Part 2 9 Electronic Transitions Outline Absorption spectroscopy

Part 2. 9: Electronic Transitions

Outline • Absorption spectroscopy • Types of transitions – atomic – molecular • • • d-d transitions Transition moment Microstates Correlation diagrams Tanabe-Sugano diagrams Selection rules
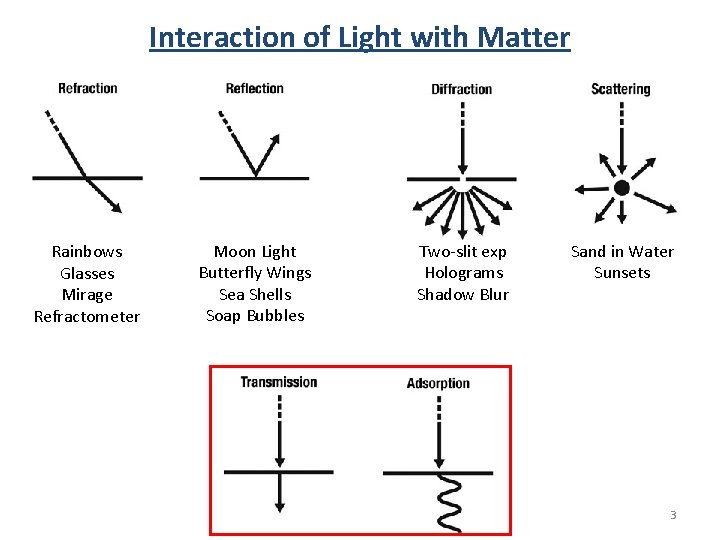
Interaction of Light with Matter Rainbows Glasses Mirage Refractometer Moon Light Butterfly Wings Sea Shells Soap Bubbles Two-slit exp Holograms Shadow Blur Sand in Water Sunsets 3
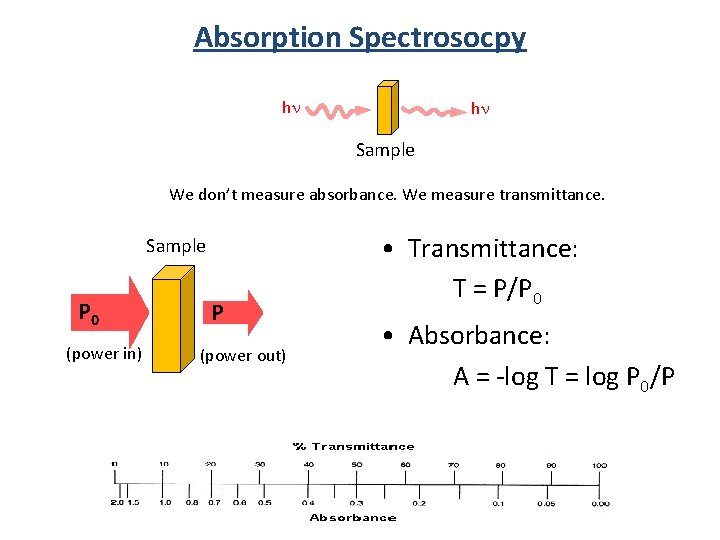
Absorption Spectrosocpy hn hn Sample We don’t measure absorbance. We measure transmittance. Sample P 0 (power in) P (power out) • Transmittance: T = P/P 0 • Absorbance: A = -log T = log P 0/P
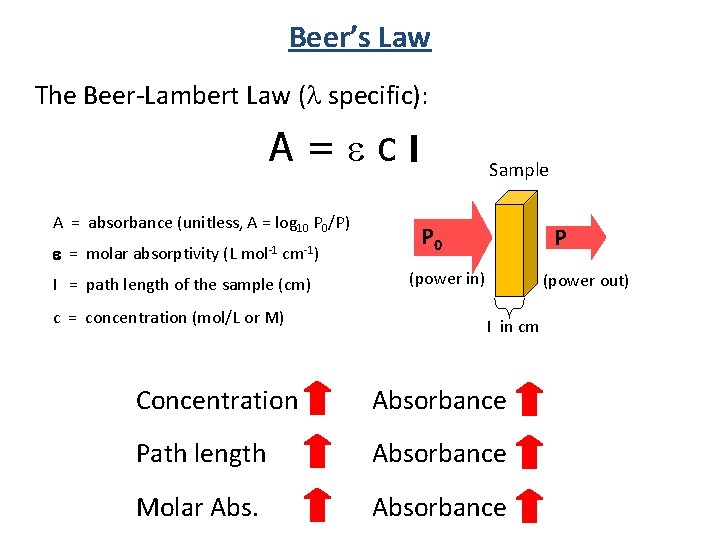
Beer’s Law The Beer-Lambert Law ( specific): A=ecl A = absorbance (unitless, A = log 10 P 0/P) e = molar absorptivity (L mol-1 cm-1) l = path length of the sample (cm) c = concentration (mol/L or M) Sample P 0 P (power in) (power out) l in cm Concentration Absorbance Path length Absorbance Molar Absorbance
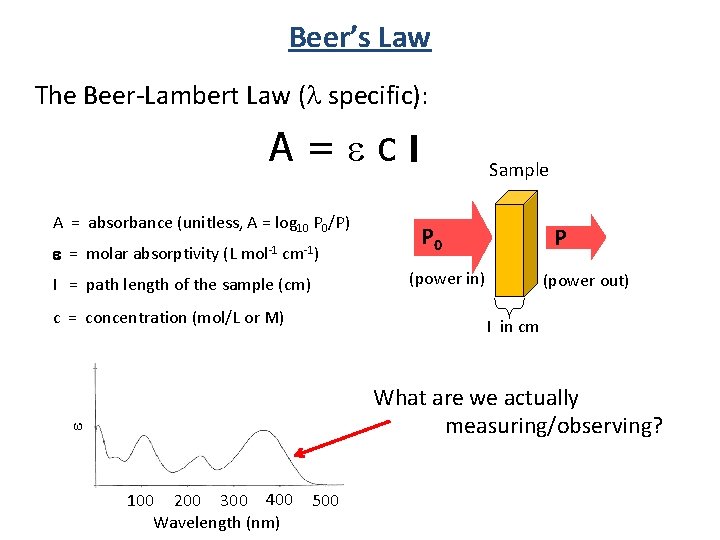
Beer’s Law The Beer-Lambert Law ( specific): A=ecl A = absorbance (unitless, A = log 10 P 0/P) e = molar absorptivity (L mol-1 cm-1) Sample P 0 P (power in) l = path length of the sample (cm) c = concentration (mol/L or M) (power out) l in cm e What are we actually measuring/observing? 100 200 300 400 Wavelength (nm) 500
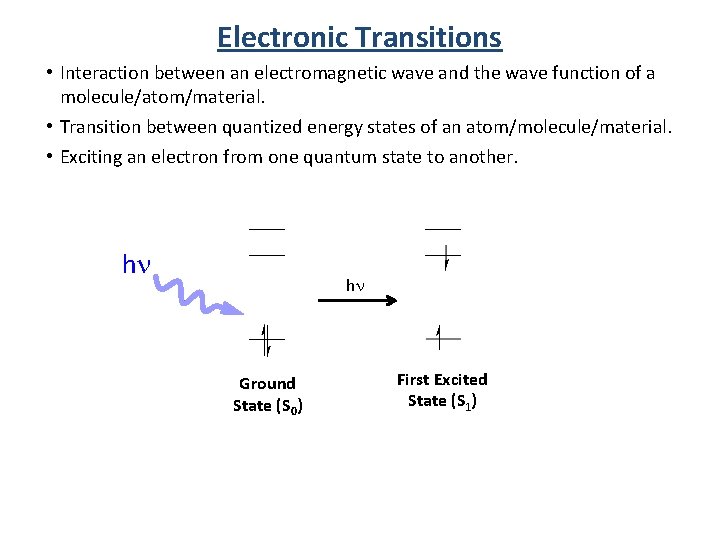
Electronic Transitions • Interaction between an electromagnetic wave and the wave function of a molecule/atom/material. • Transition between quantized energy states of an atom/molecule/material. • Exciting an electron from one quantum state to another. hn hn Ground State (S 0) First Excited State (S 1)

Electronic Transitions e Spectral Features: • Number of transitions. • Energy of the transitions. • Intensity of the transitions. 100 200 300 Wavelength (nm) 400 500
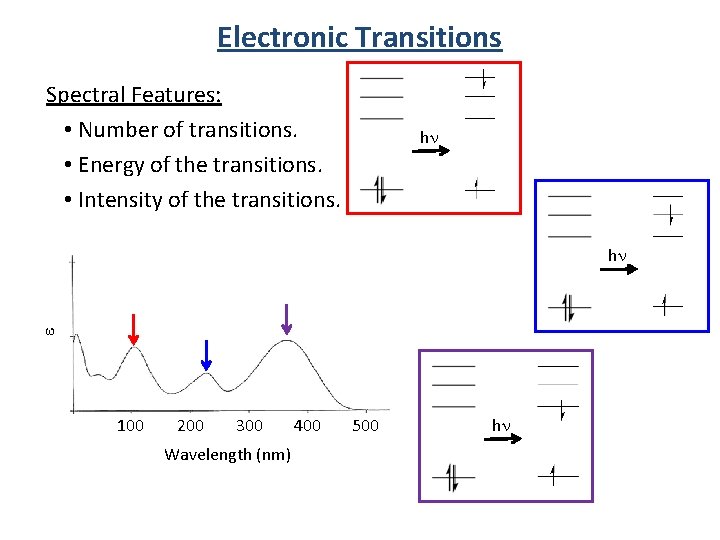
Electronic Transitions Spectral Features: • Number of transitions. • Energy of the transitions. • Intensity of the transitions. hn e hn 100 200 300 Wavelength (nm) 400 500 hn
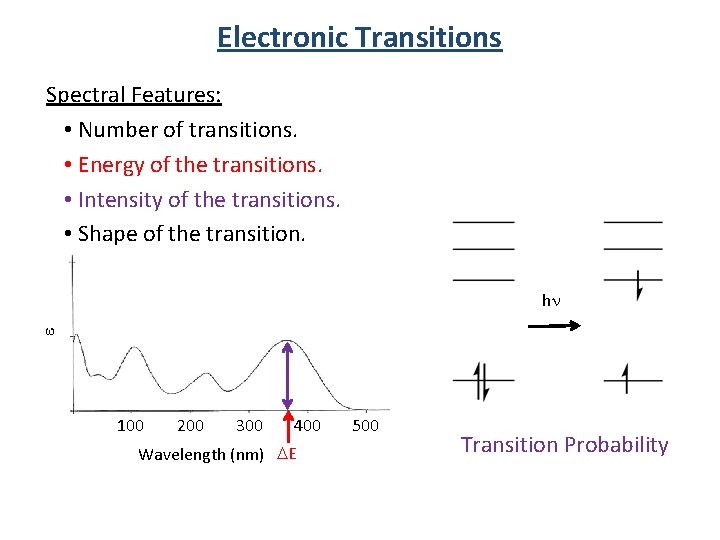
Electronic Transitions Spectral Features: • Number of transitions. • Energy of the transitions. • Intensity of the transitions. • Shape of the transition. e hn 100 200 300 400 Wavelength (nm) DE 500 Transition Probability
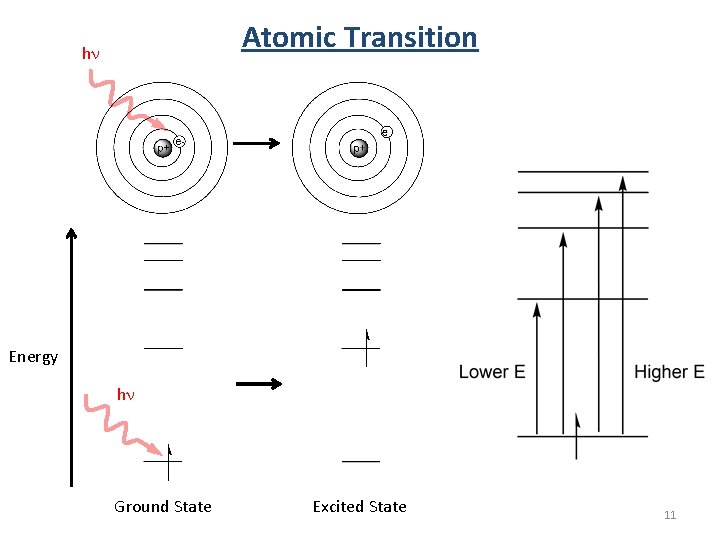
Atomic Transition hn Energy hn Ground State Excited State 11
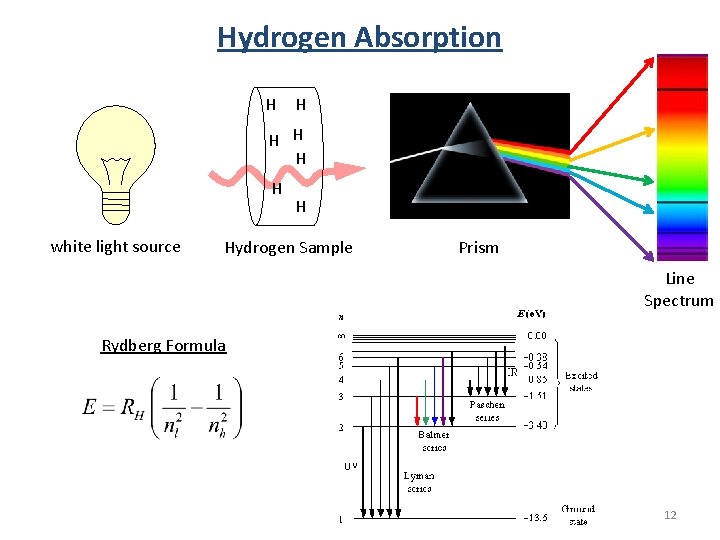
Hydrogen Absorption H H H white light source H Hydrogen Sample Prism Line Spectrum Rydberg Formula 12
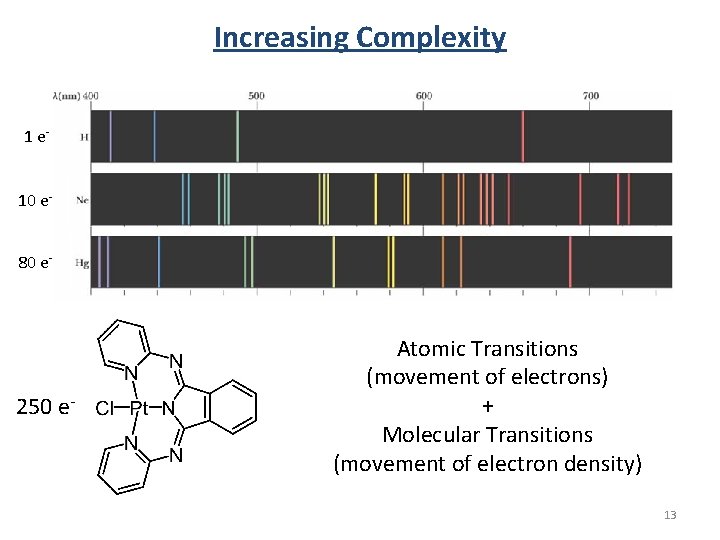
Increasing Complexity 1 e 10 e 80 e- 250 e- Atomic Transitions (movement of electrons) + Molecular Transitions (movement of electron density) 13
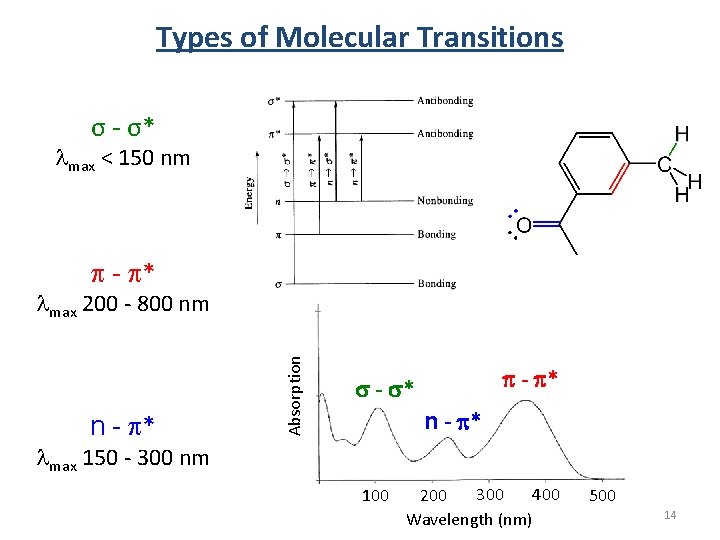
Types of Molecular Transitions σ - σ* max < 150 nm p - p* n - p* Absorption max 200 - 800 nm p - p* s - s* n - p* max 150 - 300 nm 100 400 300 200 Wavelength (nm) 500 14
![Types of Molecular Transitions [Co(H 2 O)6 ]2+ Metal Centered (MC) Focus on Metal Types of Molecular Transitions [Co(H 2 O)6 ]2+ Metal Centered (MC) Focus on Metal](http://slidetodoc.com/presentation_image/3b9c435c2687c5aa22e0c8fb76f66f33/image-15.jpg)
Types of Molecular Transitions [Co(H 2 O)6 ]2+ Metal Centered (MC) Focus on Metal Centered Transitions max 200 – 800 nm Mn. O 4 MLCT max 300 – 1000 nm LMCT max 300 – 1000 nm MMCT max 300 – 800 nm 15

