Part 3 ii Substitution Reactions Solvent Me OH
Part 3 ii Substitution Reactions: Solvent Me. OH DMSO

Content of Part 3 ii Solvent polarity and its effect on the SN 1 reaction mechanism Solvent polarity and its effect on the SN 2 reaction mechanism Protic and non-protic solvents and their effect on the SN 2 mechanism Protic and polar non-protic solvents and change of mechanism from SN 1 to SN 2 Increasing solvent polarity and change of mechanism from SN 2 to SN 1

CHM 1 C 3 – Introduction to Chemical Reactivity of Organic Compounds– – Learning Objectives Part 3 ii – Substitution Reactions: Solvent After completing PART 5 ii of this course you should have an understanding of, and be able to demonstrate, the following terms, ideas and methods. (i) Understand how changes in the polarity of the solvent can change the rate of reaction in both SN 2 and SN 1 reaction mechanisms, (ii) Understand what is meant by the dielectric constant of a solvent, (iii) Understand what is meant by protic and non-protic solvents, (iv) Understand what a hydrogen bond is, (v) Understand how protic solvents reduce the rate of reaction of nucleophiles with substrates, and (vi) Understand how increasing the polarity of the solvent can change the mechanism of substitution from SN 2 to SN 1.

Effect of Solvent Changing the solvent in which a reaction is carried out in can (i) Effect the reaction rate (ii) Result in a change in reaction mechanism
![Solvent Polarity and SN 1 Reactions Relative Rate Solvent: Rate = k[R-Br] 1 H Solvent Polarity and SN 1 Reactions Relative Rate Solvent: Rate = k[R-Br] 1 H](http://slidetodoc.com/presentation_image_h/da75b9153b8d0792f9b2519424eb23de/image-5.jpg)
Solvent Polarity and SN 1 Reactions Relative Rate Solvent: Rate = k[R-Br] 1 H 2 O/Et. OH SN 1 Solvent: Rate = k[R-Br] H 2 O SN 1 Why is reaction much faster in water alone? 30 000

Both reactions must involve the sp 2 hybridised carbocation. The carbocation is obviously a very polar species. Polar species are most stable in polar solvents. The dielectric constant (e) of a solvent is a measure of the polarity of a solvent. The larger the e the more polar the solvent. e (H 2 O) = 78 e (Et. OH) = 30 Thus, higher e solvent solvate and stabilise the carbocation more easily than lower e solvents. Thus, cations are generated more rapidly in higher e solvents.

Solvent Polarity and SN 2 Reactions Increasing solvent polarity on substrates that undergo SN 2 substitution reactions has a much less dramatic effect. As there is no cation formed there is nothing to stabilise. However, it is found that there is usually a slight decrease in rate with increasing polarity, especially when the nucleophile is charged, i. e. an anion. In much the same way as a carbocation is stabilised by higher polarity solvents, so are anions. Thus, in higher polarity solvents the nucleophile is more solvated and more stable, and therefore less reactive and the rate drops in higher polarity solvents.

Protic Solvents and Nucleophile Interaction A protic solvent is one in which there is hydrogen capable of forming a hydrogen bond to the nucleophile, i. e. a hydrogen attached to an electronegative heteroatom (O and N). e. g. H 2 O, Me. OH, Et. OH This results in a highly solvated and stable nucleophile, and therefore relative unreactive.

Non-Protic Solvents and the Nucleophile Interaction A non-protic solvent is one in which there is no hydrogen capable of forming a hydrogen bond to the nucleophile. This then results in the nucleophile being poorly solvated and relatively more reactive. DMSO DMF Me. CN Chloroform Dichloromethane Acetone Ether Ethylacetate Alkanes
![Rel. Rate Protic Solvent Me. OH Non-Protic Solvent DMF Rate = k[R-I][N 3 -] Rel. Rate Protic Solvent Me. OH Non-Protic Solvent DMF Rate = k[R-I][N 3 -]](http://slidetodoc.com/presentation_image_h/da75b9153b8d0792f9b2519424eb23de/image-10.jpg)
Rel. Rate Protic Solvent Me. OH Non-Protic Solvent DMF Rate = k[R-I][N 3 -] 1 SN 2 Rate = k[R-I][N 3 -] 45 000 SN 2 It should be noted Me. OH and DMF have similar e values, 33 and 37 respectively. Thus, any rate differences are not a result of solvating ability alone.
