Part 2 Relationship Between Atomic Absorption and Flame

Part 2

Relationship Between Atomic Absorption and Flame Emission • Flame Emission: it measures the radiation emitted by the excited atoms that is related to concentration. • Atomic Absorption: it measures the radiation absorbed by the unexcited atoms that are determined. • Atomic absorption depends only upon the number of unexcited atoms, the absorption intensity is not directly affected by the temperature of the flame. • The flame emission intensity in contrast, being dependent upon the number of excited atoms, is greatly influenced by temperature variations. Prof. Dr. Hisham Ezzat Abdellatef 2

Atomizers in emission techniques Type Arc Method of Atomization Radiation Source sample heated in an sample electric arc (4000 -5000 o. C) Spark sample excited in a high voltage spark Flame sample solution aspirated into a flame (1700 – 3200 o. C) sample Argon sample heated in an plasma argon plasma (4000 -6000 o. C) Prof. Dr. Hisham Ezzat Abdellatef sample 3

Atomizers in absorption techniques Type Method of Atomization Atomic sample solution aspirated (flame) into a flame Radiation Source HCL atomic sample solution evaporated HCL (nonflame) & ignited (2000 -3000 o. C) (Electrothermal) Hydride generation Vapor hydride generated HCL Cold vapor generated (Hg) Prof. Dr. Hisham Ezzat Abdellatef HCL 4

Atomizers in fluorescence techniques Type atomic (flame) atomic (nonflame) x-ray fluorescence Method of Atomization sample aspirated into a flame sample evaporated & ignited not required Prof. Dr. Hisham Ezzat Abdellatef Radiation Source sample 5

• • Flame Atomization: In a flame atomizer, a solution of the sample is nebulized by a flow of gaseous oxidant, mixed with a gaseous fuel, and carried into a flame where atomization occurs. The following processes then occur in the flame. Desolvation (produce a solid molecular aerosol) Dissociation (leads to an atomic gas) Ionization (to give cations and electrons) Excitation (giving atomic, ionic, and molecular emission) Prof. Dr. Hisham Ezzat Abdellatef 6
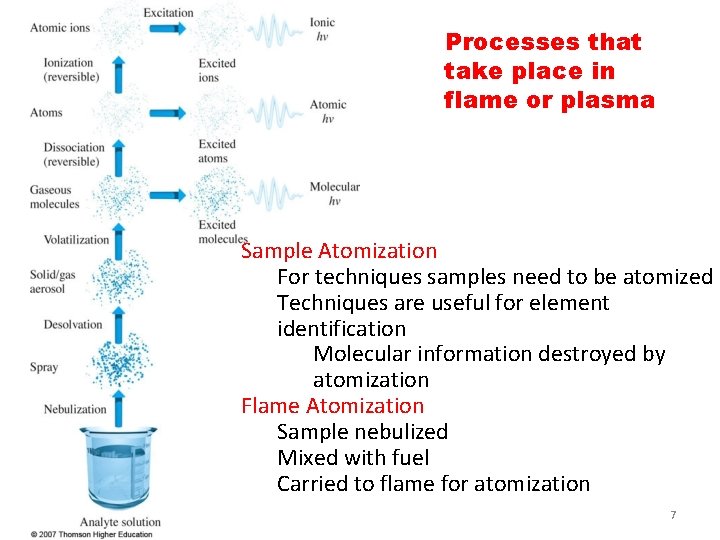
Processes that take place in flame or plasma Sample Atomization For techniques samples need to be atomized Techniques are useful for element identification Molecular information destroyed by atomization Flame Atomization Sample nebulized Mixed with fuel Carried to flame for atomization T 1 7
![The Atomization Process nebulization desolvation mist [MX]solid vaporization excitation or absorption [M 0]gas ground The Atomization Process nebulization desolvation mist [MX]solid vaporization excitation or absorption [M 0]gas ground](http://slidetodoc.com/presentation_image/00648e36965f73228dcb7f24c8c24c77/image-8.jpg)
The Atomization Process nebulization desolvation mist [MX]solid vaporization excitation or absorption [M 0]gas ground state emission [M*]gas excited state (via heat or light) Prof. Dr. Hisham Ezzat Abdellatef at om iza tio n solution [M+, X-]aq [MX]gas atomization [M+, X-]aq [M 0]gas [X 0]gas [M+]gas [X+]gas 8