Colors of Metal Ions Alexandrite Cr 3+ doped Be. Al 2 O 4
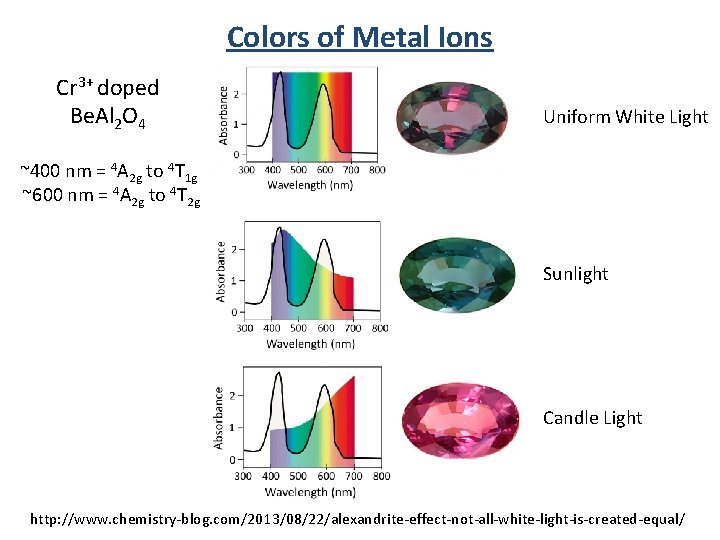
Colors of Metal Ions Cr 3+ doped Be. Al 2 O 4 Uniform White Light ~400 nm = 4 A 2 g to 4 T 1 g ~600 nm = 4 A 2 g to 4 T 2 g Sunlight Candle Light http: //www. chemistry-blog. com/2013/08/22/alexandrite-effect-not-all-white-light-is-created-equal/

Colors of Metal Ions Ruby ~1% Cr 3+ doped Al 2 O 3 Absorbs yellow-green region Emits red Most expensive ruby (1. 6 cm 3) = $6. 7 million Al 2 O 3 (1. 5 cm 3) = ~$500
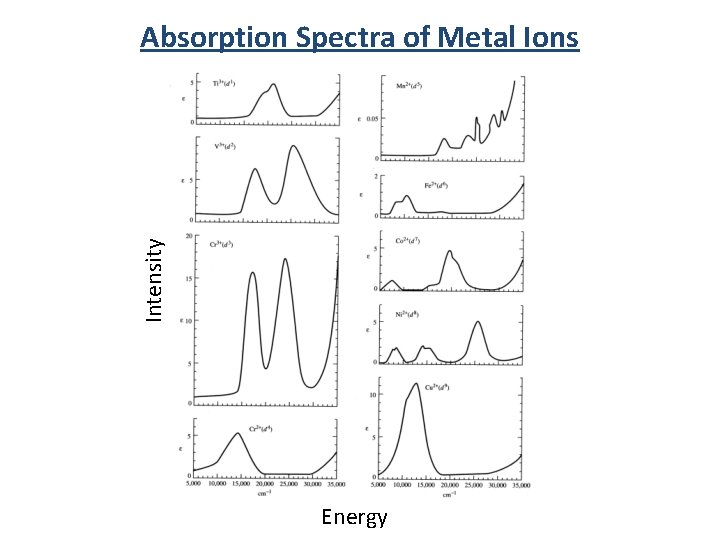
Intensity Absorption Spectra of Metal Ions Energy

Electronic Transitions A=ecl e A = absorbance (unitless, A = log 10 P 0/P) e = molar absorptivity (L mol-1 cm-1) 100 200 300 400 Wavelength (nm) 500 l = path length of the sample (cm) c = concentration (mol/L or M) Transition probability –the probability of a particular transition taking place. Depends on: 1) Energy of the transition/incident light. 2) Orientation of the molecule/material. 3) Symmetry of the initial and final states. 4) Angular momentum (spin).
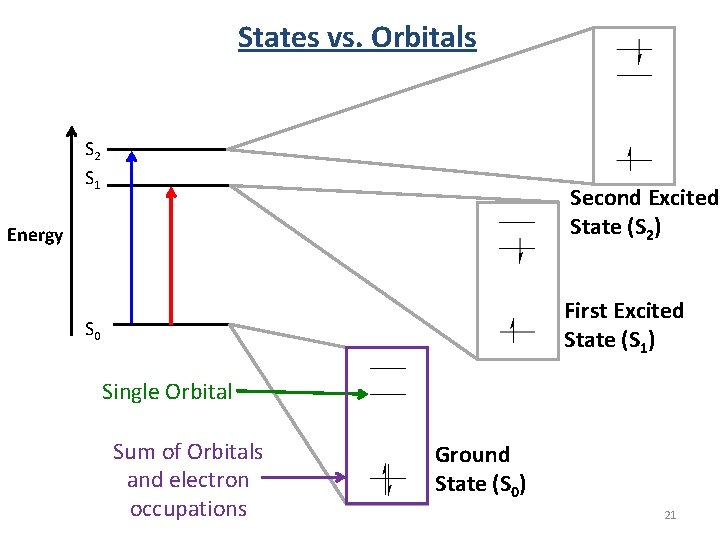
States vs. Orbitals S 2 S 1 Second Excited State (S 2) Energy First Excited State (S 1) S 0 Single Orbital Sum of Orbitals and electron occupations Ground State (S 0) 21
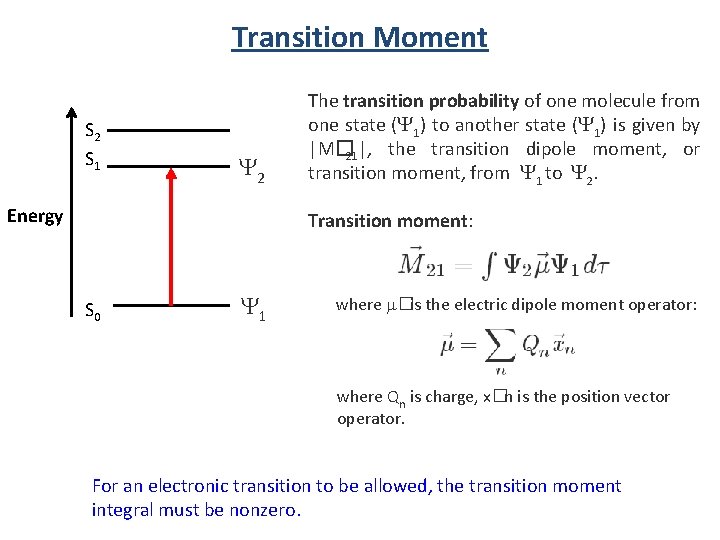
Transition Moment S 2 S 1 Y 2 Energy The transition probability of one molecule from one state (Y 1) to another state (Y 1) is given by |M� 21|, the transition dipole moment, or transition moment, from Y 1 to Y 2. Transition moment: S 0 Y 1 where m� is the electric dipole moment operator: where Qn is charge, x� n is the position vector operator. For an electronic transition to be allowed, the transition moment integral must be nonzero.
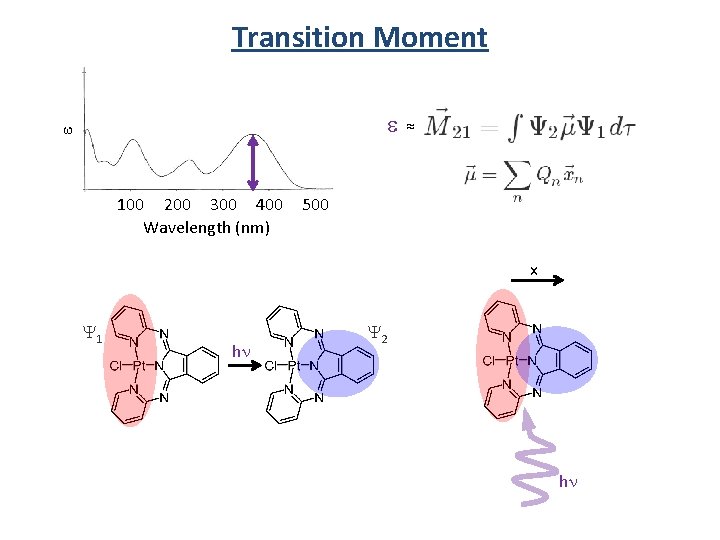
Transition Moment e e 100 200 300 400 Wavelength (nm) ≈ 500 x Y 1 hn Y 2 hn
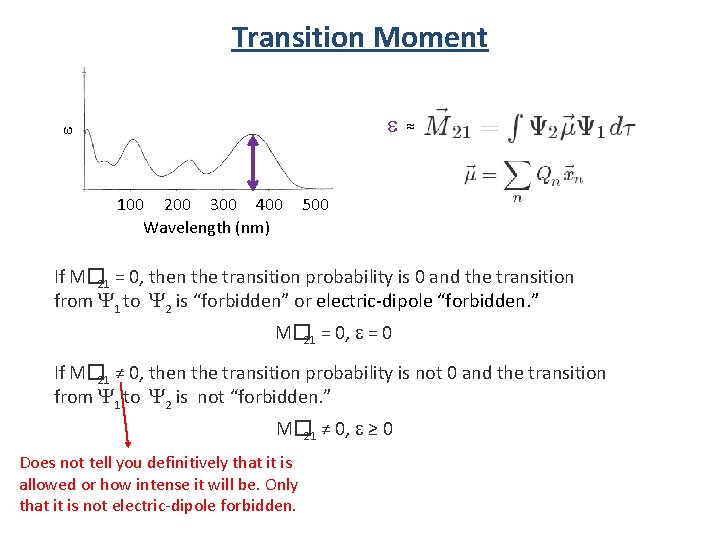
Transition Moment e e 100 200 300 400 Wavelength (nm) ≈ 500 If M� 21 = 0, then the transition probability is 0 and the transition from Y 1 to Y 2 is “forbidden” or electric-dipole “forbidden. ” M� 21 = 0, e = 0 If M� 21 ≠ 0, then the transition probability is not 0 and the transition from Y 1 to Y 2 is not “forbidden. ” M� 21 ≠ 0, e ≥ 0 Does not tell you definitively that it is allowed or how intense it will be. Only that it is not electric-dipole forbidden.
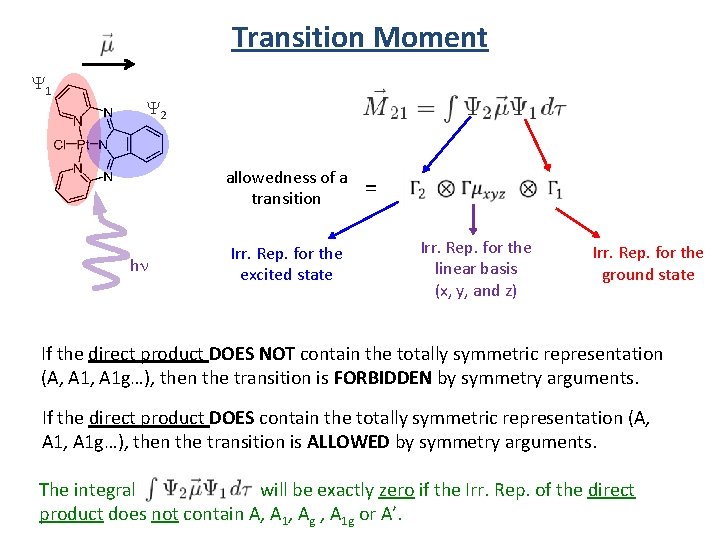
Transition Moment Y 1 Y 2 allowedness of a transition hn Irr. Rep. for the excited state = Irr. Rep. for the linear basis (x, y, and z) Irr. Rep. for the ground state If the direct product DOES NOT contain the totally symmetric representation (A, A 1 g…), then the transition is FORBIDDEN by symmetry arguments. If the direct product DOES contain the totally symmetric representation (A, A 1, A 1 g…), then the transition is ALLOWED by symmetry arguments. The integral will be exactly zero if the Irr. Rep. of the direct product does not contain A, A 1, Ag , A 1 g or A’.