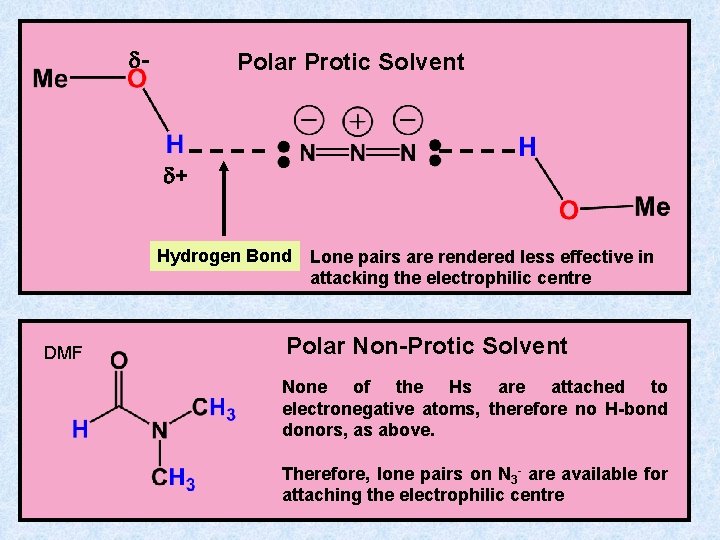
d- Polar Protic Solvent d+ Hydrogen Bond DMF Lone pairs are rendered less effective in attacking the electrophilic centre Polar Non-Protic Solvent None of the Hs are attached to electronegative atoms, therefore no H-bond donors, as above. Therefore, lone pairs on N 3 - are available for attaching the electrophilic centre
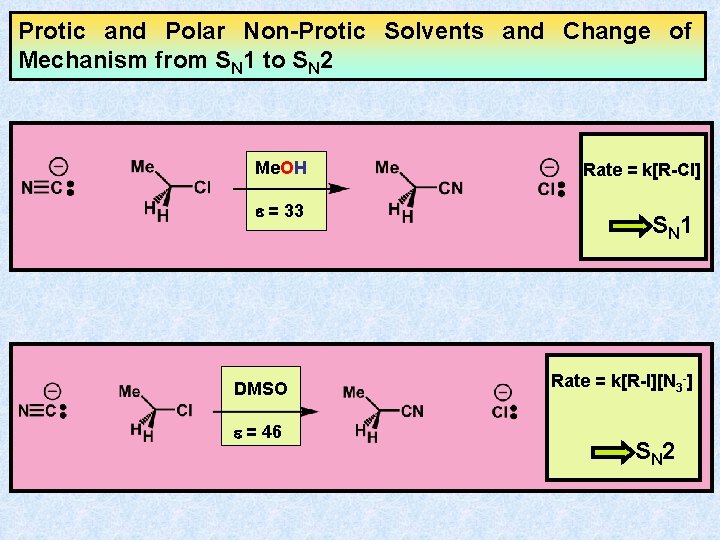
Protic and Polar Non-Protic Solvents and Change of Mechanism from SN 1 to SN 2 Me. OH e = 33 DMSO e = 46 Rate = k[R-Cl] S N 1 Rate = k[R-I][N 3 -] S N 2
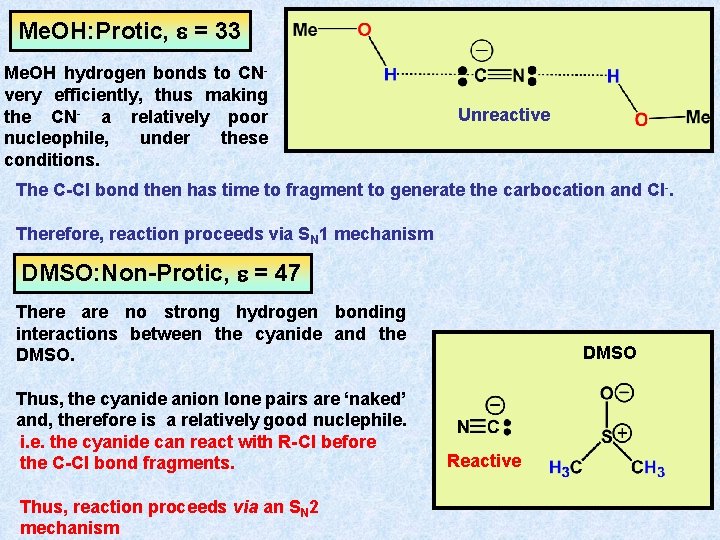
Me. OH: Protic, e = 33 Me. OH hydrogen bonds to CNvery efficiently, thus making the CN- a relatively poor nucleophile, under these conditions. Unreactive The C-Cl bond then has time to fragment to generate the carbocation and Cl-. Therefore, reaction proceeds via SN 1 mechanism DMSO: Non-Protic, e = 47 There are no strong hydrogen bonding interactions between