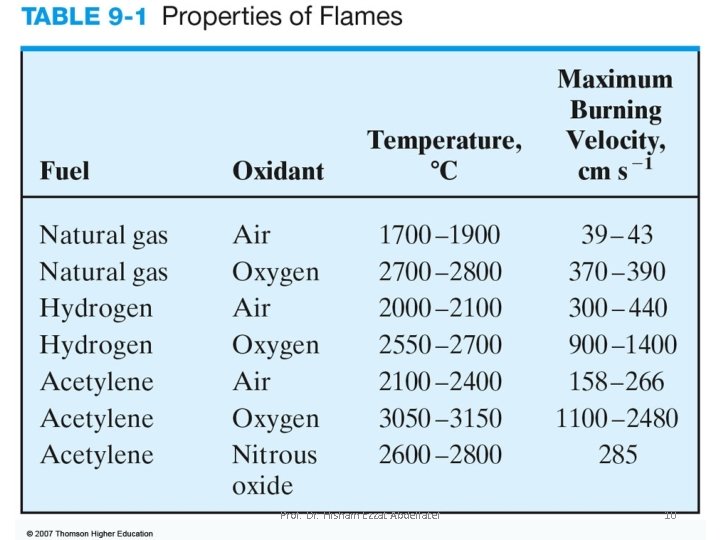
Types of Flames: Several common fuels and oxidants can be employed in flame spectroscopy depending on temperature needed. Temperatures of 1700 o. C to 2400 o. C are obtained with the various fuels when air serves as the oxidant. At these temperature, only easily decomposed samples are atomized. For more refractory samples, oxygen or nitrous oxide must be employed as the oxidant. With the common fuels these oxidants produce temperatures of 2500 o. C to 3100 o. C. Prof. Dr. Hisham Ezzat Abdellatef 9
Prof. Dr. Hisham Ezzat Abdellatef 10

Burning Velocity: The burning velocities are of considerable importance because flames are stable in certain ranges of gas flow rates only. If the gas flow rate does not exceed the burning velocity, the flame propagates itself back in to the burner, giving flashback. As the flow rate increases, the flame rises until it reaches a point above the burner where the flow velocity and the burning velocity are equal. This region is where the flame is stable. At higher flow rates, the flame rises and eventually reaches a point where it blows off of the burner. Prof. Dr. Hisham Ezzat Abdellatef 11

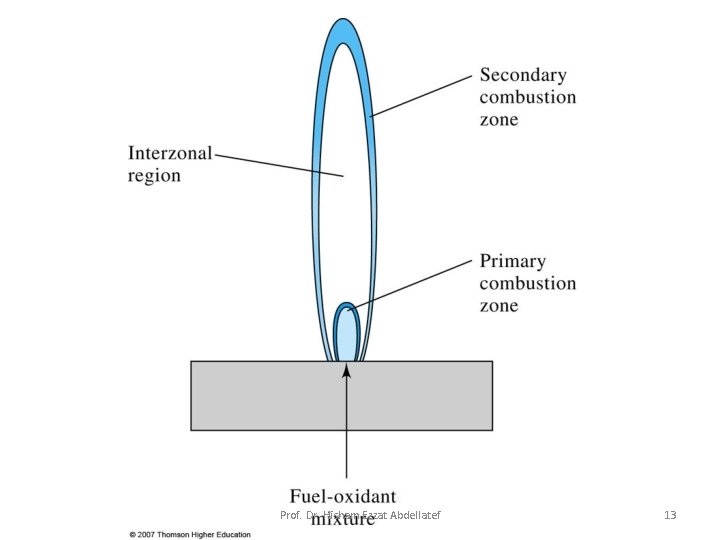
• Flame Structure: Important regions of a flame include: 1. primary combustion zone 2. interzonal region 3. secondary combustion zone 1. Primary combustion zone: Thermal equilibrium is ordinarily not reached in this region, and it is, therefore, seldom used for flame spectroscopy. Prof. Dr. Hisham Ezzat Abdellatef 12
Prof. Dr. Hisham Ezzat Abdellatef 13

2. Interzonal region: This area is relatively narrow in stoichiometric hydrocarbon flames, is often rich in free atoms and is the most widely used part of the flame for spectroscopy. 3. Secondary combustion zone: In the secondary reaction zone, the products of the inner core are converted to stable molecular oxides that are then dispersed into the surroundings. Prof. Dr. Hisham Ezzat Abdellatef 14