Direct Product Direct product: The representation of the product of two representations is given by the product of the characters of the two representations. 26
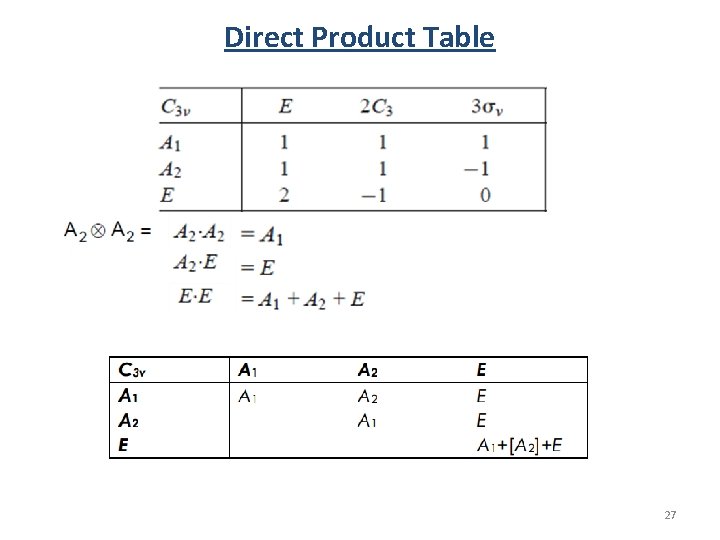
Direct Product Table 27
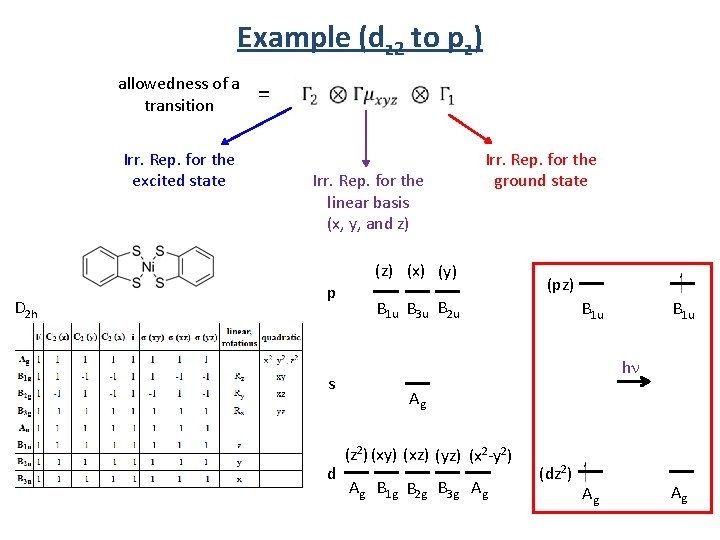
Example (dz 2 to pz) allowedness of a transition Irr. Rep. for the excited state D 2 h = Irr. Rep. for the linear basis (x, y, and z) p s d Irr. Rep. for the ground state (z) (x) (y) (pz) B 1 u B 3 u B 2 u B 1 u hn Ag (z 2) (xy) (xz) (yz) (x 2 -y 2) Ag B 1 g B 2 g B 3 g Ag (dz 2) Ag Ag
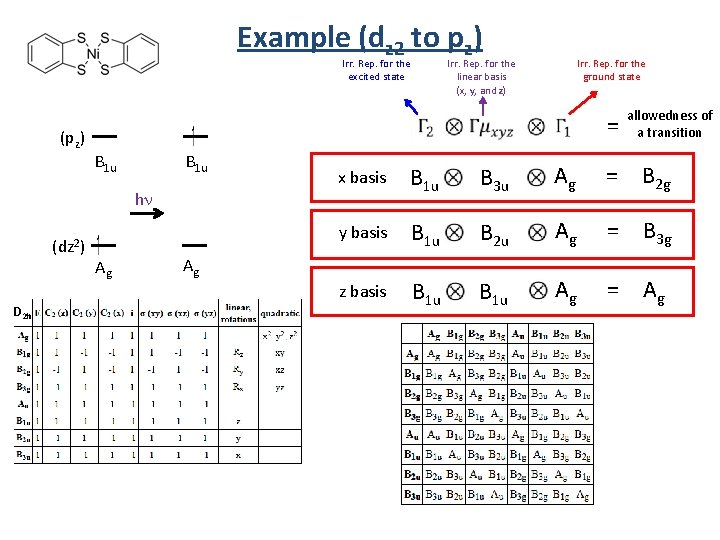
Example (dz 2 to pz) Irr. Rep. for the excited state (pz) = B 1 u hn (dz 2) D 2 h Irr. Rep. for the ground state Irr. Rep. for the linear basis (x, y, and z) Ag allowedness of a transition x basis B 1 u B 3 u Ag = B 2 g y basis B 1 u B 2 u Ag = B 3 g z basis B 1 u Ag = Ag Ag
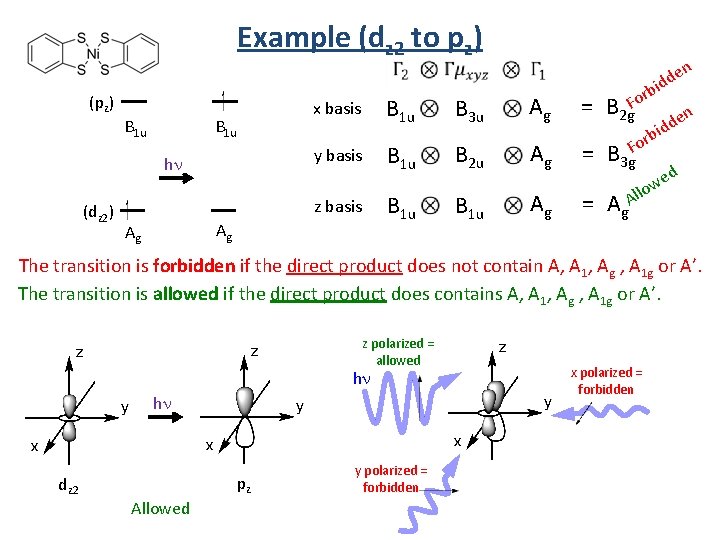
Example (dz 2 to pz) (pz) B 1 u y basis hn (dz 2) B 1 u x basis z basis Ag Ag B 3 u n Ag rb Fo = B 2 g e idd n e d d i rb Fo B 1 u B 2 u Ag = B 3 g B 1 u Ag = Ag. Al d e w o l The transition is forbidden if the direct product does not contain A, A 1, Ag , A 1 g or A’. The transition is allowed if the direct product does contains A, A 1, Ag , A 1 g or A’. z polarized = allowed hn hn dz 2 pz Allowed y polarized = forbidden x polarized = forbidden
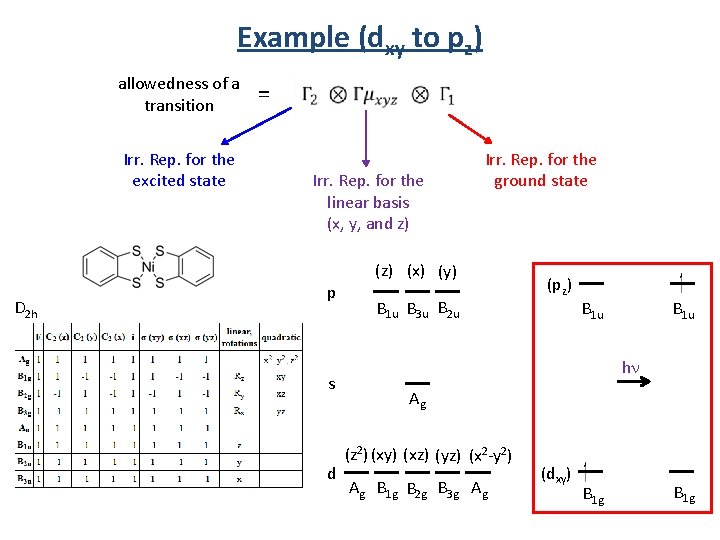
Example (dxy to pz) allowedness of a transition Irr. Rep. for the excited state D 2 h = Irr. Rep. for the linear basis (x, y, and z) p s d Irr. Rep. for the ground state (z) (x) (y) B 1 u B 3 u B 2 u (pz) B 1 u hn Ag (z 2) (xy) (xz) (yz) (x 2 -y 2) Ag B 1 g B 2 g B 3 g Ag (dxy) B 1 g
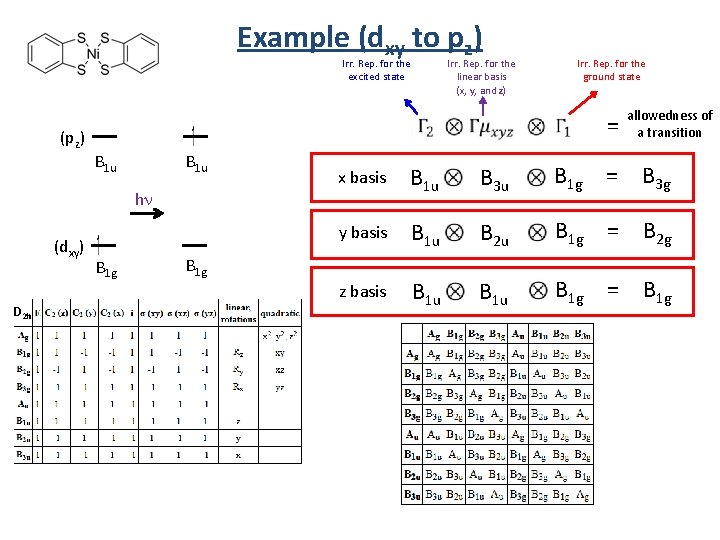
Example (dxy to pz) Irr. Rep. for the excited state (pz) D 2 h Irr. Rep. for the ground state = B 1 u hn (dxy) Irr. Rep. for the linear basis (x, y, and z) B 1 g allowedness of a transition x basis B 1 u B 3 u B 1 g = B 3 g y basis B 1 u B 2 u B 1 g = B 2 g z basis B 1 u B 1 g = B 1 g
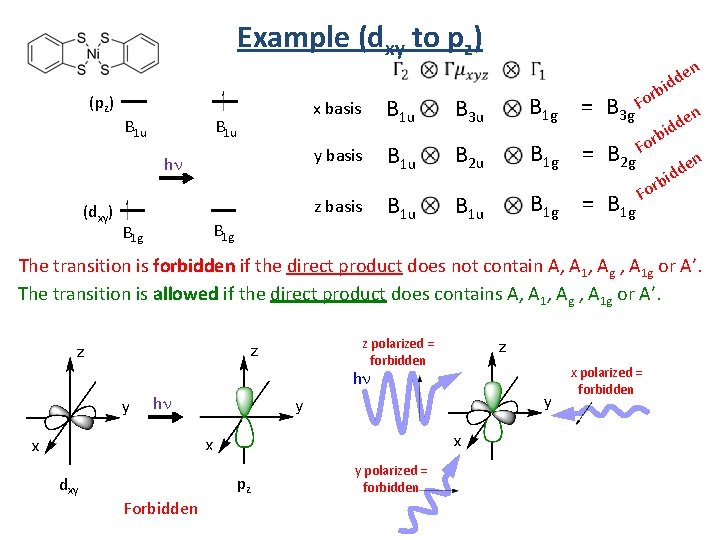
Example (dxy to pz) (pz) B 1 u y basis hn (dxy) B 1 u x basis z basis B 1 g B 3 u n rb B 1 g = B 3 g Fo Fo B 1 u B 2 u B 1 g = B 2 g B 1 u B 1 g = B 1 g e idd n e d d i rb rb o F n e idd The transition is forbidden if the direct product does not contain A, A 1, Ag , A 1 g or A’. The transition is allowed if the direct product does contains A, A 1, Ag , A 1 g or A’. z polarized = forbidden hn hn dxy pz Forbidden y polarized = forbidden x polarized = forbidden
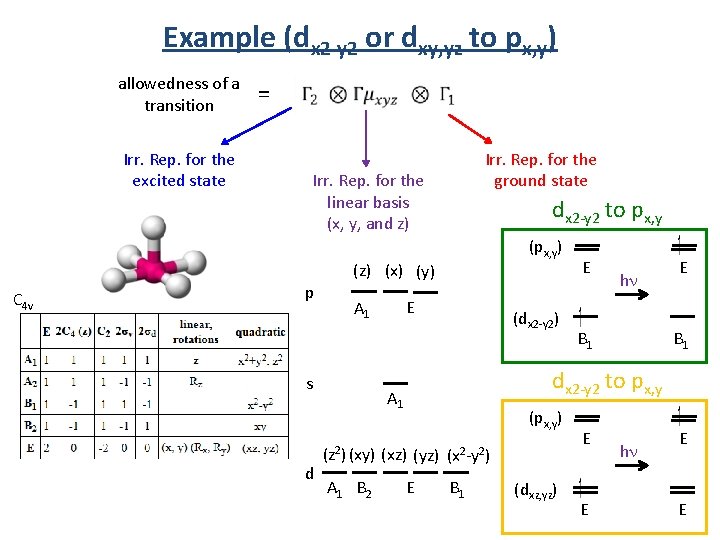
Example (dx 2 -y 2 or dxy, yz to px, y) allowedness of a transition Irr. Rep. for the excited state C 4 v = Irr. Rep. for the ground state Irr. Rep. for the linear basis (x, y, and z) p (px, y) (z) (x) (y) E A 1 s d dx 2 -y 2 to px, y (dx 2 -y 2) hn B 1 E B 1 dx 2 -y 2 to px, y A 1 (px, y) (z 2) (xy) (xz) (yz) (x 2 -y 2) A 1 B 2 E E B 1 (dxz, yz) E E hn E E
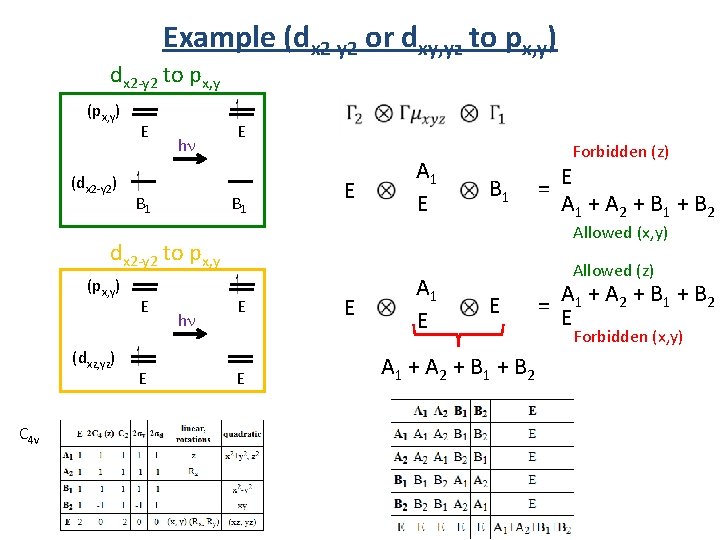
Example (dx 2 -y 2 or dxy, yz to px, y) dx 2 -y 2 to px, y (px, y) (dx 2 -y 2) E hn B 1 E A 1 E Forbidden (z) B 1 Allowed (x, y) dx 2 -y 2 to px, y (px, y) (dxz, yz) C 4 v E E hn = E A 1 + A 2 + B 1 + B 2 E E E A 1 E Allowed (z) E A 1 + A 2 + B 1 + B 2 = A E Forbidden (x, y)
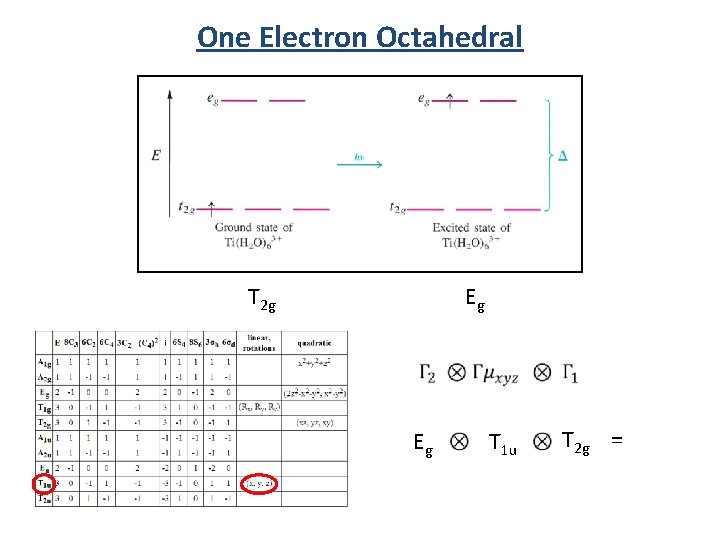
One Electron Octahedral Eg T 2 g Eg T 1 u T 2 g =

One Electron Octahedral Eg Eg Forbidden (x, y, z) T 1 u T 2 g A 2 u + Eu + T 1 u + T 2 u Eu + A 1 u + A 2 u + Eu + T 1 u + T 2 u

