engineering stress (6. 1) F:the instantaneous load applied newtons (N) or pounds force (lbf) ; A 0:original cross-sectional area (m 2 or in. 2); : engineering stress, MPa (SI) (where 1 MPa = 106 N/m 2), and pounds force per square inch, psi (Customary U. S. ). 2 72

#27 ◎ Engineering strain (6. 2) l 0:the original length; li :the instantaneous length; Δl= li - l 0 Engineering strain, :unitless (meters per meter or inches per inch ; or a percentage) 73

#27 74

#27 75

metallic Metallic + ionic or covalent intermetallic (compounds): Al. Li, Ni 3 Al, Al 3 V, Ti 3 Al, Cu. Zn, … metals: Al, Ti, Li, Ni, Zn… ionic or covalent ceramics: Al 2 O 3, Zr. O 2, Si. O 2, … ductile ductility ↓ electrically conductive electrical conductivity ↑ electrically insulation low mechanical strength ↓ brittle high mechanical strength (low stittness) density ↑ high stittness high density temp. resistance ↑ low density low m. p. ★ strength to weight ratio ↑ (light, tough, hightemp materials) high m. p. 76

water ice O=C=O dry ice (sublim Temp <<0℃) (m. p. =~ 1500℃)

Bond Force Curve slope corresponding slope