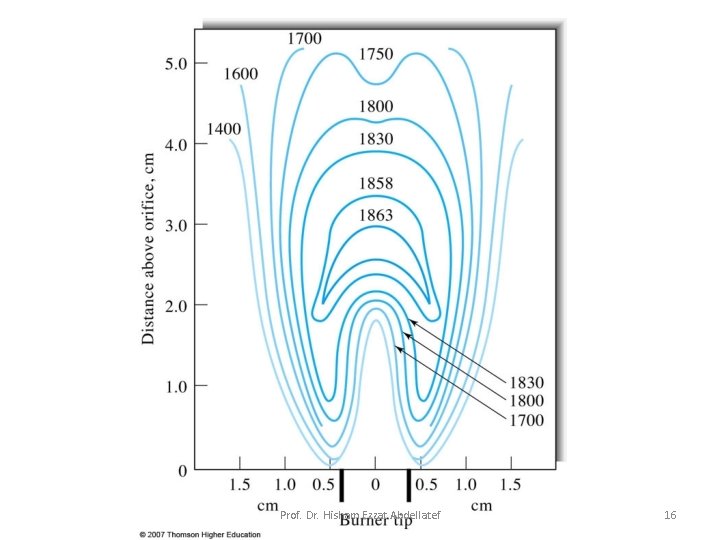
Temperature Profiles: A temperature profile of a typical flame for atomic spectroscopy is shown in Fig. 9 -3. The maximum temperature is located in the flame about 1 cm above the primary combustion zone. It is important– particularly for emission methods – to focus the same part of the flame on the entrance slit for all calibrations and analytical measurements. Prof. Dr. Hisham Ezzat Abdellatef 15
Prof. Dr. Hisham Ezzat Abdellatef 16
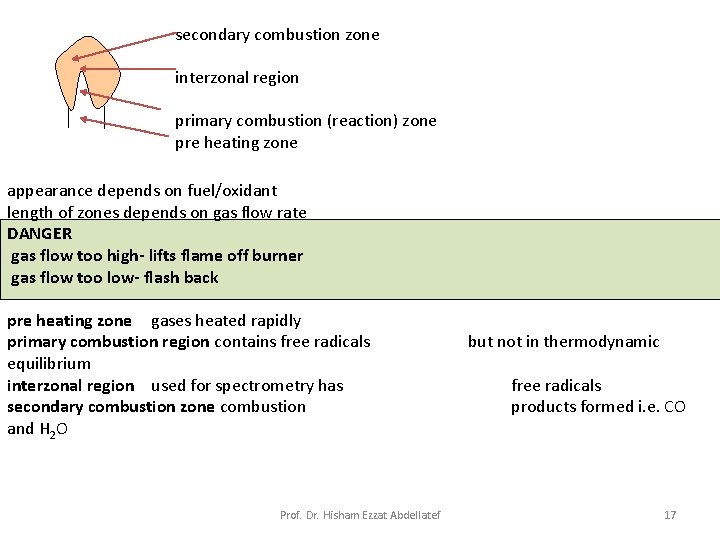
secondary combustion zone interzonal region primary combustion (reaction) zone pre heating zone appearance depends on fuel/oxidant length of zones depends on gas flow rate DANGER gas flow too high- lifts flame off burner gas flow too low- flash back pre heating zone gases heated rapidly primary combustion region contains free radicals equilibrium interzonal region used for spectrometry has secondary combustion zone combustion and H 2 O but not in thermodynamic Prof. Dr. Hisham Ezzat Abdellatef free radicals products formed i. e. CO 17

Types of fuel/oxidant air/acetylene 2300 o. C most widely used. C 2 H 2 + 2. 502 + 10 N 2 → 2 CO 2 + H 2 O + 10 N 2 stoichiometric reaction nitrous oxide/acetylene 2750 o. C hot and reducing red feather zone - due to CN very reactive free radical scavenger for 02 → lowers partial pressure of 02 in zone reducing atmosphere C 2 H 2 + 5 N 2 O → 2 CO 2 + H 2 O + 5 N 2 Prof. Dr. Hisham Ezzat Abdellatef 18

Why do you need a different burner for different oxidants? because to prevent flash back linear gas flow rate needs to 3 x speed of which flame can travel, burning velocity). Prof. Dr. Hisham Ezzat Abdellatef 19

Role of Chemistry in the Flame sample atomised by thermal and chemical dissociation H 2 + Q → H + H O 2 + Q → O + O H + O 2 → OH + O O + H 2 → OH + H equilibrium achieved by 3 rd body collision (B) i. e. N 2, O 2 H + B → H 2 + B + Q H + OH + B → H 2 O + B + Q Free reductions may react with sample to produce atoms i. e. H + HO + Na. Cl → H 2 O + Na + Cl Na + Q → Na * 10 Prof. Dr. Hisham Ezzat Abdellatef 20

Flame Atomisation Process Sample must be in the form of a fine mist so as not to put out flame. Breaks down sample into very fine drops to form liquid aerosol or mist. This assist atomisation as sample only in flame ≈ 0. 025 s Sample drawn up capillary tube at high velocity Sample oxidant Prof. Dr. Hisham Ezzat Abdellatef 21

Suction caused by high flows of oxidant gas and Venturi effect. The high gas flow rate at the end of the capillary creates a pressure drop in the capillary – the pressure in capillary is below atmospheric pressure and sample solution is pulled up. The high speed gas breaks the solution into a fine mist by turbulence as it emerges from capillary. How do we get a better aerosol? • use impact bead (glass or alloy) to encourage aerosol formation and remove large droplets. Prof. Dr. Hisham Ezzat Abdellatef 22

Flame absorbance Profiles: Fig. 9 -4 shows typical absorption profiles for three elements. Magnesium exhibits a maximum in absorbance at the middle of the flame. The behavior of silver, which is not readily oxidized, is quite different, a continuous increase in the number of atoms, and thus the absorbance, is observed from the base to the periphery of the flame. Chromium, which forms very stable oxides, shows a continuous decrease in absorbance beginning close to the burner tip. Prof. Dr. Hisham Ezzat Abdellatef 23
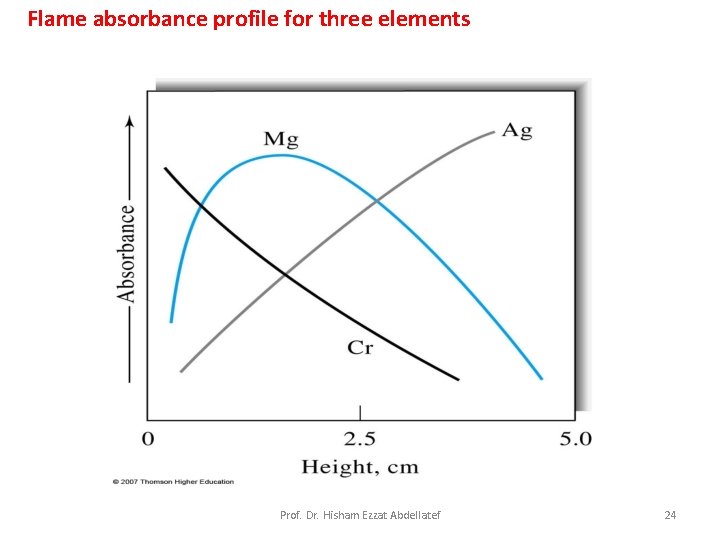
Flame absorbance profile for three elements Prof. Dr. Hisham Ezzat Abdellatef 24

