Parkinsons Disease Bradykinesia tremor rigidity postural reflect impairment

Parkinson’s Disease Bradykinesia, tremor, rigidity, postural reflect impairment n Destruction of dopaminergic neurons in the pars compacta of the substantia nigra (with Lewy inclusion bodies) n Lifetime risk up to 2% men, 1. 3% women; rates rising among >75 yo n

Parkinson’s Disease and the Environment: the Potential Contribution of Metal-Gene Interactions Howard Hu, M. D. , M. P. H. , Sc. D. Professor of Occupational and Environmental Medicine Harvard School of Public Health

PD causation n Twin studies: must be mostly environmental or gene-env >50 yo n Environment – IVDA exposed to MPTP – Pesticides • Paraquat—induces oxygen free radicals leading to lipid peroxidation in neurons • Heptachlor, rontenone, dieldrin—may accelerate alpha-synuclein fibril formation – Solvents—n-hexane, toluene – Smoking (inverse) – Metals? ? ?

PD causation: Metals? n Metals – Manganese—miners, welder, smelters – Mercury—case-control study – Lead—Gorell study: population casecontrol, occupational exposure to lead —OR of 5. 24 (95%CI: 1. 59 -17); exposure rated by IH blinded to casecontrol status n Mechanism – Catalyzation of Fenton reaction generating reactive oxygen species – Synergy with iron?
Metals Epidemiology Research Group n Director: Howard Hu n Primary base: – Dept. Environ Health, HSPH (also, Depts. Of Epi, Biostat, Health and Social Behavior, Maternal and Child Health, Cell and Cancer Biology) – Channing Lab, Brigham&Women’s, HMS n Collaborations: – Boston: Boston VA Hospital, Normative Aging Study, BU Neuro, Children’s Hospital, Mass Coll of Pharmacy – Outside: UC-Santa Cruz, NIEHS, U. Pittsburgh, Brookhaven Nat’l Lab, Nat’l Inst. Public Health. Mexico` n Funding: NIEHS, NHLBI, EPA, CDC, ATSDR,

Lead as cause of PD High population exposures with longlived body stores n Animal studies: lead decreases dopamine synthesis, turnover, uptake in the basal ganglia n Increased spontaneous release of dopamine; dopamine auto-oxidized to 6 -OHDA, facilitating Fenton reaction; places these neurons at increased risk of oxidative toxicity n Lead also has direct oxidative properties, perhaps mediated by ALA n

