Part B Definitions Empirical Formula a formula giving

Part B. Definitions • Empirical Formula = a formula giving the ratios (proportions) of the elements present in a compound but not the actual numbers or arrangement of atoms… aka the reduced/simplest form • Examples: CH 2 O --- this is the empirical form for glucose, C 6 H 12 O 6 C 3 H 4 O --- the subscripts cannot be simplified so must be in their reduced form, making this empirical

Part B. Definitions • Molecular Formula = a formula giving the number of atoms of each of the elements present in one molecule of a specific compound (not necessarily the reduced form, but it could be) • Examples: C 6 H 12 O 6 (sugar) --- Dividing all subscripts by 6 would give the empirical form, CH 2 O N 2 O 4 --- dividing all subscripts by 2 would give the empirical form NO 2 H 2 O --- this is the molecular and empirical form because this is actually what the H 2 O molecule looks like
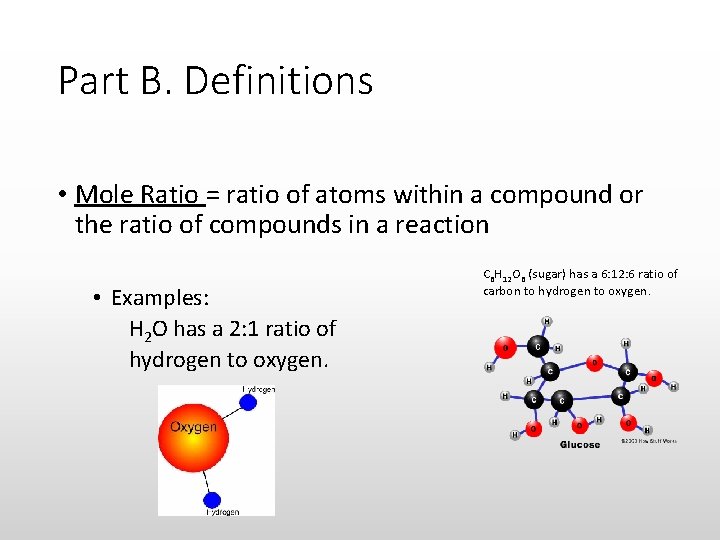
Part B. Definitions • Mole Ratio = ratio of atoms within a compound or the ratio of compounds in a reaction • Examples: H 2 O has a 2: 1 ratio of hydrogen to oxygen. C 6 H 12 O 6 (sugar) has a 6: 12: 6 ratio of carbon to hydrogen to oxygen.

PART C. Identify the following as empirical or molecular 1. Formaldehyde CH 2 O 2. dinitrogen tetroxide N 2 O 4 3. carbon monoxide CO 4. acetic acid C 2 H 4 O 2 5. Hydrogen peroxide H 2 O 2

PART C. Identify the following as empirical or molecular 6. Acetylene C 2 H 2 7. Water H 2 O 8. Carbon dioxide CO 2 9. Butene C 4 H 8 10. glucose C 6 H 12 O 6
- Slides: 5