Six Electron Octahedral (Low spin) Ground State A 1 g Excited State T 2 g Eg T 1 g + T 2 g
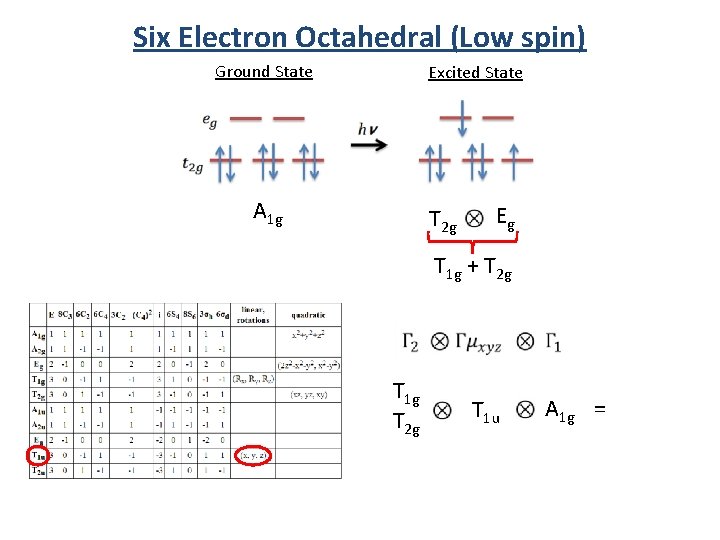
Six Electron Octahedral (Low spin) Ground State Excited State A 1 g T 2 g Eg T 1 g + T 2 g T 1 g T 2 g T 1 u A 1 g =
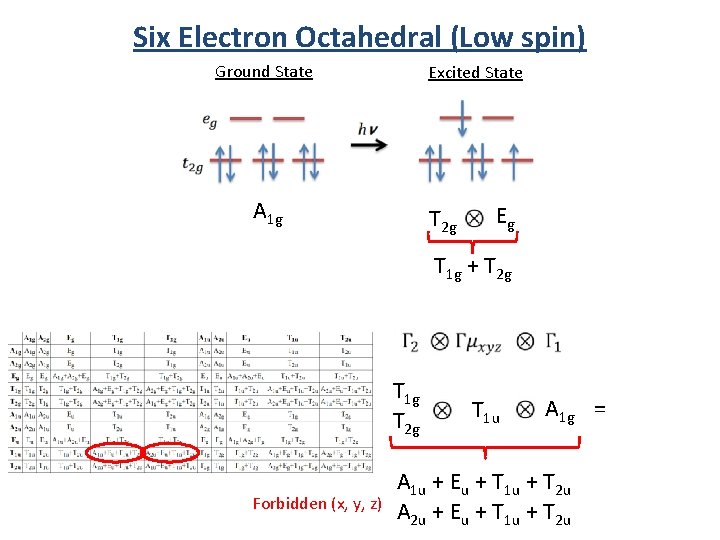
Six Electron Octahedral (Low spin) Ground State Excited State A 1 g T 2 g Eg T 1 g + T 2 g T 1 g T 2 g Forbidden (x, y, z) T 1 u A 1 g = A 1 u + Eu + T 1 u + T 2 u A 2 u + Eu + T 1 u + T 2 u
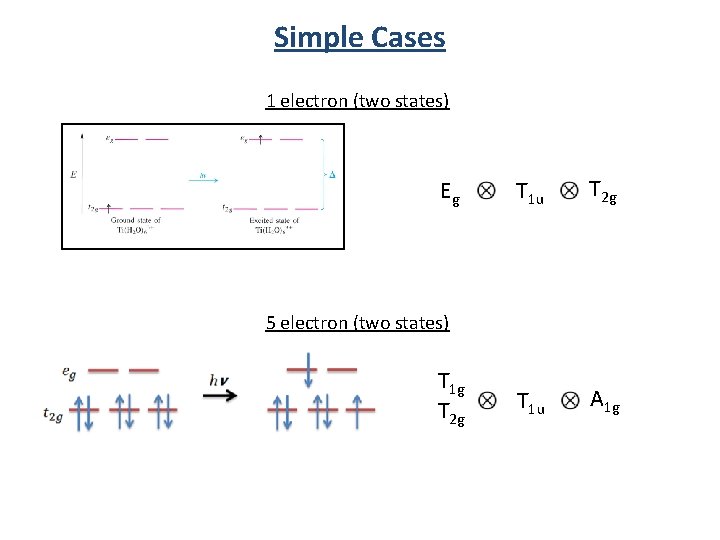
Simple Cases 1 electron (two states) Eg T 1 u T 2 g T 1 u A 1 g 5 electron (two states) T 1 g T 2 g
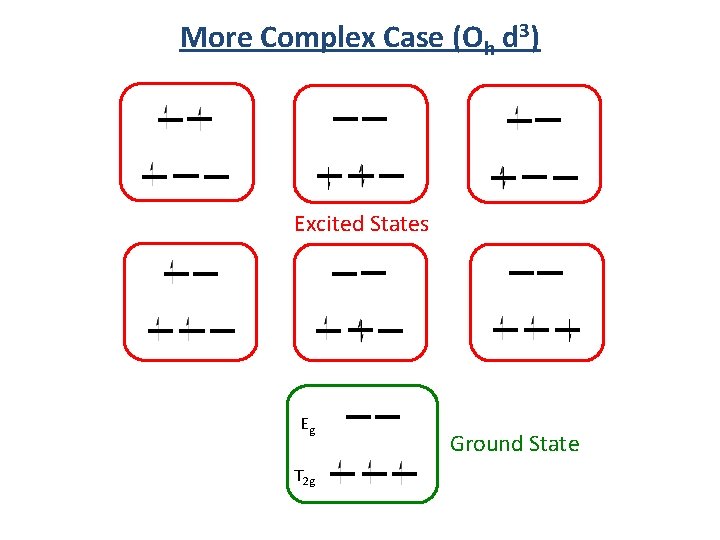
More Complex Case (Oh d 3) Excited States Eg T 2 g Ground State

More Complex Case (Oh d 2, d 3, d 2, d 8) There has to be an easier way to describe transitions between states!
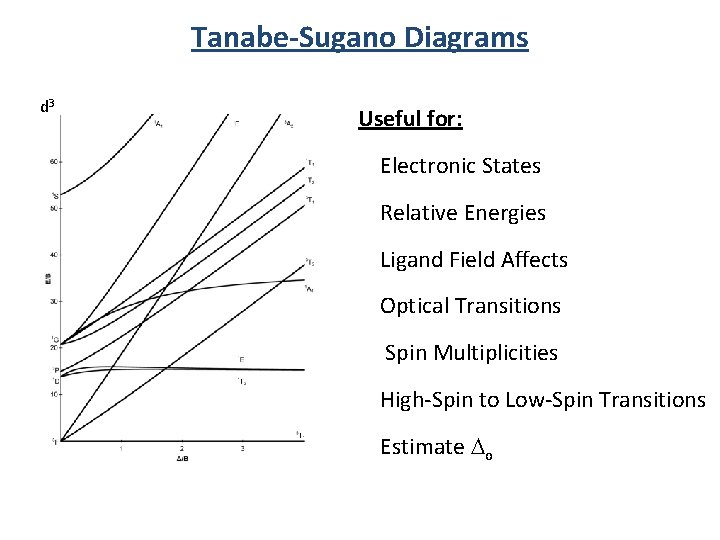
Tanabe-Sugano Diagrams d 3 Useful for: Electronic States Relative Energies Ligand Field Affects Optical Transitions Spin Multiplicities High-Spin to Low-Spin Transitions Estimate Do

Getting to Tanabe-Sugano Diagrams Electronic States Term symbols Microstate tables Correlation diagrams Tanabe-Sugano diagrams Selection rules

Quantum Numbers • PRINCIPAL (n): energy level, the distance the orbital is from the nucleus n = 1, 2, 3, 4… • ANGULAR MOMENTUM: l, shape of the orbital s = 0, p = 1, d = 2, f = 3 • MAGNETIC: ml , spatial orientation ml = 0 for s; -1, 0, +1 for p; -2, -1, 0, +1, +2 for d, etc. • SPIN: ms spin ms = +1/2 or -1/2)

Quantum Numbers F atom 1 s 2 s 2 p The third electron is in the 2 s orbital. n = 2 l = 0 ms= +1/2 The eighth electron is in a 2 p orbital. n = 2 l = 1 ml = -1 ms= -1/2 Only describes single electron states! What about multielectron states?

Many Electron States Many electron interactions are described by Russel-Saunders or L-S coupling scheme ML = total orbital angular momentum =Σml MS = total spin angular momentum = Σms Summarized by term symbols that contain: - spin multiplicity (2 S+1) - angular momentum quantum number (L) - the total angular momentum (J ) 2 S+1 L J The interactions produce atomic states called
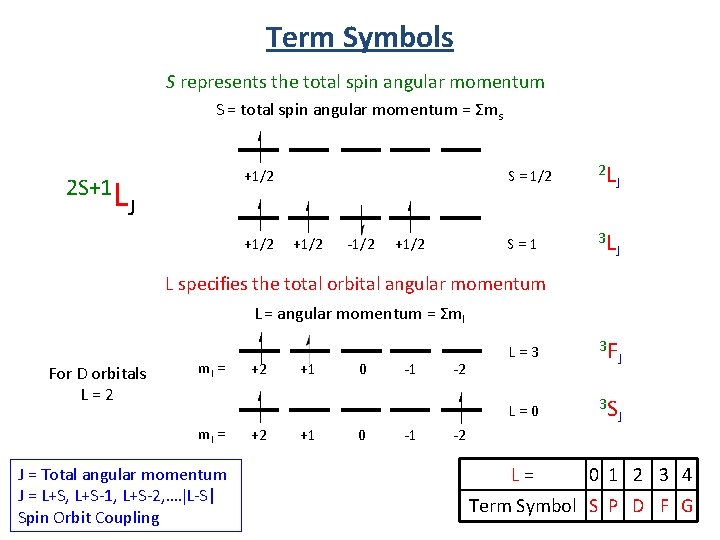
Term Symbols S represents the total spin angular momentum S = total spin angular momentum = Σms 2 S+1 L +1/2 S = 1/2 2 L J S = 1 3 L J J +1/2 -1/2 +1/2 L specifies the total orbital angular momentum L = angular momentum = Σml For D orbitals L = 2 ml = J = Total angular momentum J = L+S, L+S-1, L+S-2, …. L-S| Spin Orbit Coupling +2 +2 +1 +1 0 0 -1 -1 -2 L = 3 3 F J L = 0 3 S J -2 L = 0 1 2 3 4 Term Symbol S P D F G
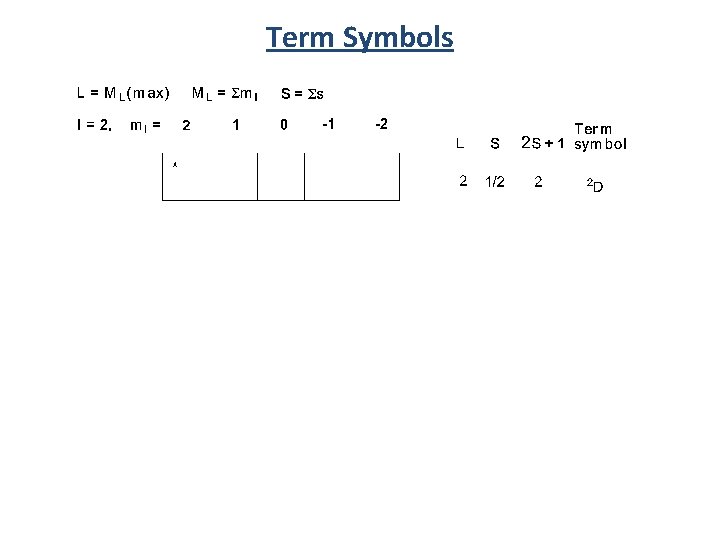
Term Symbols