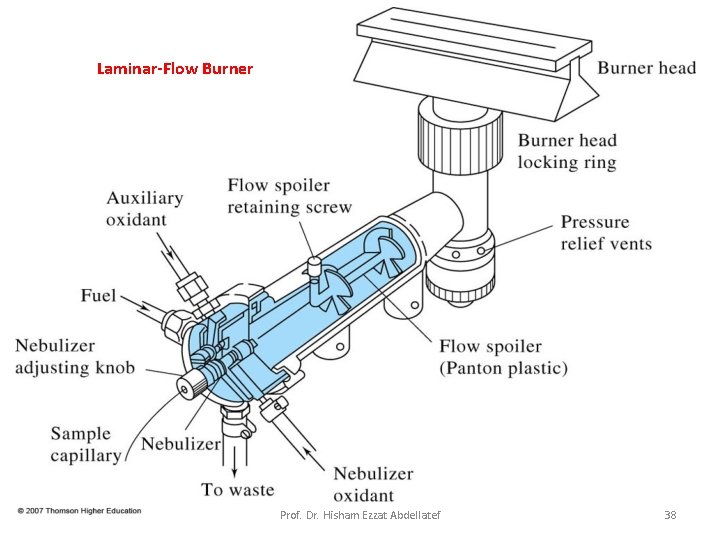
Flame Atomizers: Figure 9 -5 is a diagram of a typical commercial laminar flow burner that employs a concentric tube nebulizer. The aerosol is mixed with fuel. The aerosol, oxidant, and fuel are then burned in a slotted burner that provides a flame that is usually 5 or 10 cm in length. Prof. Dr. Hisham Ezzat Abdellatef 25
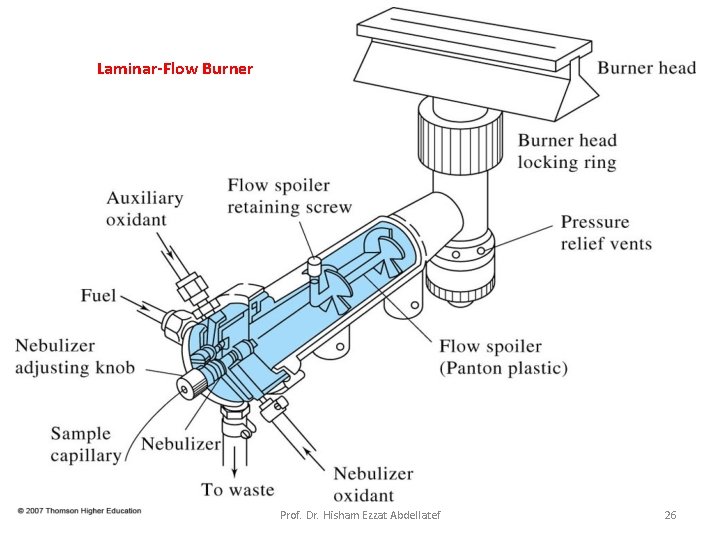
Laminar-Flow Burner Prof. Dr. Hisham Ezzat Abdellatef 26

Advantages: 1. Uniform dropsize 2. Homogeneous flame 3. Quiet flame and a long path length Disadvantages: 1. Flash back if Vburning > Vflow 2. ~90% of sample is lost 3. Large mixing volume Prof. Dr. Hisham Ezzat Abdellatef 27

Sample introduction techniques Prof. Dr. Hisham Ezzat Abdellatef 28

Methods of Sample Introduction in Atomic Spectroscopy Prof. Dr. Hisham Ezzat Abdellatef 29