Our relevant prior research n We have developed and used a new biological marker of cumulative lead dose to elucidate lead’s impact on chronic disease n We have also examined other leadgene interactions n Examples…
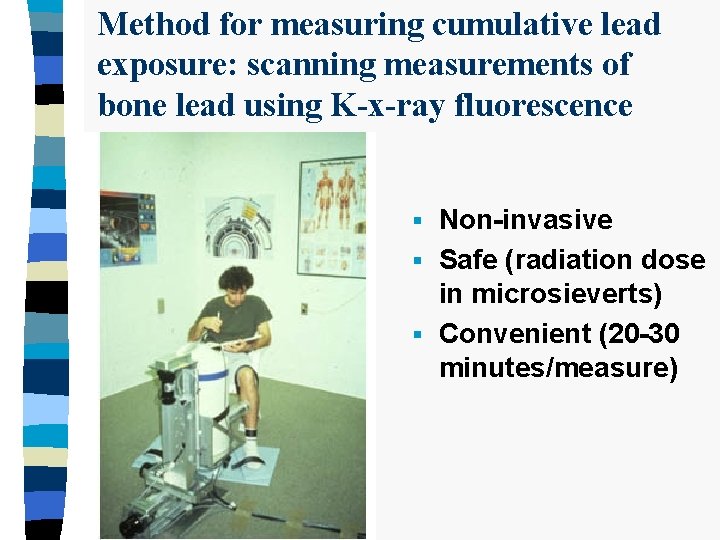
Method for measuring cumulative lead exposure: scanning measurements of bone lead using K-x-ray fluorescence Non-invasive § Safe (radiation dose in microsieverts) § Convenient (20 -30 minutes/measure) §
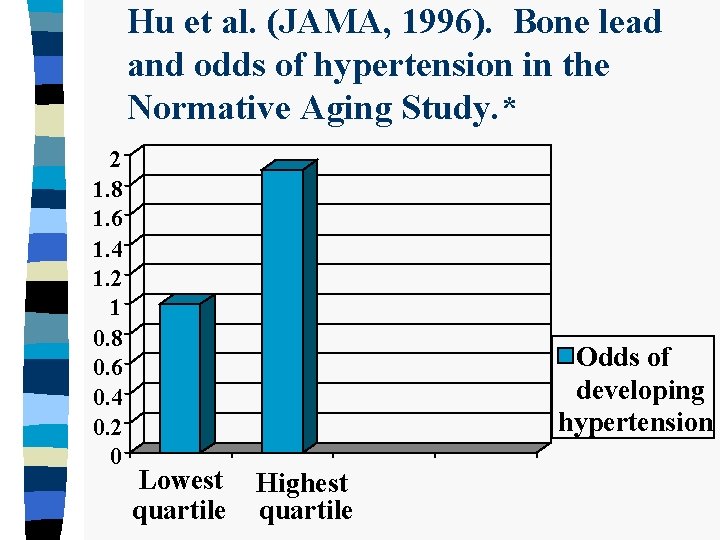
Hu et al. (JAMA, 1996). Bone lead and odds of hypertension in the Normative Aging Study. * 2 1. 8 1. 6 1. 4 1. 2 1 0. 8 0. 6 0. 4 0. 2 0 Odds of developing hypertension Lowest quartile Highest quartile
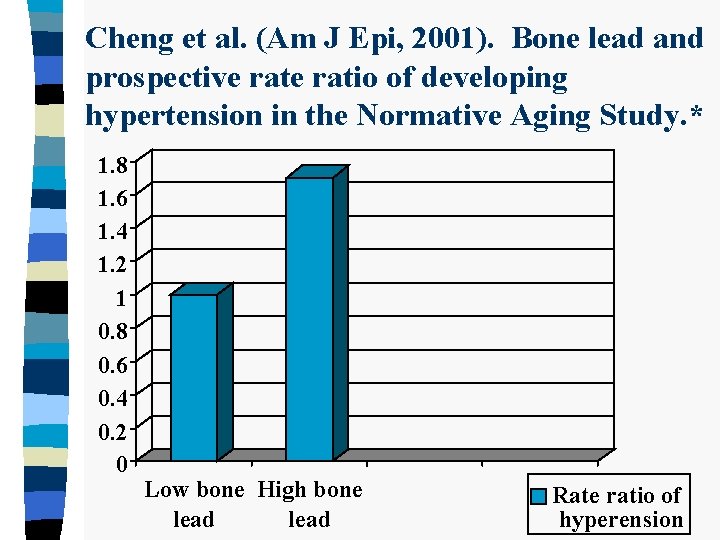
Cheng et al. (Am J Epi, 2001). Bone lead and prospective ratio of developing hypertension in the Normative Aging Study. * 1. 8 1. 6 1. 4 1. 2 1 0. 8 0. 6 0. 4 0. 2 0 Low bone High bone lead Rate ratio of hyperension
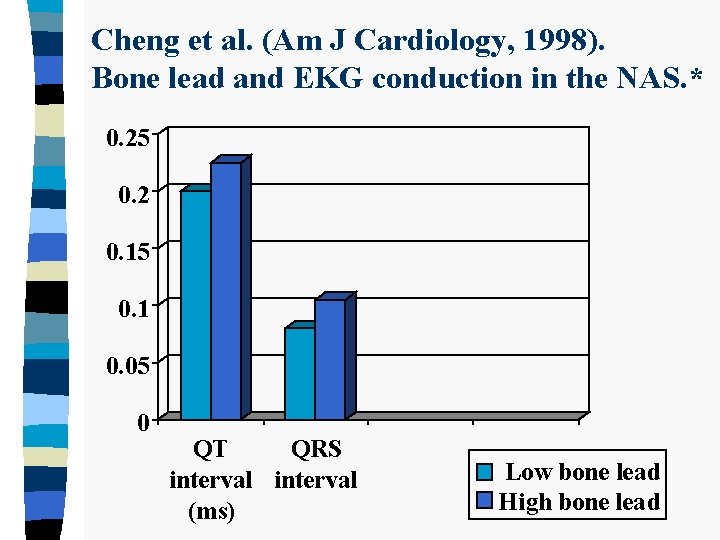