Term Symbols We are only assigning one state at a time! To assign all the states we turn to a microstate table!
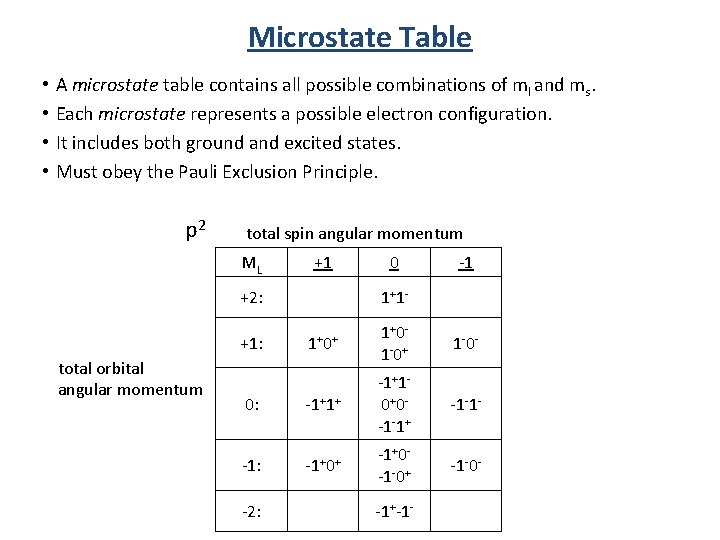
Microstate Table • • A microstate table contains all possible combinations of ml and ms. Each microstate represents a possible electron configuration. It includes both ground and excited states. Must obey the Pauli Exclusion Principle. p 2 total spin angular momentum ML +1 +2: 1+1 - -1 1+0+ 1+0 - 1 -0+ 1 -0 - 0: -1+1+ -1+1 - 0+0 - -1 -1+ -1 -1 - -1: -1+0+ -1+0 - -1 -0+ -1 -0 - +1: total orbital angular momentum 0 -2: -1+-1 -
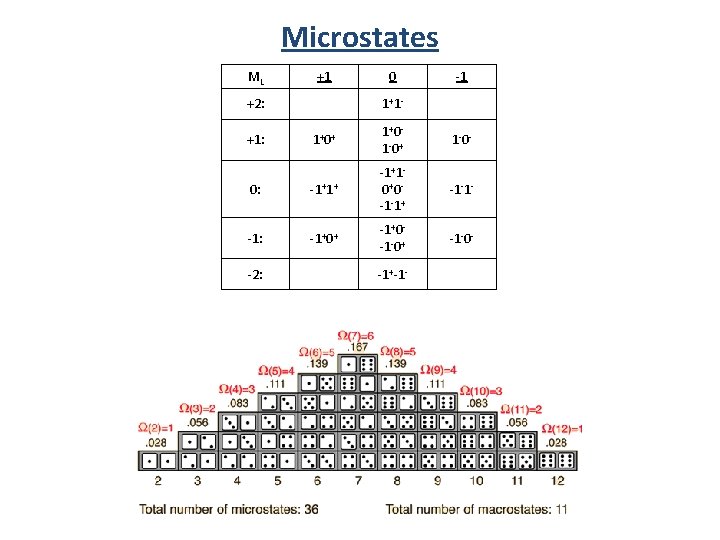
Microstates ML +1 +2: 0 1+ 1 - -1 1+ 0+ 1+ 0 - 1 - 0+ 1 - 0 - 0: -1+1+ -1+1 - 0+ 0 - -1 -1+ -1 -1 - -1: -1+0+ -1+0 - -1 -0+ -1 -0 - +1: -2: -1+-1 -
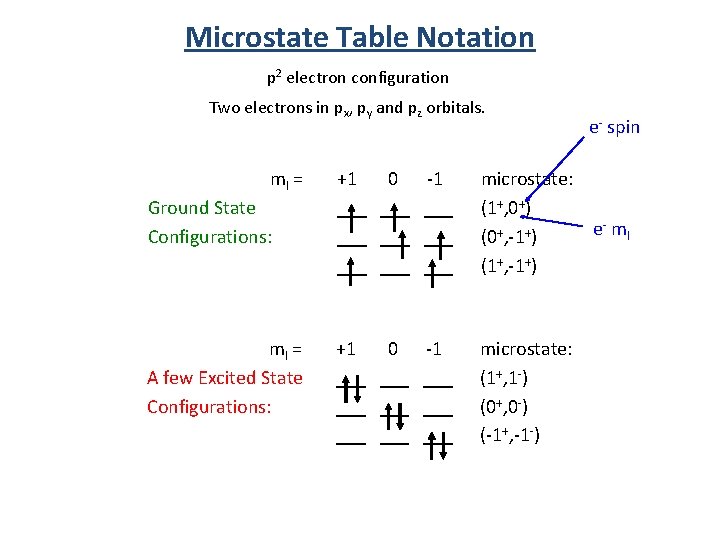
Microstate Table Notation p 2 electron configuration Two electrons in px, py and pz orbitals. e- spin ml = Ground State Configurations: +1 0 -1 ___ ___ ___ microstate: (1+, 0+) e- ml (0+, -1+) (1+, -1+) ml = A few Excited State Configurations: +1 0 -1 ___ ___ ___ microstate: (1+, 1 -) (0+, 0 -) (-1+, -1 -)
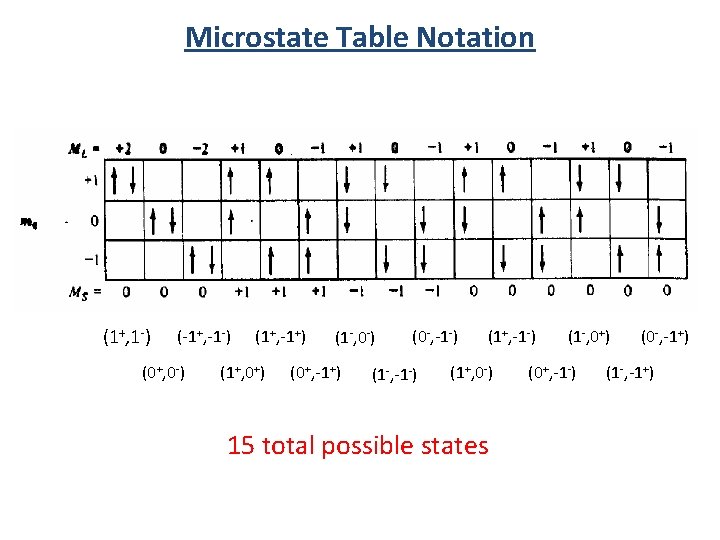
Microstate Table Notation (1+, 1 -) (-1+, -1 -) (0+, 0 -) (1+, -1+) (1+, 0+) (1 -, 0 -) (0+, -1+) (0 -, -1 -) (1+, 0 -) 15 total possible states (1 -, 0+) (0+, -1 -) (0 -, -1+) (1 -, -1+)
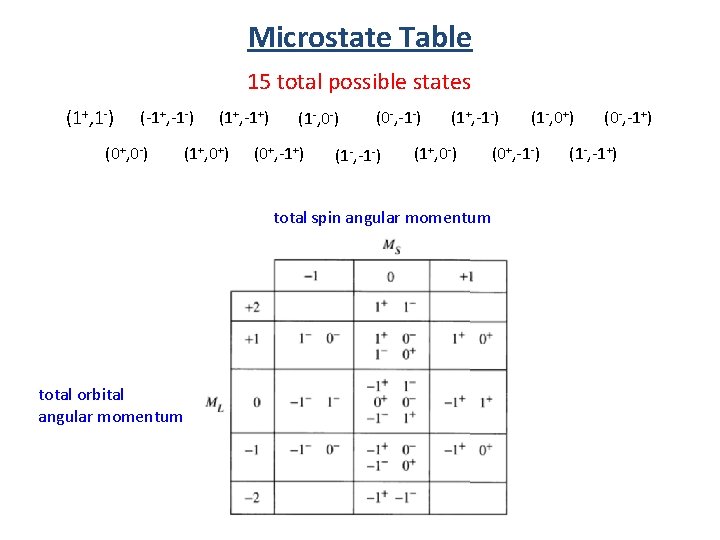
Microstate Table 15 total possible states (1+, 1 -) (-1+, -1 -) (0+, 0 -) (1+, -1+) (1+, 0+) (1 -, 0 -) (0+, -1+) (0 -, -1 -) (1+, 0 -) (0+, -1 -) total spin angular momentum total orbital angular momentum (1 -, 0+) (0 -, -1+) (1 -, -1+)
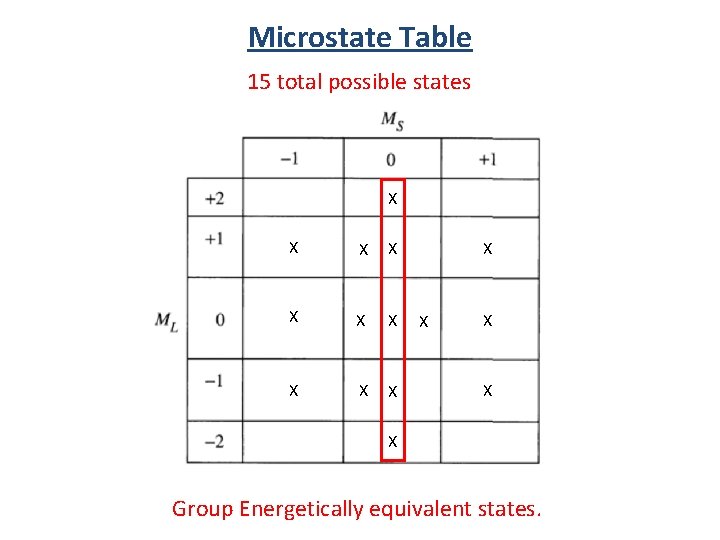
Microstate Table 15 total possible states X X X X Group Energetically equivalent states.
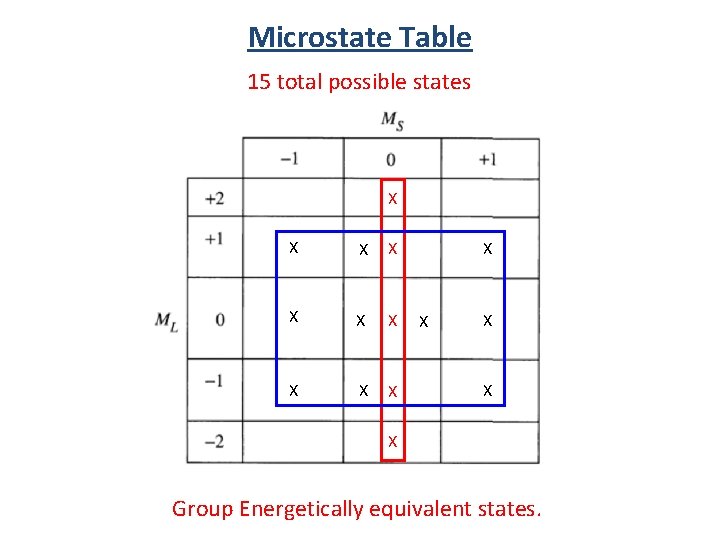
Microstate Table 15 total possible states X X X X Group Energetically equivalent states.
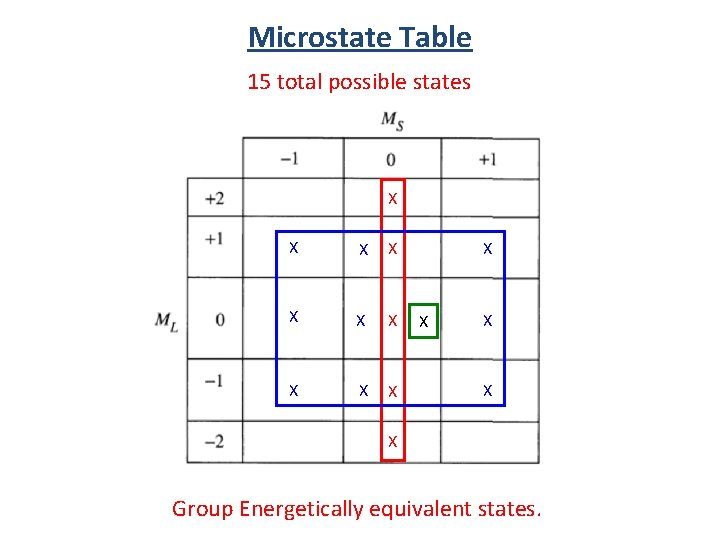
Microstate Table 15 total possible states X X X X Group Energetically equivalent states.
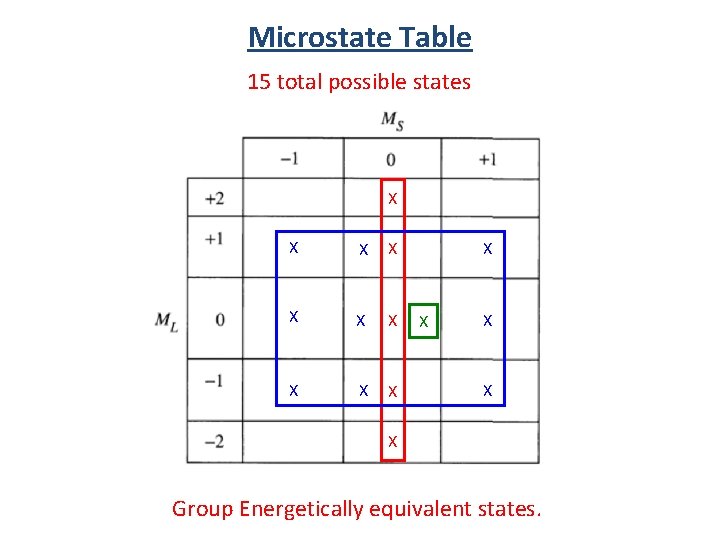
Microstate Table 15 total possible states X X X X Group Energetically equivalent states.
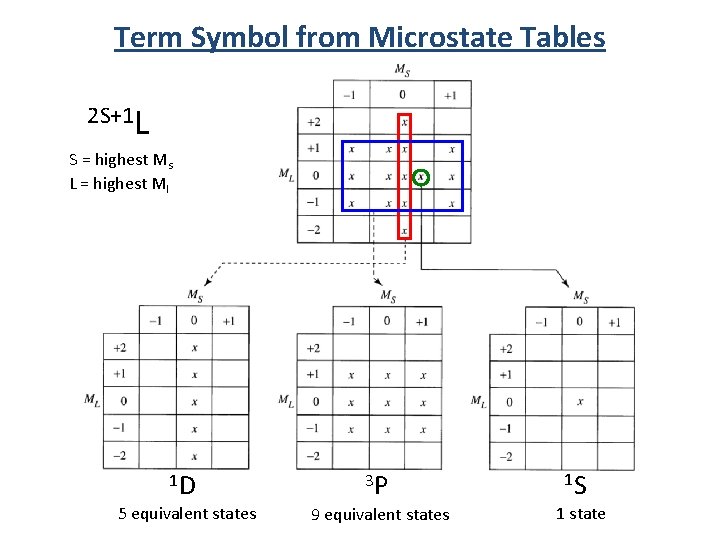
Term Symbol from Microstate Tables 2 S+1 L S = highest Ms L = highest Ml 1 D 3 P 5 equivalent states 9 equivalent states 1 S 1 state

Relative Energies 1 S 1 D 3 P 5 E equivalent states 9 E equivalent states 1 state 1. For a given electron configuration, the term with the greatest multiplicity lies lowest in energy. (Hund’s rule. ) 2. For a term of a given multiplicity, the greater the value of L, the lower the energy. Lowest E 3 P Highest E < 1 D < 1 S Note: The rules for predicting the ground state always work, but they may fail in predicting the order of energies for excited states.
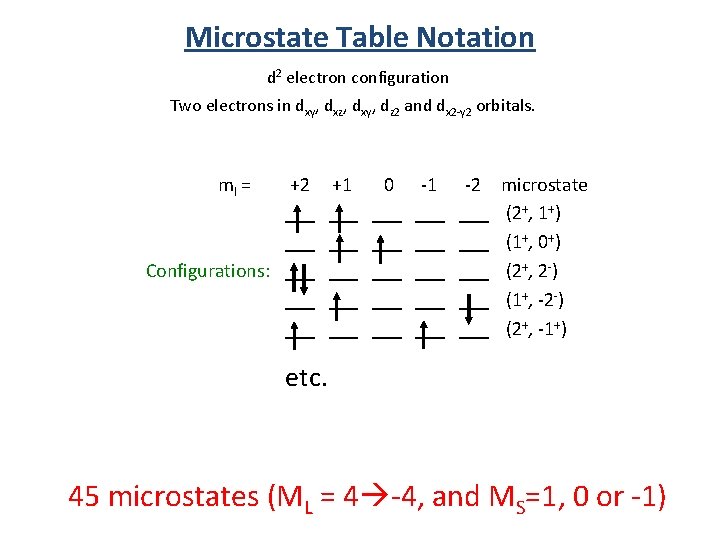
Microstate Table Notation d 2 electron configuration Two electrons in dxy, dxz, dxy, dz 2 and dx 2 -y 2 orbitals. ml = +2 +1 0 -1 -2 ___ ___ ___ Configurations: ___ ___ ___ ___ ___ microstate (2+, 1+) (1+, 0+) (2+, 2 -) (1+, -2 -) (2+, -1+) etc. 45 microstates (ML = 4 -4, and MS=1, 0 or -1)
Microstate Table Notation d 2 electron configuration Two electrons in dxy, dxz, dxy, dz 2 and dx 2 -y 2 orbitals.
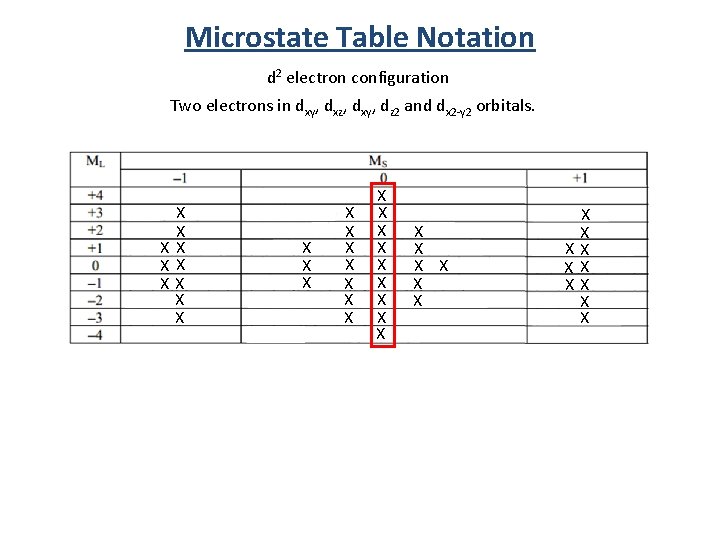
Microstate Table Notation d 2 electron configuration Two electrons in dxy, dxz, dxy, dz 2 and dx 2 -y 2 orbitals. X X XX X X X X X X X XX XX XX X X
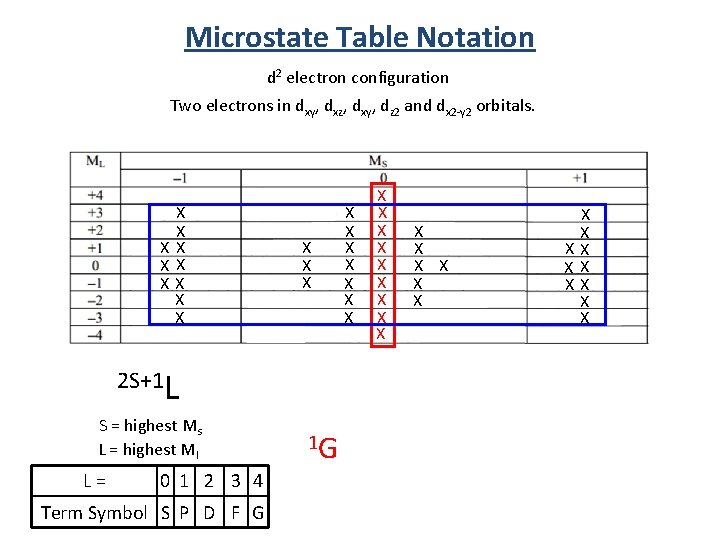
Microstate Table Notation d 2 electron configuration Two electrons in dxy, dxz, dxy, dz 2 and dx 2 -y 2 orbitals. X X XX X X X 2 S+1 L S = highest Ms L = highest Ml L = 0 1 2 3 4 Term Symbol S P D F G 1 G X X X X X X XX XX XX X X
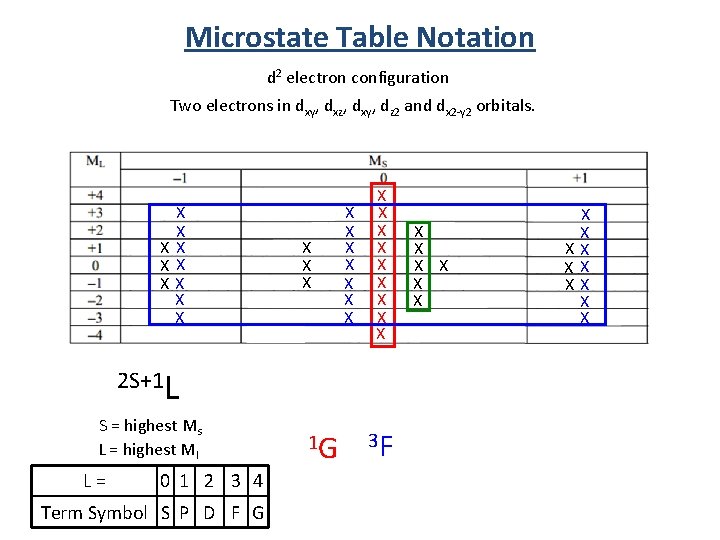
Microstate Table Notation d 2 electron configuration Two electrons in dxy, dxz, dxy, dz 2 and dx 2 -y 2 orbitals. X X XX X X X X X 2 S+1 L S = highest Ms L = highest Ml L = 0 1 2 3 4 Term Symbol S P D F G 1 G 3 F X X X X XX XX XX X X
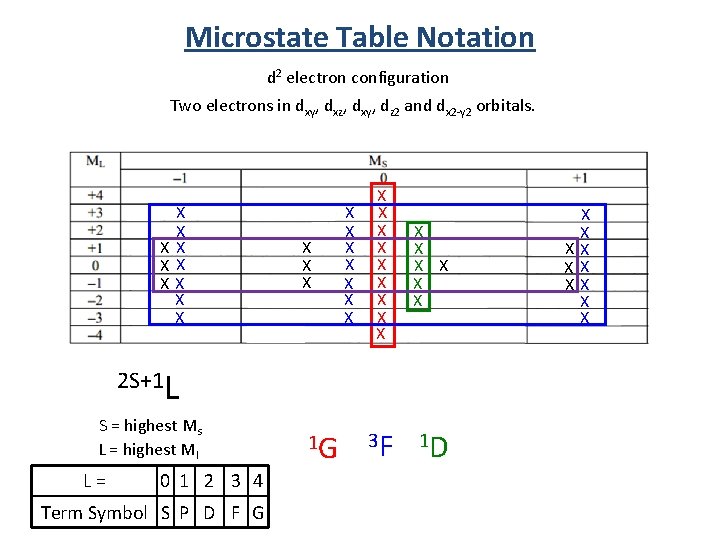
Microstate Table Notation d 2 electron configuration Two electrons in dxy, dxz, dxy, dz 2 and dx 2 -y 2 orbitals. X X XX X X X X X X X 3 F 1 D 2 S+1 L S = highest Ms L = highest Ml L = 0 1 2 3 4 Term Symbol S P D F G 1 G X X XX X X
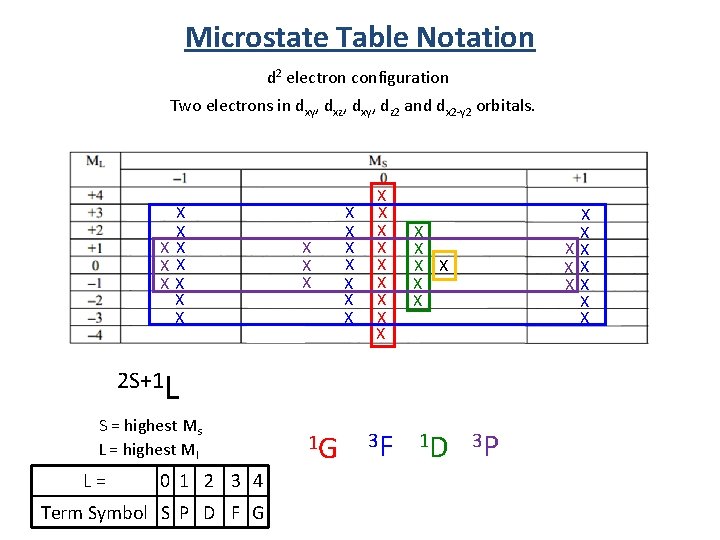
Microstate Table Notation d 2 electron configuration Two electrons in dxy, dxz, dxy, dz 2 and dx 2 -y 2 orbitals. X X XX X X X X X X X 3 F 1 D X X XX X X 2 S+1 L S = highest Ms L = highest Ml L = 0 1 2 3 4 Term Symbol S P D F G 1 G 3 P
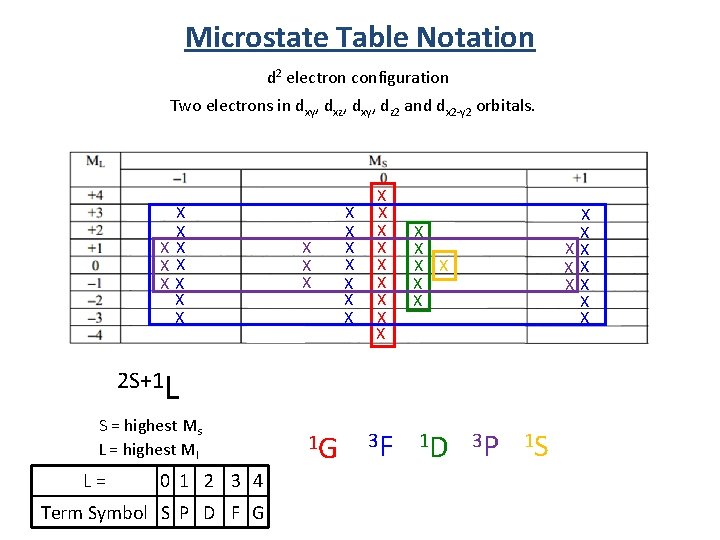
Microstate Table Notation d 2 electron configuration Two electrons in dxy, dxz, dxy, dz 2 and dx 2 -y 2 orbitals. X X XX X X X X X X X 3 F 1 D X X XX X X 2 S+1 L S = highest Ms L = highest Ml L = 0 1 2 3 4 Term Symbol S P D F G 1 G 3 P 1 S