Nebulization • Nebulization is conversion of a sample to a fine mist of finely divided droplets using a jet of compressed gas. – The flow carries the sample into the atomization region. • Pneumatic Nebulizers (most common) Four types of pneumatic nebulizers: • Concentric tube - the liquid sample is sucked through a capillary tube by a high pressure jet of gas flowing around the tip of the capillary (Bennoulli effect). – This is also referred to aspiration. The high velocity breaks the sample into a mist and carries it to the atomization region. Prof. Dr. Hisham Ezzat Abdellatef 30
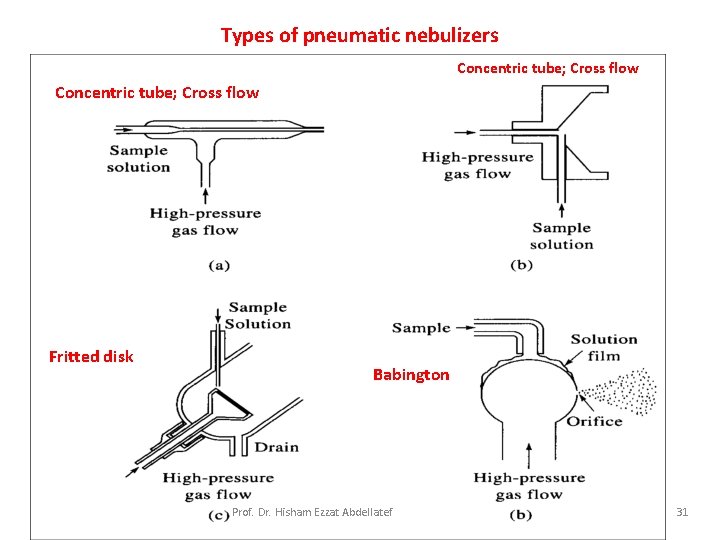
Types of pneumatic nebulizers Concentric tube; Cross flow Fritted disk Babington Prof. Dr. Hisham Ezzat Abdellatef 31

• Cross-flow The jet stream flows at right angles to the capillary tip. The sample is sometimes pumped through the capillary. • Fritted disk The sample is pumped onto a fritted disk through which the gas jet is flowing. Gives a finer aerosol than the others. • Babington Jet is pumped through a small orifice in a sphere on which a thin film of sample flows. This type is less prone to clogging and used for high salt content samples. • Ultrasonic Nebulizer • The sample is pumped onto the surface of a vibrating piezoelectric crystal. • The resulting mist is denser and more homogeneous than pneumatic nebulizers. • Electro-thermal Vaporizers (Etv) An electro thermal vaporizer contains an evaporator in a closed chamber through which an inert gas carries the vaporized sample into the atomizer. Prof. Dr. Hisham Ezzat Abdellatef 32
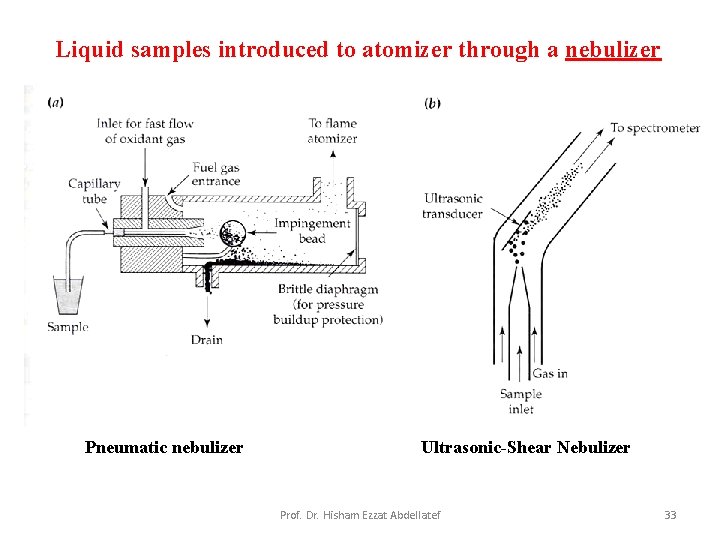
Liquid samples introduced to atomizer through a nebulizer Pneumatic nebulizer Ultrasonic-Shear Nebulizer Prof. Dr. Hisham Ezzat Abdellatef 33

Atomization Atomizers Flame Electrothermal Special Glow Discharge Hydride Generation Cold-Vapor Prof. Dr. Hisham Ezzat Abdellatef 34

Flame Chemistry Flames are used in atomic emission spectrometry for excitation (emission spectrometry) but in atomic absorption flames are used as Atom Cells to produce gaseous atoms. Why must the atoms not be excited for atomic absorption spectrometry? If the atom is already in the excited state it cannot absorb the light. Prof. Dr. Hisham Ezzat Abdellatef 35
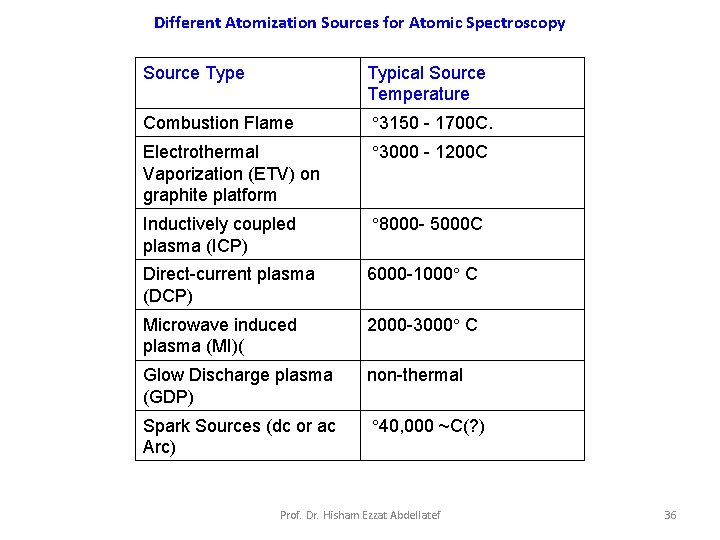
Different Atomization Sources for Atomic Spectroscopy Source Typical Source Temperature Combustion Flame ° 3150 - 1700 C. Electrothermal Vaporization (ETV) on graphite platform ° 3000 - 1200 C Inductively coupled plasma (ICP) ° 8000 - 5000 C Direct-current plasma (DCP) 6000 -1000° C Microwave induced plasma (MI)( 2000 -3000° C Glow Discharge plasma (GDP) non-thermal Spark Sources (dc or ac Arc) ° 40, 000 ~C(? ) Prof. Dr. Hisham Ezzat Abdellatef 36