the cyanide and the DMSO. Thus, the cyanide anion lone pairs are ‘naked’ and, therefore is a relatively good nuclephile. i. e. the cyanide can react with R-Cl before the C-Cl bond fragments. Thus, reaction proceeds via an SN 2 mechanism DMSO Reactive
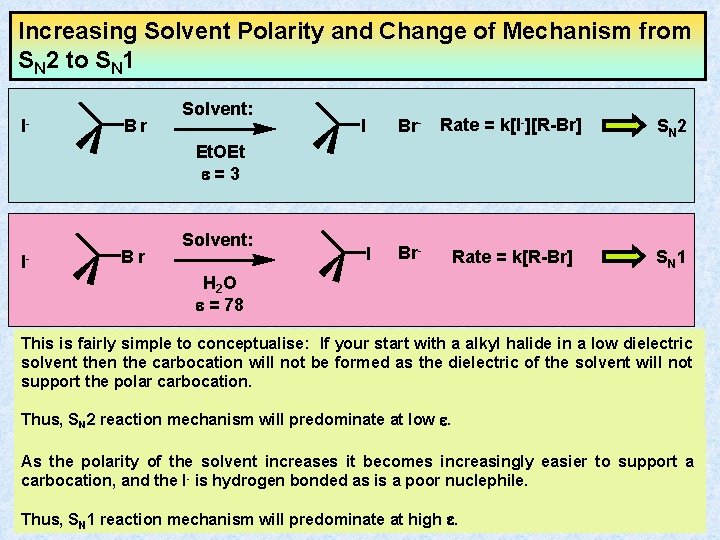
Increasing Solvent Polarity and Change of Mechanism from SN 2 to SN 1 I- Br Solvent: I Br- Rate = k[I-][R-Br] SN 2 Br- SN 1 Et. OEt e=3 I- Br Solvent: I Rate = k[R-Br] H 2 O e = 78 This is fairly simple to conceptualise: If your start with a alkyl halide in a low dielectric solvent then the carbocation will not be formed as the dielectric of the solvent will not support the polar carbocation. Thus, SN 2 reaction mechanism will predominate at low e. As the polarity of the solvent increases it becomes increasingly easier to support a carbocation, and the I- is hydrogen bonded as is a poor nuclephile. Thus, SN 1 reaction mechanism will predominate at high e.