Microstate Table Notation d 2 electron configuration Two electrons in dxy, dxz, dxy, dz 2 and dx 2 -y 2 orbitals. 1 G 3 F 1 D 3 P 1 S 1. For a given electron configuration, the term with the greatest multiplicity lies lowest in energy. (Hund’s rule. ) 2. For a term of a given multiplicity, the greater the value of L, the lower the energy. Lowest E 3 F Highest E < 3 P < 1 G < 1 D < 1 S Note: The rules for predicting the ground state always work, but they may fail in predicting the order of energies for excited states. Lowest E Real Order 3 F < 1 D < Highest E 3 P < 1 G < 1 S
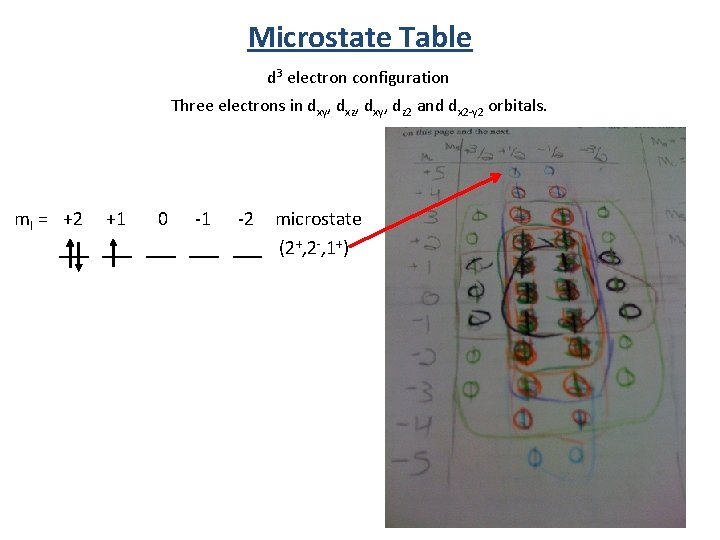
Microstate Table d 3 electron configuration Three electrons in dxy, dxz, dxy, dz 2 and dx 2 -y 2 orbitals. ml = +2 +1 0 -1 -2 microstate ___ ___ (2+, 2 -, 1+)
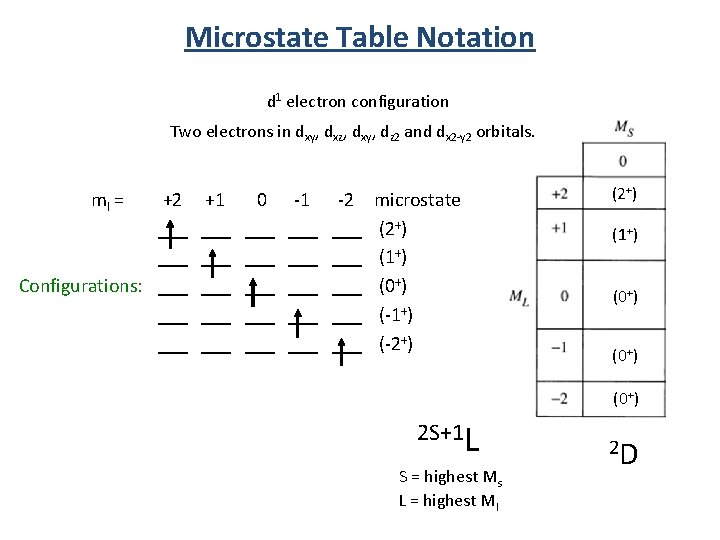
Microstate Table Notation d 1 electron configuration Two electrons in dxy, dxz, dxy, dz 2 and dx 2 -y 2 orbitals. ml = +2 +1 0 -1 -2 ___ ___ ___ Configurations: ___ ___ ___ ___ ___ microstate (2+) (1+) (0+) (-1+) (-2+) (1+) (0+) 2 S+1 L S = highest Ms L = highest Ml 2 D
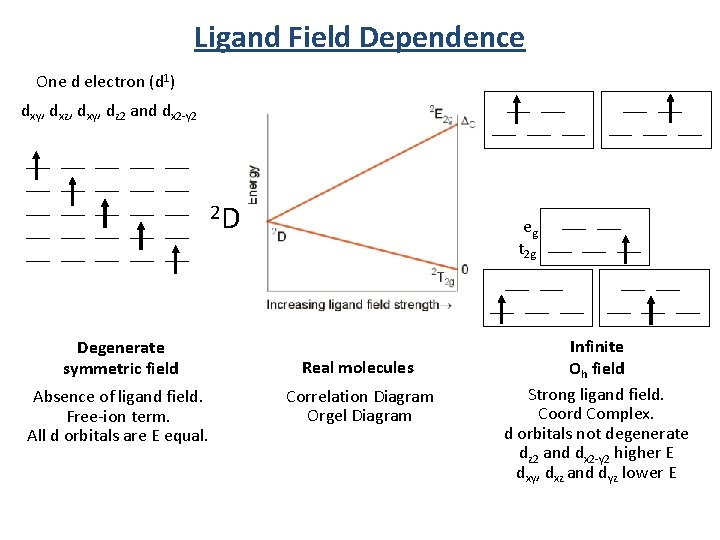
Ligand Field Dependence One d electron (d 1) ___ ___ dxy, dxz, dxy, dz 2 and dx 2 -y 2 ___ ___ ___ ___ ___ ___ ___ 2 D eg ___ t 2 g ___ ___ Degenerate symmetric field Absence of ligand field. Free-ion term. All d orbitals are E equal. ___ ___ Real molecules Correlation Diagram Orgel Diagram ___ ___ Infinite Oh field Strong ligand field. Coord Complex. d orbitals not degenerate dz 2 and dx 2 -y 2 higher E dxy, dxz and dyz lower E
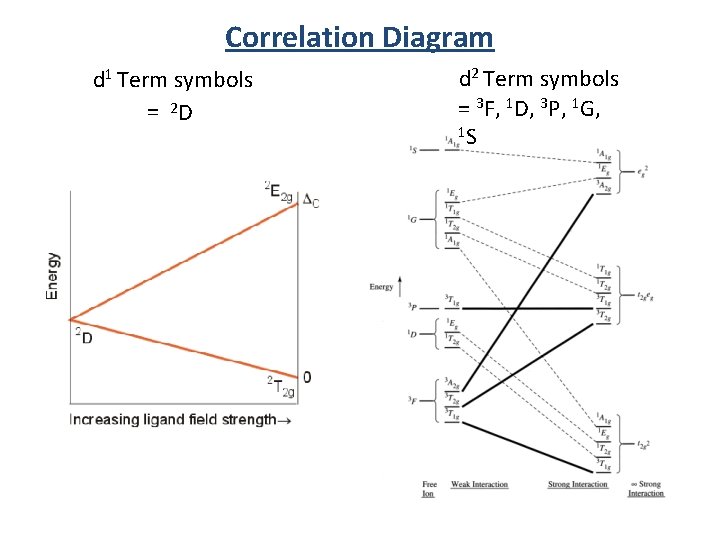
Correlation Diagram d 1 Term symbols = 2 D d 2 Term symbols = 3 F, 1 D, 3 P, 1 G, 1 S
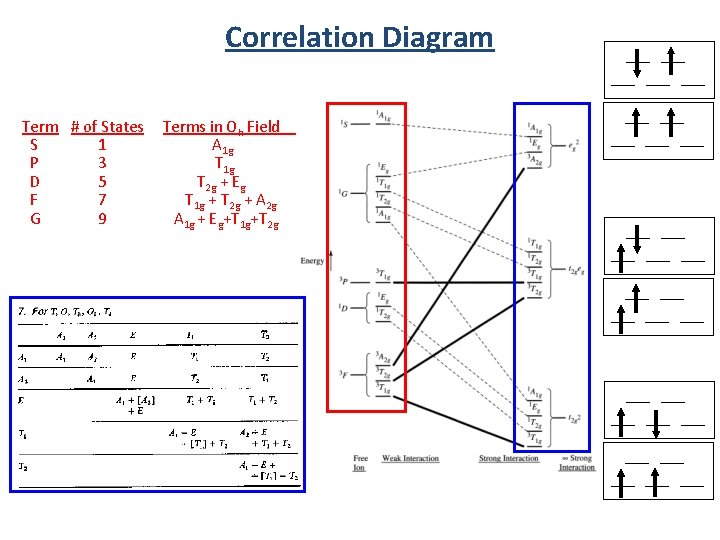
Term S P D F G Correlation Diagram # of States Terms in Oh Field 1 A 1 g 3 T 1 g 5 T 2 g + Eg 7 T 1 g + T 2 g + A 2 g 9 A 1 g + Eg+T 1 g+T 2 g ___ ___ ___ ___ ___ ___ ___ ___
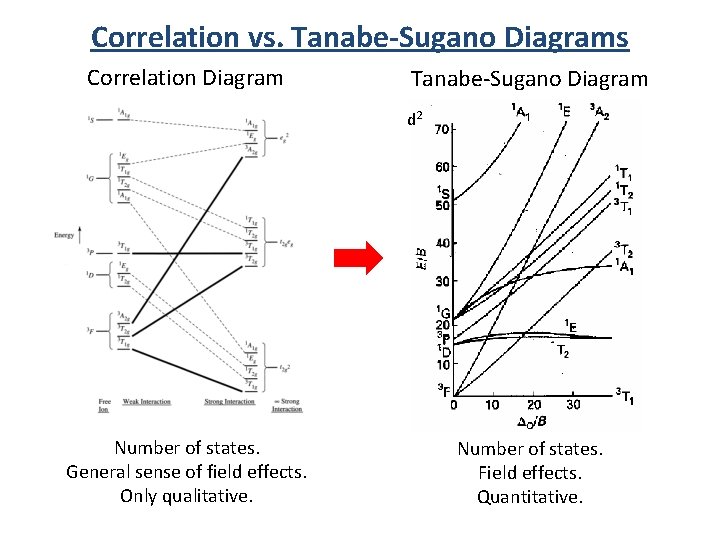
Correlation vs. Tanabe-Sugano Diagrams Correlation Diagram Tanabe-Sugano Diagram d 2 Number of states. General sense of field effects. Only qualitative. Number of states. Field effects. Quantitative.
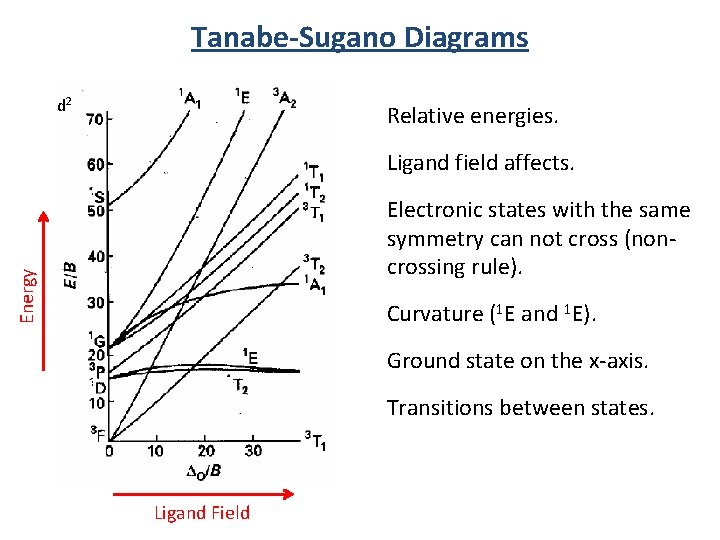
Tanabe-Sugano Diagrams d 2 Relative energies. Ligand field affects. Energy Electronic states with the same symmetry can not cross (noncrossing rule). Curvature (1 E and 1 E). Ground state on the x-axis. Transitions between states. Ligand Field
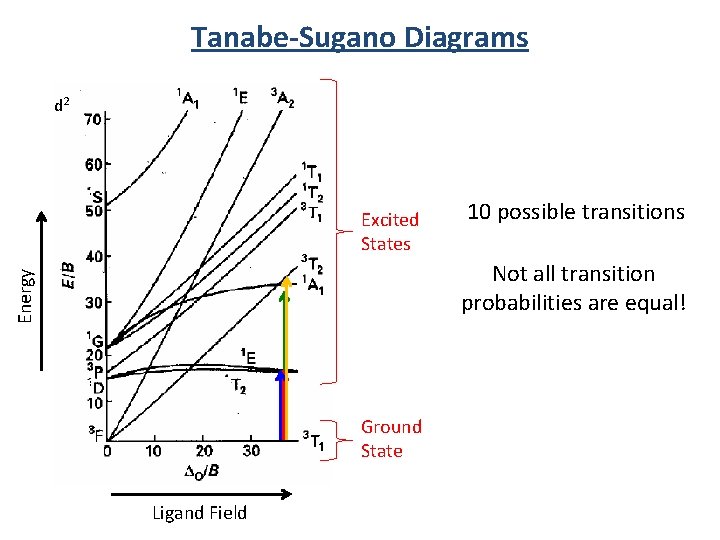
Tanabe-Sugano Diagrams d 2 Excited States 10 possible transitions Energy Not all transition probabilities are equal! Ground State Ligand Field