Flame Atomizers • Superior method for reproducible liquid sample introduction for atomic absorption and fluorescence spectroscopy. • Other methods better in terms of sampling efficiency and sensitivity. Prof. Dr. Hisham Ezzat Abdellatef 37
Laminar-Flow Burner Prof. Dr. Hisham Ezzat Abdellatef 38
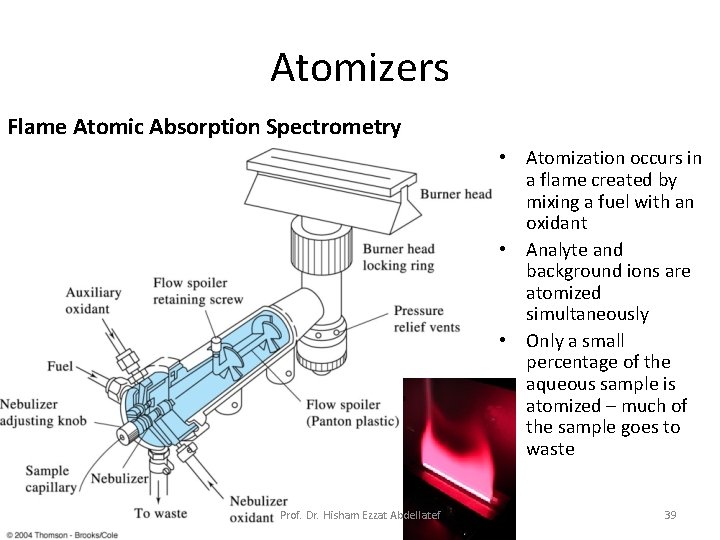
Atomizers Flame Atomic Absorption Spectrometry • Atomization occurs in a flame created by mixing a fuel with an oxidant • Analyte and background ions are atomized simultaneously • Only a small percentage of the aqueous sample is atomized – much of the sample goes to waste Prof. Dr. Hisham Ezzat Abdellatef 39
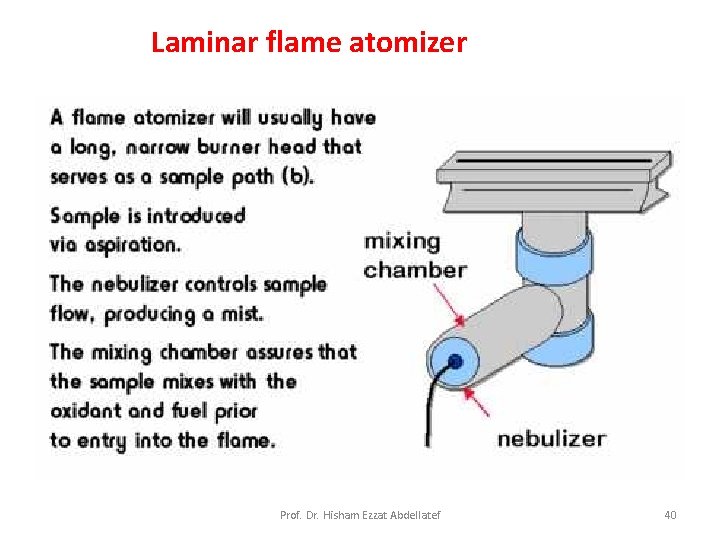
Laminar flame atomizer Prof. Dr. Hisham Ezzat Abdellatef 40

Prof. Dr. Hisham Ezzat Abdellatef 41

Prof. Dr. Hisham Ezzat Abdellatef 42

Performance Characteristics Of Flame Atomizers In terms of reproducible behavior, flame atomization appears to be superior to all other methods for liquid sample introduction. In terms of sampling efficiency and thus sensitivity, however, other atomization methods are markedly better. A large portion of the sample flows down the drain and the residence time of individual atoms in the optical path in the flame is brief (~10 -4 s). Prof. Dr. Hisham Ezzat Abdellatef 43

Electrothermal Atomization • Atomization of entire sample in short period • Average sample time in optical path is seconds – Evaporation of sample • Microliter volume • Low temperature – Sample ashed at higher temperature – Increase current • Sample temperature goes to 2000 -3000 °C – Sample measured above heated surface • High sensitivity for small samples Prof. Dr. Hisham Ezzat Abdellatef 44