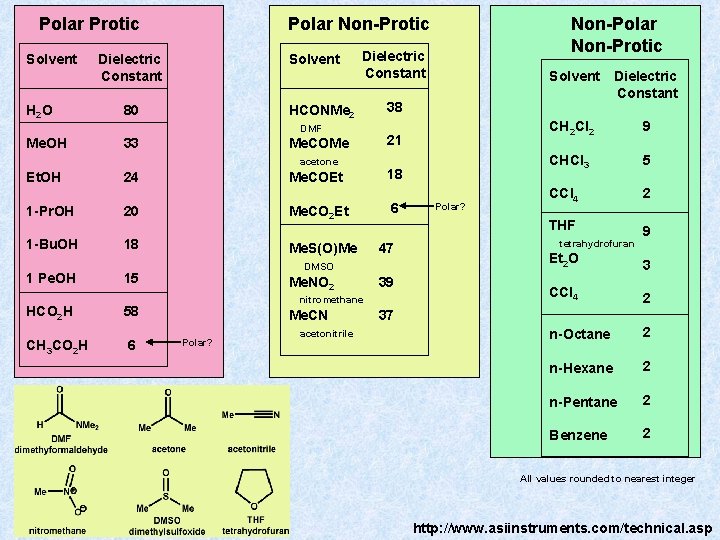
Polar Protic Solvent H 2 O Polar Non-Protic Solvent Dielectric Constant HCONMe 2 80 DMF Me. OH Me. COMe 33 acetone Et. OH 24 1 -Pr. OH 20 1 -Bu. OH 18 1 Pe. OH HCO 2 H 58 CH 3 CO 2 H 6 21 Me. COEt Me. CO 2 Et 6 47 DMSO Me. NO 2 39 nitromethane Me. CN Polar? acetonitrile Solvent Dielectric Constant 38 18 Me. S(O)Me 15 Non-Polar Non-Protic Polar? CH 2 Cl 2 9 CHCl 3 5 CCl 4 2 THF 9 tetrahydrofuran Et 2 O 3 CCl 4 2 n-Octane 2 n-Hexane 2 n-Pentane 2 Benzene 2 37 All values rounded to nearest integer http: //www. asiinstruments. com/technical. asp
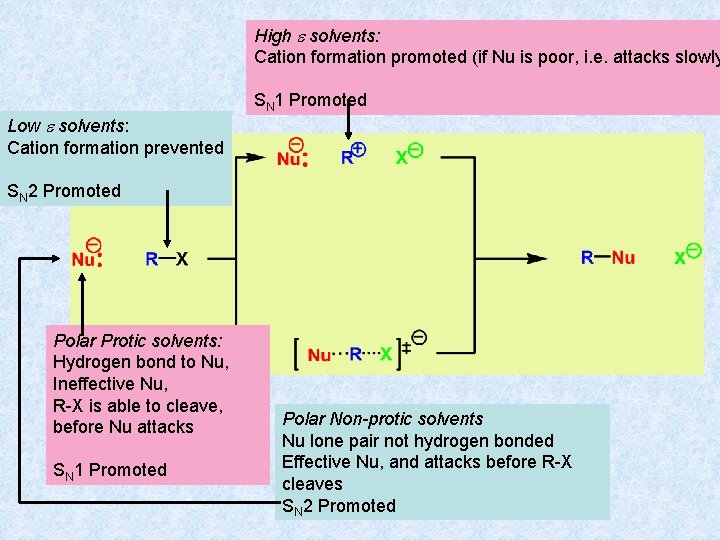
High e solvents: Cation formation promoted (if Nu is poor, i. e. attacks slowly Low e solvents: Cation formation prevented SN 1 Promoted SN 2 Promoted Polar Protic solvents: Hydrogen bond to Nu, Ineffective Nu, R-X is able to cleave, before Nu attacks SN 1 Promoted Polar Non-protic solvents Nu lone pair not hydrogen bonded Effective Nu, and attacks before R-X cleaves SN 2 Promoted

CHM 1 C 3 – Introduction to Chemical Reactivity of Organic Compounds– – Summary Sheet Part 3 ii – Substitution Reactions: Solvent If one considers an SN 1 reaction in which a carbocation is formed, then increasing the polarity of the solvent will lead to better solvation of the carbocation, and therefore greater stability. Thus, increasing the solvent polarity from, for example Et. OH (e = 30) to H 2 O (e = 78), the rate of reaction increases by several orders of magnitude as the stability of the carbocation is so much greater in H 2 O than Et. OH. Indeed, a reaction in a low dielectric solvent may go via an SN 2 reaction (i. e. no