Cheng et al. (Am J Cardiology, 1998). Bone lead and EKG conduction in the NAS. * 0. 25 0. 2 0. 15 0. 1 0. 05 0 QT QRS interval (ms) Low bone lead High bone lead
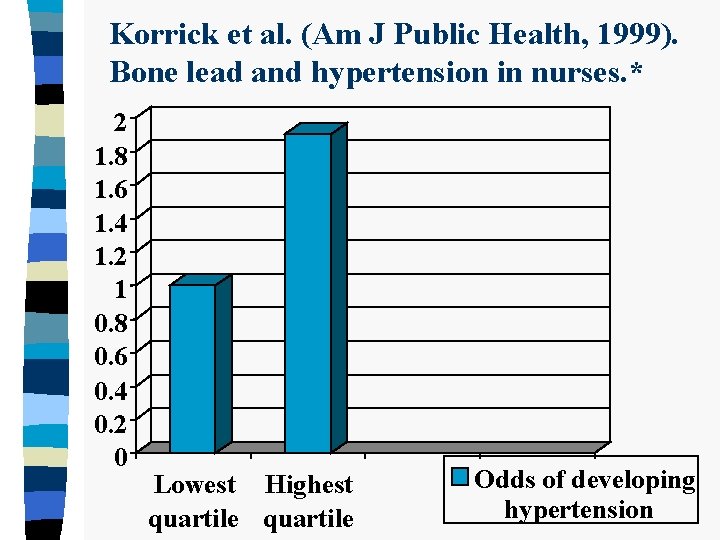
Korrick et al. (Am J Public Health, 1999). Bone lead and hypertension in nurses. * 2 1. 8 1. 6 1. 4 1. 2 1 0. 8 0. 6 0. 4 0. 2 0 Lowest Highest quartile Odds of developing hypertension
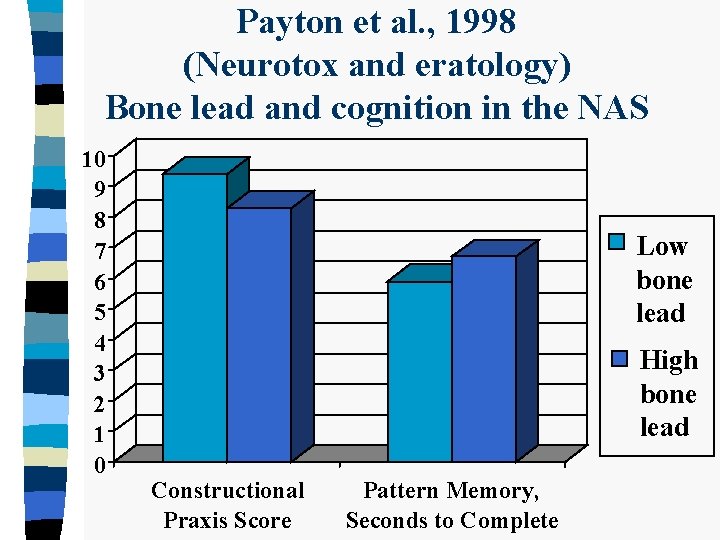
Payton et al. , 1998 (Neurotox and eratology) Bone lead and cognition in the NAS 10 9 8 7 6 5 4 3 2 1 0 Low bone lead High bone lead Constructional Praxis Score Pattern Memory, Seconds to Complete
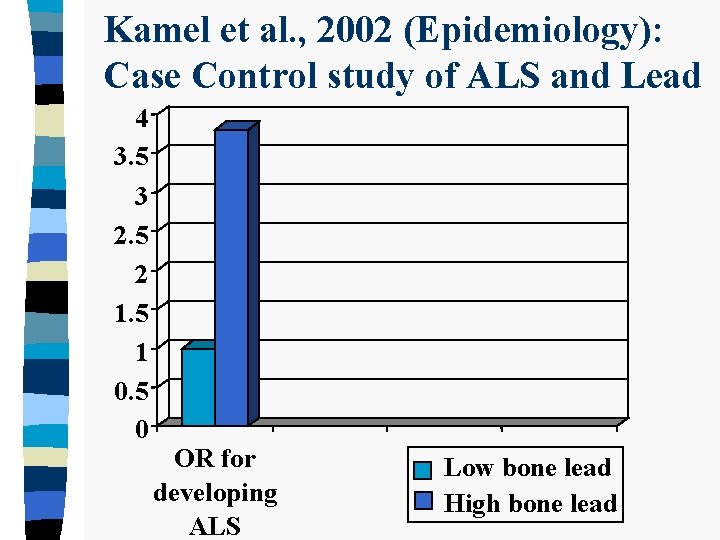
Kamel et al. , 2002 (Epidemiology): Case Control study of ALS and Lead 4 3. 5 3 2. 5 2 1. 5 1 0. 5 0 OR for developing ALS Low bone lead High bone lead
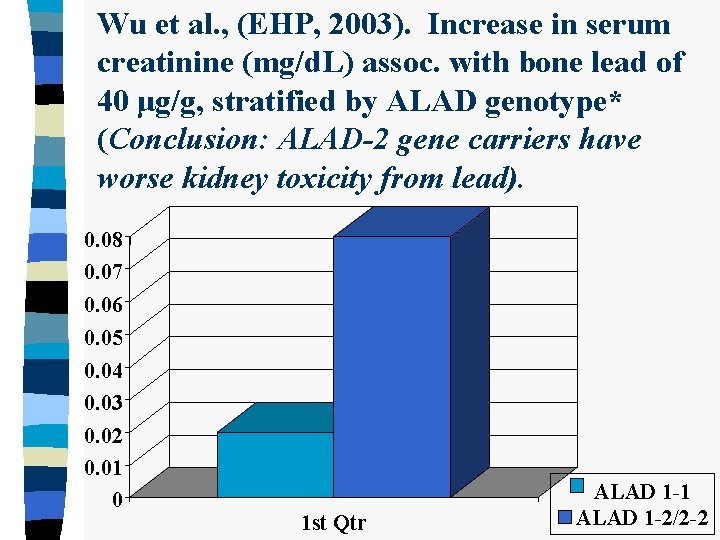
Wu et al. , (EHP, 2003). Increase in serum creatinine (mg/d. L) assoc. with bone lead of 40 μg/g, stratified by ALAD genotype* (Conclusion: ALAD-2 gene carriers have worse kidney toxicity from lead). 0. 08 0. 07 0. 06 0. 05 0. 04 0. 03 0. 02 0. 01 0 1 st Qtr ALAD 1 -1 ALAD 1 -2/2 -2