Electrothermal Atomization It provides enhanced sensitivity because the entire sample is atomized in a short period, and the average residence time of the atoms in the optical path is a second or more. A few microliters of sample are first evaporated at a low temperature and then ashed at a somewhat higher temperature in an electrically heated graphite tube or in a graphite cup. Then the current is rapidly increased to several hundred amperes, which caused the temperature to soar to perhaps 2000 o. C to 3000 o. C; atomization of the sample occurs in a period of a few milliseconds to seconds. Prof. Dr. Hisham Ezzat Abdellatef 45
Prof. Dr. Hisham Ezzat Abdellatef 46
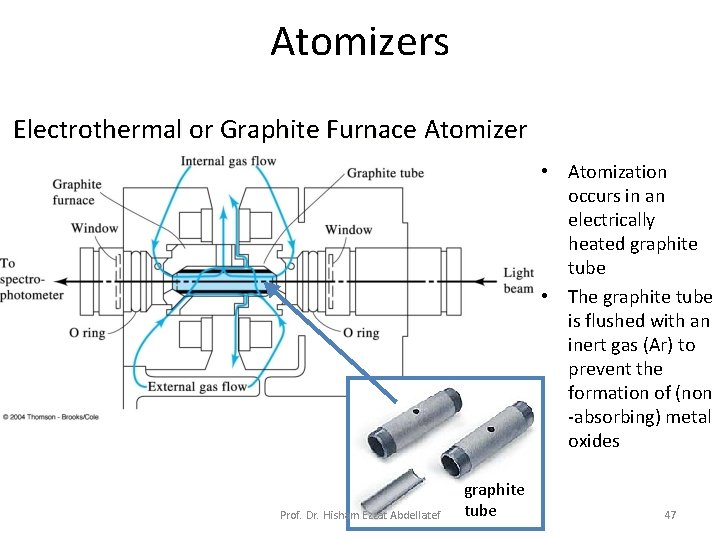
Atomizers Electrothermal or Graphite Furnace Atomizer • Atomization occurs in an electrically heated graphite tube • The graphite tube is flushed with an inert gas (Ar) to prevent the formation of (non -absorbing) metal oxides Prof. Dr. Hisham Ezzat Abdellatef graphite tube 47