Selection Rules Selection rules determine the probability (intensity) of the transition. Symmetry (Laporte) Selection Rule: The initial and final wavefunctions must change in parity. Parity is related to the orbital angular momentum summation over all elections Σli, which can be even or odd; only even ↔ odd transitions are allowed. Transitions between the orbitals of the same sub shell are forbidden. Spin Selection Rule: There must be no change in the spin multiplicity (DS = 0) during the transition. i. e. the spin of the electron must not change during the transition.

Selection Rules Selection rules determine the probability (intensity) of the transition. Symmetry (Laporte) Selection Rule: The initial and final wavefunctions must change in parity. Only even (g) ↔ odd (u) transitions are allowed. Transitions between the orbitals of the same sub shell are forbidden. g→g u→u g→u u→g Allowed Forbidden For Oh complexes allowedness of a transition = T 1 u = Forbidden u = g u u u g u Allowed g = g u u Allowed g = u u g Forbidden Direct Product Rules g g u u = u g = g u = g

Selection Rules Selection rules determine the probability (intensity) of the transition. Symmetry (Laporte) Selection Rule: The initial and final wavefunctions must change in parity. Only even (g) ↔ odd (u) transitions are allowed. Transitions between the orbitals of the same sub shell are forbidden. g→g u→u g→u u→g Forbidden Allowed For Oh complexes d → d t 2 g → eg Forbidden d → p t 2 g → t 1 u Allowed p → p t 1 u → t 1 u Forbidden

Selection Rules Selection rules determine the probability (intensity) of the transition. Symmetry (Laporte) Selection Rule: The initial and final wavefunctions must change in parity. Parity is related to the orbital angular momentum summation over all elections Σli, which can be even or odd; only even ↔ odd transitions are allowed. Transitions between the orbitals of the same sub shell are forbidden. Spin Selection Rule: There must be no change in the spin multiplicity (DS = 0) during the transition. i. e. the spin of the electron must not change during the transition.
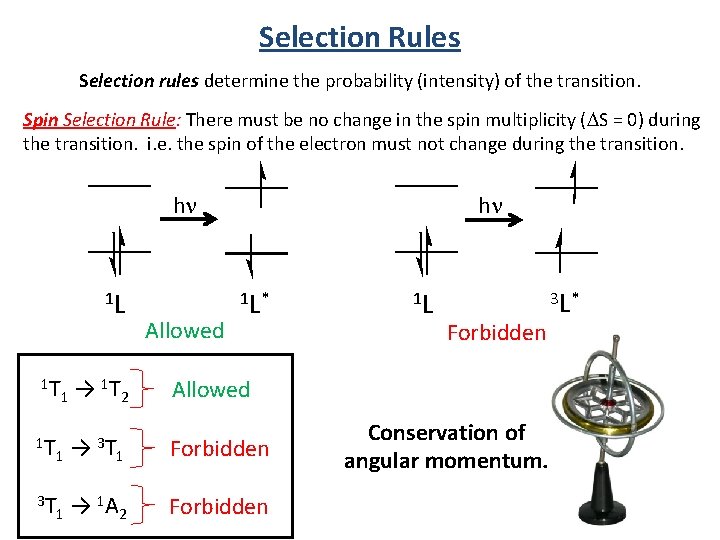
Selection Rules Selection rules determine the probability (intensity) of the transition. Spin Selection Rule: There must be no change in the spin multiplicity (DS = 0) during the transition. i. e. the spin of the electron must not change during the transition. hn 1 L 1 T 1 → 1 T → 3 T 1 → 1 T 2 1 1 A 2 Allowed hn 1 L* 1 L Forbidden Allowed Forbidden Conservation of angular momentum. 3 L*
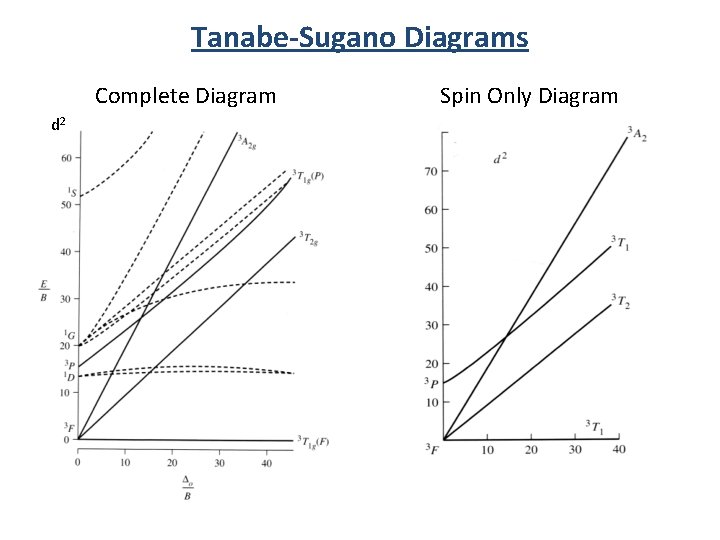
Tanabe-Sugano Diagrams Complete Diagram d 2 Spin Only Diagram

Selection Rules Selection rules determine the probability (intensity) of the transition. Symmetry (Laporte) Selection Rule: The initial and final wavefunctions must change in parity. Spin Selection Rule: The spin of the electron must not change during the transition. εmax (M 1 cm 1) Transition Spin and Symmetry forbidden "d-d" bands 0. 02 - 1 Spin allowed and Symmetry forbidden "d-d" bands 1 - 10 Spin and Symmetry allowed CT bands 103 - 5 x 104
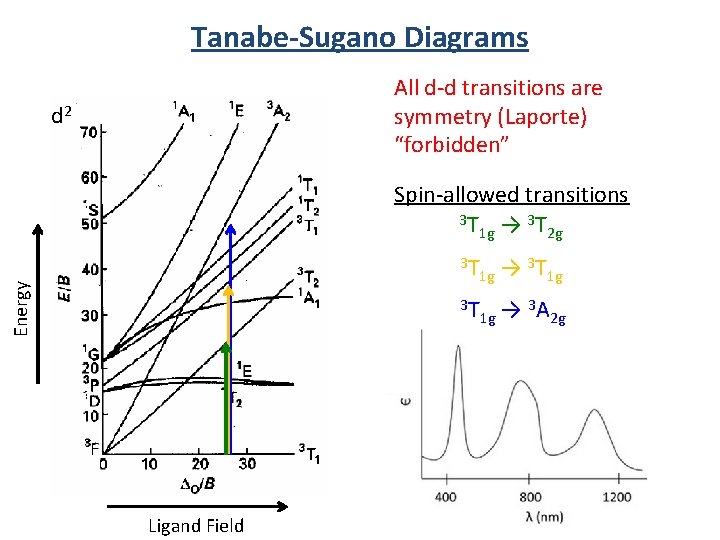
Tanabe-Sugano Diagrams All d-d transitions are symmetry (Laporte) “forbidden” d 2 Spin-allowed transitions 3 T → 3 T 1 g 2 g Energy 3 T 3 T → 1 g 1 g 3 T Ligand Field 1 g → 3 A 2 g
![d 1 and d 9 Tanabe-Sugano Diagram d 1 [Ti(H 2 O)6]3+ d 9 d 1 and d 9 Tanabe-Sugano Diagram d 1 [Ti(H 2 O)6]3+ d 9](http://slidetodoc.com/presentation_image/3b9c435c2687c5aa22e0c8fb76f66f33/image-88.jpg)
d 1 and d 9 Tanabe-Sugano Diagram d 1 [Ti(H 2 O)6]3+ d 9
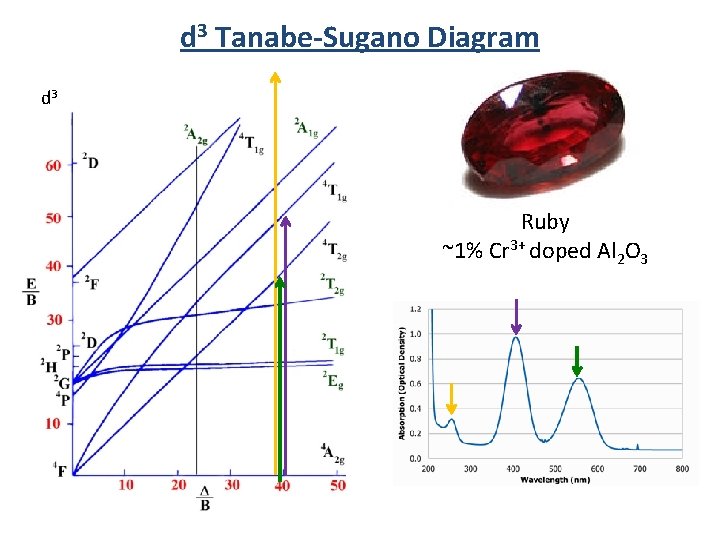
d 3 Tanabe-Sugano Diagram d 3 Ruby ~1% Cr 3+ doped Al 2 O 3
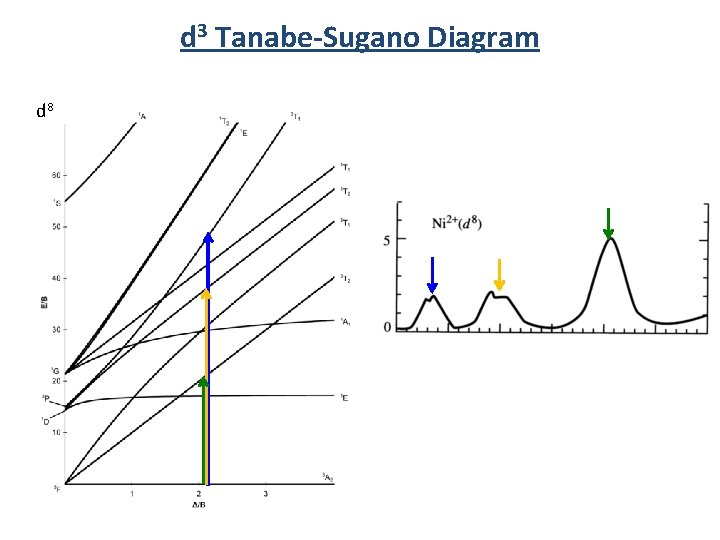
d 3 Tanabe-Sugano Diagram d 8
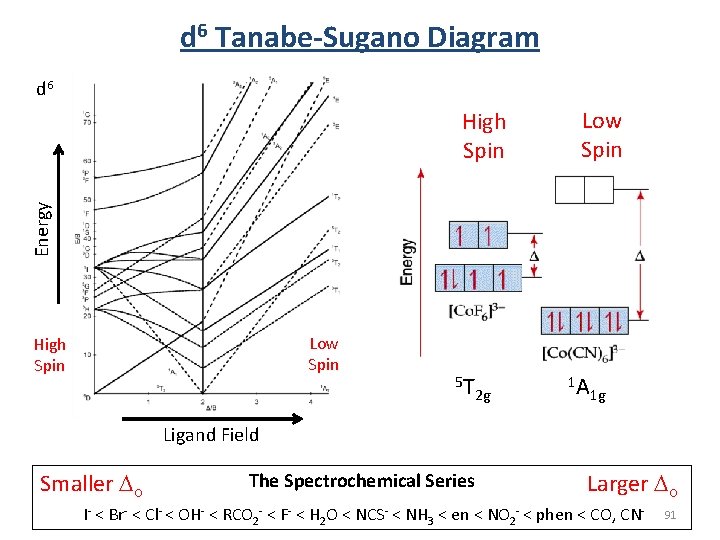
d 6 Tanabe-Sugano Diagram d 6 Low Spin Energy High Spin Low Spin High Spin 5 T 2 g 1 A 1 g Ligand Field Smaller Do The Spectrochemical Series Larger Do I- < Br- < Cl- < OH- < RCO 2 - < F- < H 2 O < NCS- < NH 3 < en < NO 2 - < phen < CO, CN- 91
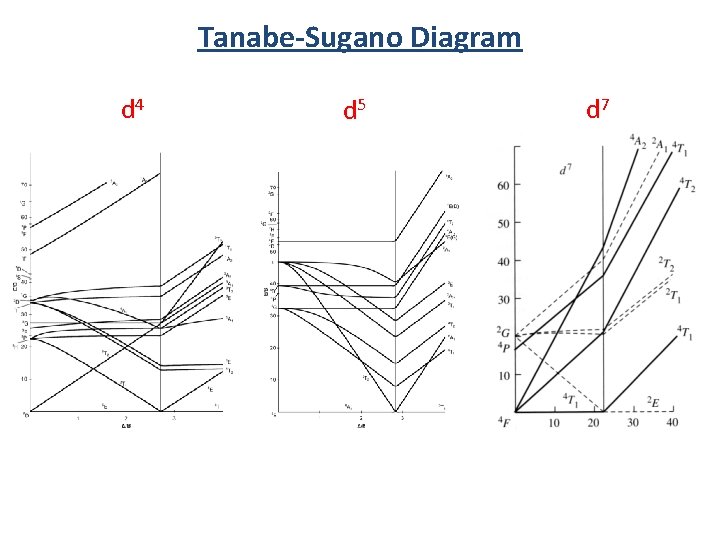
Tanabe-Sugano Diagram d 4 d 5 d 7
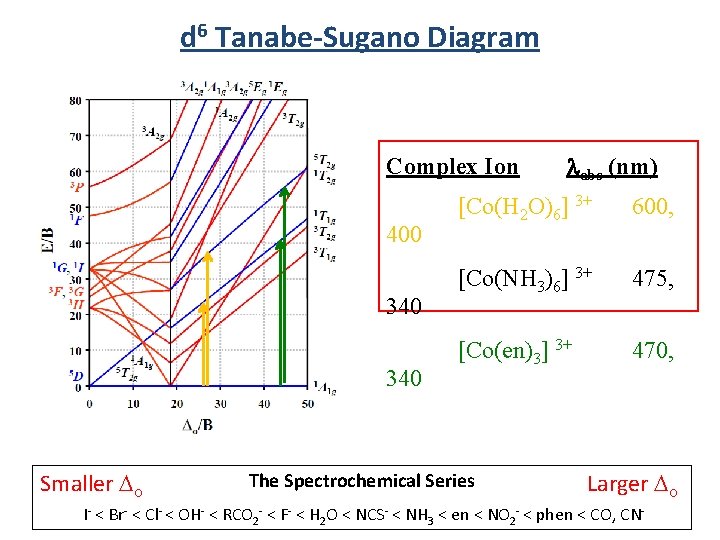
d 6 Tanabe-Sugano Diagram Complex Ion 400 340 Smaller Do labs (nm) [Co(H 2 O)6] 3+ 600, [Co(NH 3)6] 3+ 475, [Co(en)3] 3+ 470, The Spectrochemical Series Larger Do I- < Br- < Cl- < OH- < RCO 2 - < F- < H 2 O < NCS- < NH 3 < en < NO 2 - < phen < CO, CN-

Selection Rules Selection rules determine the probability (intensity) of the transition. Symmetry (Laporte) Selection Rule: The initial and final wavefunctions must change in parity. Spin Selection Rule: The spin of the electron must not change during the transition. εmax (M 1 cm 1) Transition Spin and Symmetry forbidden "d-d" bands 0. 02 - 1 Spin allowed and Symmetry forbidden "d-d" bands 1 - 10 Spin and Symmetry allowed CT bands 103 - 5 x 104 Why do we see “forbidden” transitions at all?