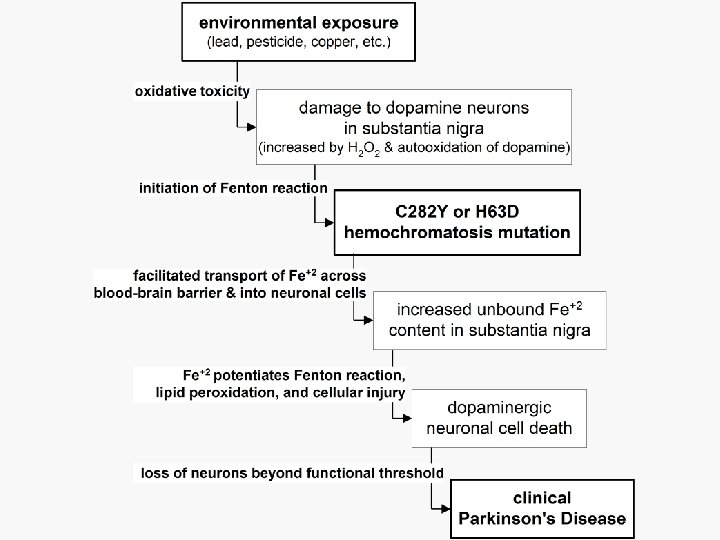
Candidate genes for gene-metal interactions and Parkinson’s n HFE—hemochromatosis (C 282 Y, H 63 D) – Increases intracellular iron that can • catalyze reactions to produce toxicfree radicals • promote the Fenton reaction

Our Study Design: Case Control Epidemiologic Study NIEHS R 01 ES 10798 to Channing Lab, 5 yrs n Major collaboration with the late R. Feldman, Marie St-Hilaire, and BU-PDC n Existing PD patients (n=1, 233) + new PD patients (n=1, 080) n Critieria: PD sx<10 y, meet case def, within 2 hrs drive n Controls: spouses and in-laws n

Case definition Complete hx and clinical eval by neurol n 2 of 3: resting tremor, cogwheel rigidity, bradykinesia n Assymetry n None of: [supranuclear gaze palsy, postural instability, dysautonomia] out of proportion for PD; unexplained [cerebellar findings, hyperreflexia]; no response to L-dopa; nonprogressive; MRI or CT with infarcts n Recent clinical exam, with at least 2 exams n Sx < 10 yrs n

Protocol n Questionnaire (exposures, smoking, diet, etc) n KXRF measures of bone lead, blood lead n Toenails for manganese, copper n Blood for genotyping of HFE status and for future genotyping, other studies
- Slides: 20