requirement for stabilisation of a carbocation), but when transferred to a higher dielectric solvent may change mechanism to SN 1. Not only is the stabilisation of the carbocation important by the solvent, but also the interaction of the nucleophile with the solvent. Nucleophiles have by definition a lone pair of electrons for donation to electrophilic centres, i. e. the carbon attached to the leaving group. However, if the solvent has a hydrogen atom attached to a electronegative heteroatom (O, S, N, such as an alcohol, amine or thiol) then this hydrogen atom will carry a partial positive charge and interact as hydrogen bond acceptor with the lone pair of electrons on the nucleophile. Thus, the lone pair of electrons will now be less effective in a nucleophilic sense. Thus, SN 2 mechanisms might be slowed down on transferring to protic solvents, or even change to SN 1 if the media is even just slight more polar due to the increased time available for scission of the leaving group bond. C-
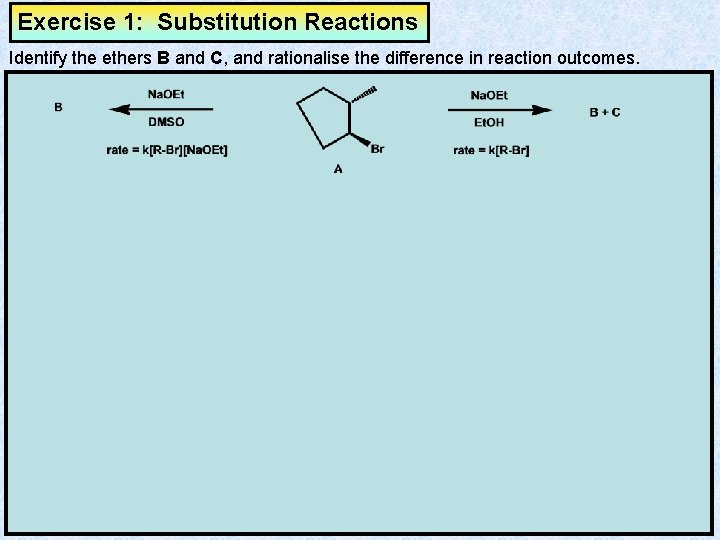
Exercise 1: Substitution Reactions Identify the ethers B and C, and rationalise the difference in reaction outcomes.
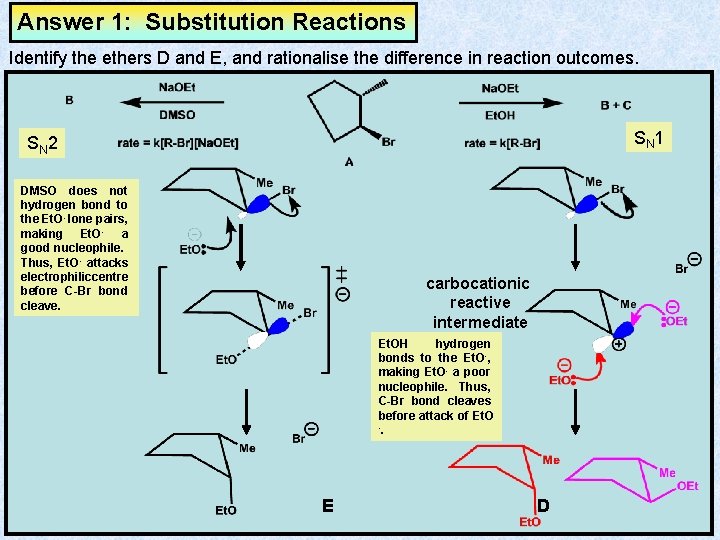
Answer 1: Substitution Reactions Identify the ethers D and E, and rationalise the difference in reaction outcomes. SN 1 SN 2 DMSO does not hydrogen bond to the Et. O- lone pairs, making Et. O- a good nucleophile. Thus, Et. O- attacks electrophilic centre before C-Br bond cleave. carbocationic reactive intermediate Et. OH hydrogen bonds to the Et. O-, making Et. O- a poor nucleophile. Thus, C-Br bond cleaves before attack of Et. O -. E D
- Slides: 19