Performance Characteristics: Electrothermal atomizers offer the advantage of unusually high sensitivity for small volumes of sample. Typically, sample volumes between 0. 5 and 10 L are used; absolute detection limits lie in the range of 10 -10 to 10 -13 g of analyte. Furnace methods are slow-typically requiring several minutes per element. A final disadvantage is that the analytical range is low, being usually less than two orders of magnitude. Prof. Dr. Hisham Ezzat Abdellatef 48
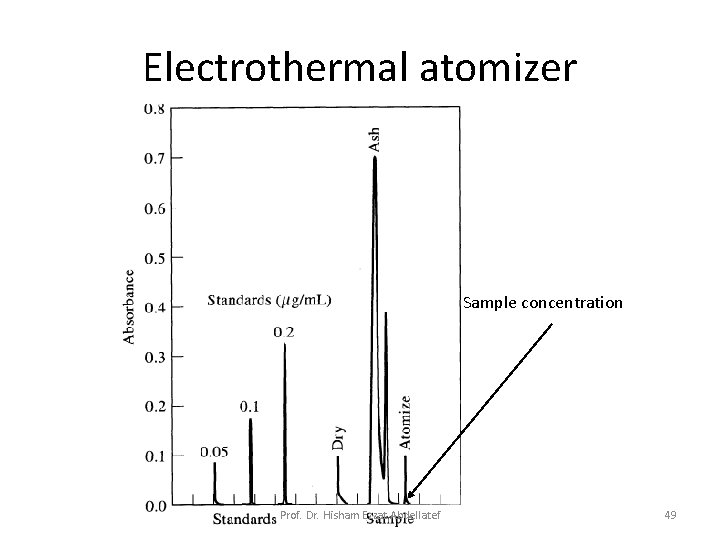
Electrothermal atomizer Sample concentration Prof. Dr. Hisham Ezzat Abdellatef 49
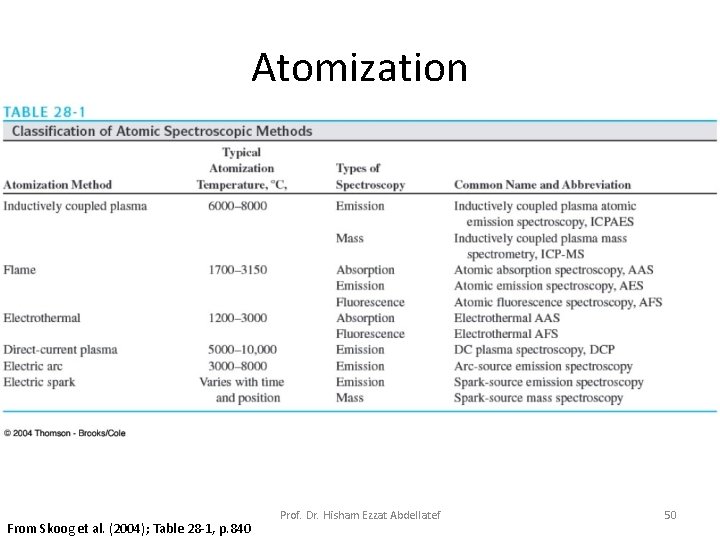
Atomization From Skoog et al. (2004); Table 28 -1, p. 840 Prof. Dr. Hisham Ezzat Abdellatef 50
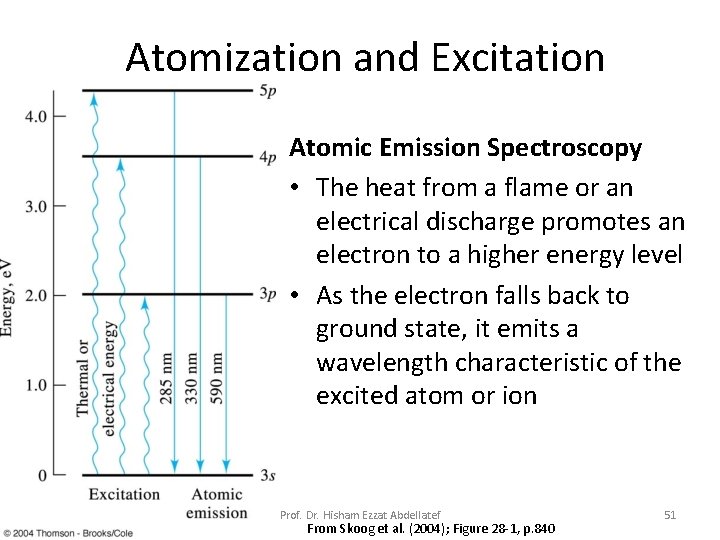
Atomization and Excitation Atomic Emission Spectroscopy • The heat from a flame or an electrical discharge promotes an electron to a higher energy level • As the electron falls back to ground state, it emits a wavelength characteristic of the excited atom or ion Prof. Dr. Hisham Ezzat Abdellatef From Skoog et al. (2004); Figure 28 -1, p. 840 51
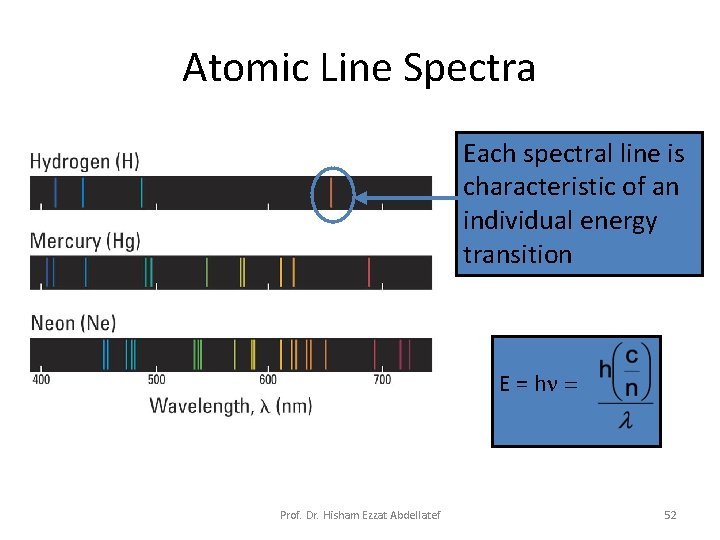
Atomic Line Spectra Each spectral line is characteristic of an individual energy transition E = hn = Prof. Dr. Hisham Ezzat Abdellatef 52
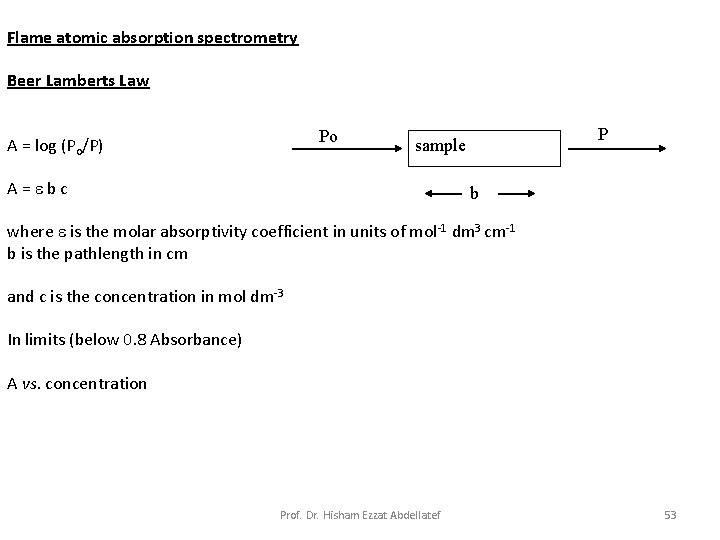
Flame atomic absorption spectrometry Beer Lamberts Law Po A = log (Po/P) sample A = b c b where is the molar absorptivity coefficient in units of mol-1 dm 3 cm-1 b is the pathlength in cm P and c is the concentration in mol dm-3 In limits (below 0. 8 Absorbance) A vs. concentration Prof. Dr. Hisham Ezzat Abdellatef 53
- Slides: 53