Allowing “Forbidden” Transitions Mechanisms that make “forbidden” electronic transitions to be “allowed” 1) Vibronic Coupling: Electronic states coupled to vibrational states help overcome the Laporte selection rule. 2) Spin-orbit Coupling: Spin and orbital angular momenta can interact to make spin “forbidden” transitions allowed. j 3) Mixing of states: π-acceptor and π-donor ligands can mix with the d-orbitals transitions are no longer purely d-d.
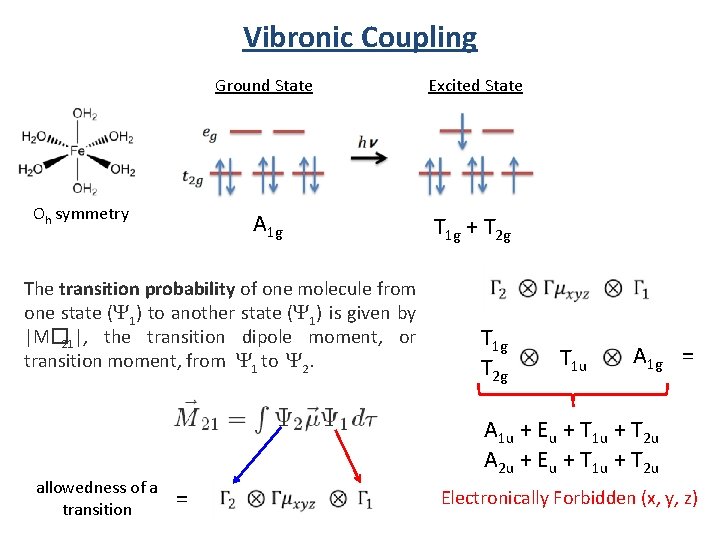
Vibronic Coupling Oh symmetry Ground State Excited State A 1 g T 1 g + T 2 g The transition probability of one molecule from one state (Y 1) to another state (Y 1) is given by |M� 21|, the transition dipole moment, or transition moment, from Y 1 to Y 2. allowedness of a transition T 1 g T 2 g T 1 u A 1 g = A 1 u + Eu + T 1 u + T 2 u A 2 u + Eu + T 1 u + T 2 u = Electronically Forbidden (x, y, z)
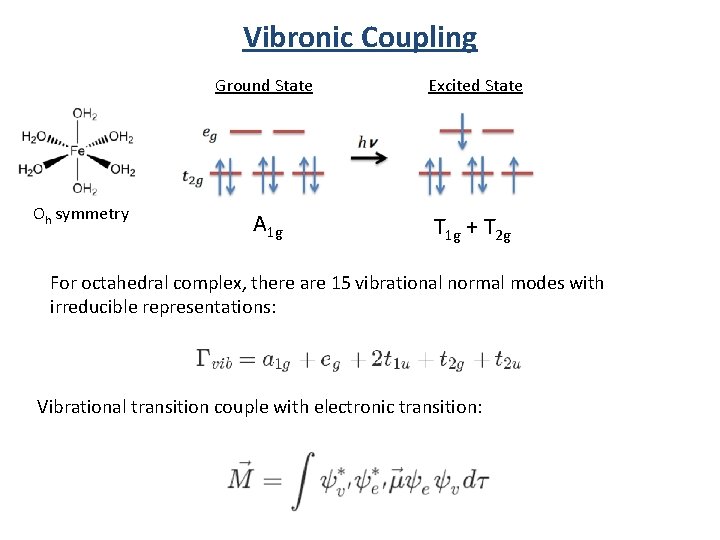
Vibronic Coupling Oh symmetry Ground State Excited State A 1 g T 1 g + T 2 g For octahedral complex, there are 15 vibrational normal modes with irreducible representations: Vibrational transition couple with electronic transition:
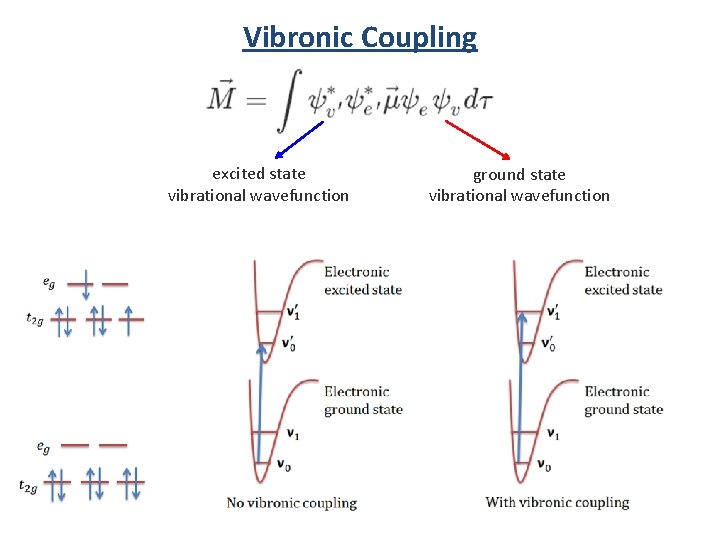
Vibronic Coupling excited state vibrational wavefunction ground state vibrational wavefunction
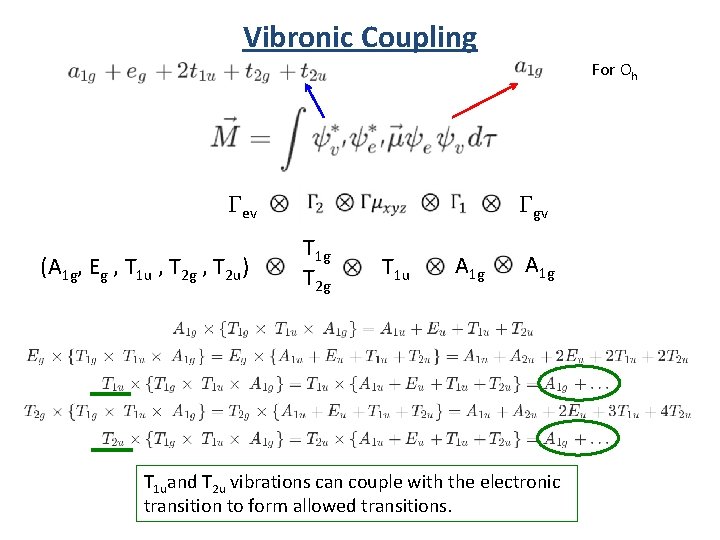
Vibronic Coupling For Oh Gev (A 1 g, Eg , T 1 u , T 2 g , T 2 u) Ggv T 1 g T 2 g T 1 u A 1 g T 1 uand T 2 u vibrations can couple with the electronic transition to form allowed transitions.
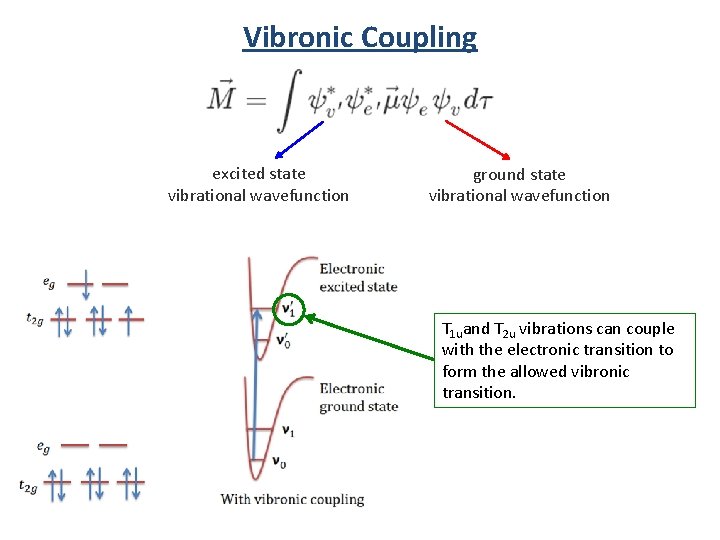
Vibronic Coupling excited state vibrational wavefunction ground state vibrational wavefunction T 1 uand T 2 u vibrations can couple with the electronic transition to form the allowed vibronic transition.
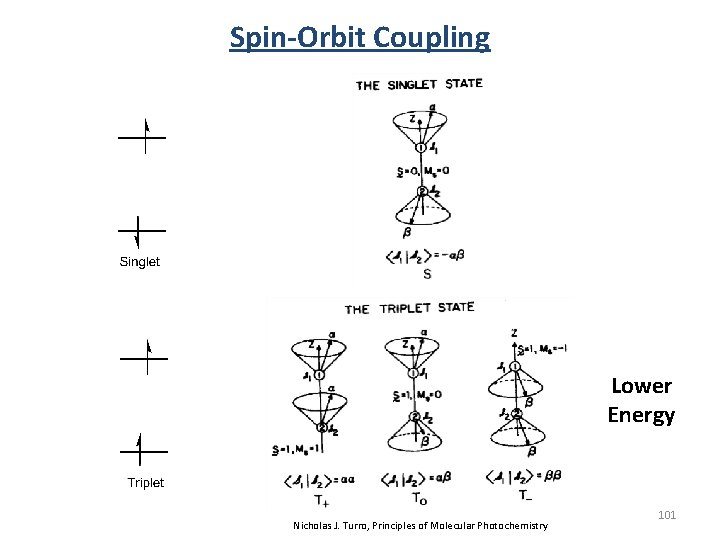
Spin-Orbit Coupling Lower Energy Nicholas J. Turro, Principles of Molecular Photochemistry 101
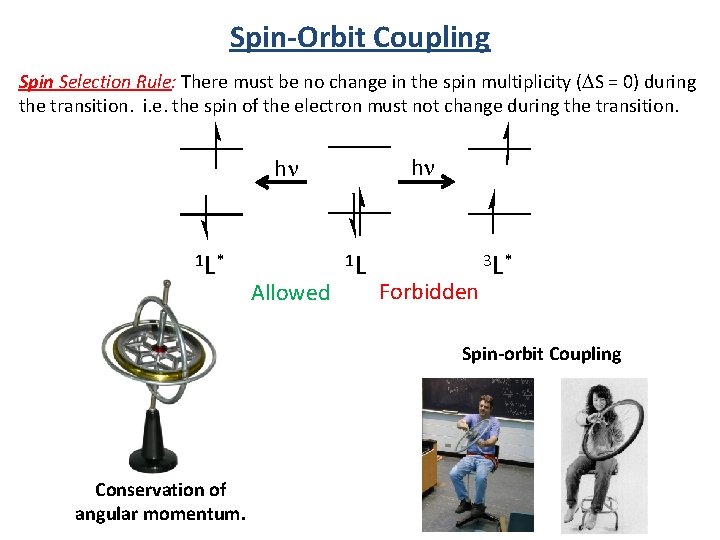
Spin-Orbit Coupling Spin Selection Rule: There must be no change in the spin multiplicity (DS = 0) during the transition. i. e. the spin of the electron must not change during the transition. hn hn 1 L* Allowed 1 L Forbidden 3 L* Spin-orbit Coupling Conservation of angular momentum.
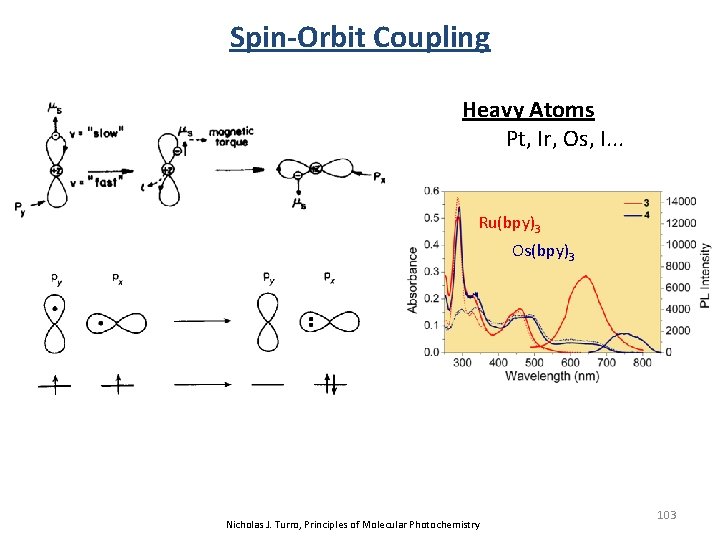
Spin-Orbit Coupling Heavy Atoms Pt, Ir, Os, I. . . Ru(bpy)3 Os(bpy)3 Nicholas J. Turro, Principles of Molecular Photochemistry 103
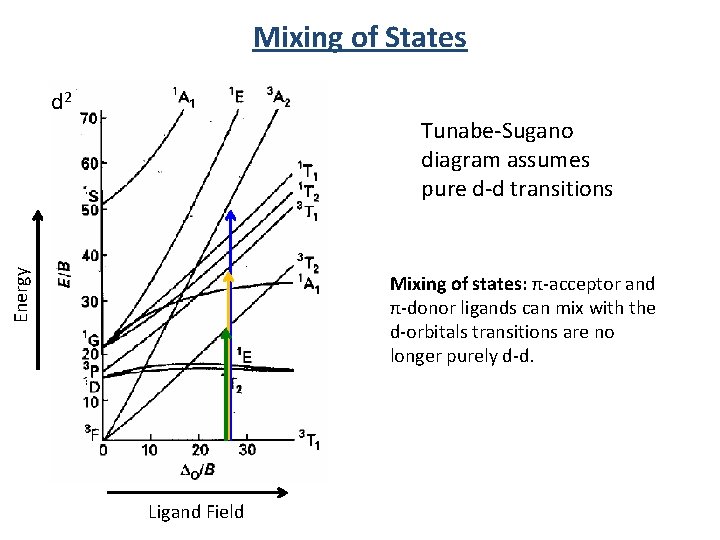
Mixing of States d 2 Energy Tunabe-Sugano diagram assumes pure d-d transitions Mixing of states: π-acceptor and π-donor ligands can mix with the d-orbitals transitions are no longer purely d-d. Ligand Field

Selection Rules Selection rules determine the probability (intensity) of the transition. Symmetry (Laporte) Selection Rule: The initial and final wavefunctions must change in parity. Spin Selection Rule: The spin of the electron must not change during the transition. εmax (M 1 cm 1) Transition Spin and Symmetry forbidden "d-d" bands 0. 02 - 1 Spin allowed and Symmetry forbidden "d-d" bands 1 - 10 Spin and Symmetry allowed CT bands 103 - 5 x 104

Outline • Absorption spectroscopy • Types of transitions – atomic – molecular • • • d-d transitions Transition moment Microstates Correlation diagrams Tanabe-Sugano diagrams Selection rules
- Slides: 106