Parasitology lab 1 Fecal Examination Specimen Collection Collect

Parasitology lab. 1 Fecal Examination

Specimen Collection � � � Collect the stool in a dry, clean, leakproof container. Make sure no urine, water, soil or other material gets in the container. Fresh stool should be examined, processed, or preserved immediately. An exception is specimens kept under refrigeration when preservatives are not available; these specimens are suitable for antigen testing only. Preserve the specimen as soon as possible. If using a commercial collection kit, follow the kit’s instructions. If kits are not available, the specimen should be divided and stored in two different preservatives, 10% formalin and PVA (polyvinyl-alcohol), using suitable containers. Add one volume of the stool specimen to three volumes of the preservative

� � � Insure that the specimen is mixed well with the preservative. Formed stool needs to be well broken up. Insure that the specimen containers are sealed well. Reinforce with parafilm or other suitable material. Insert the container in a plastic bag. Certain drugs and compounds will render the stool specimens unsatisfactory for examination. The specimens should be collected before these substances are administered, or collection must be delayed until after the effects have passed. Such substances include: antacids, kaolin, mineral oil and other oily materials, nonabsorbable antidiarrheal preparations, barium or bismuth (7 -10 days needed for clearance of effects), antimicrobial agents (2 -3 weeks), and gallbladder dyes (3 weeks).

� Specimen collection may need to be repeated if the first examination is negative. If possible, three specimens passed at intervals of 2 -3 days should be examined.
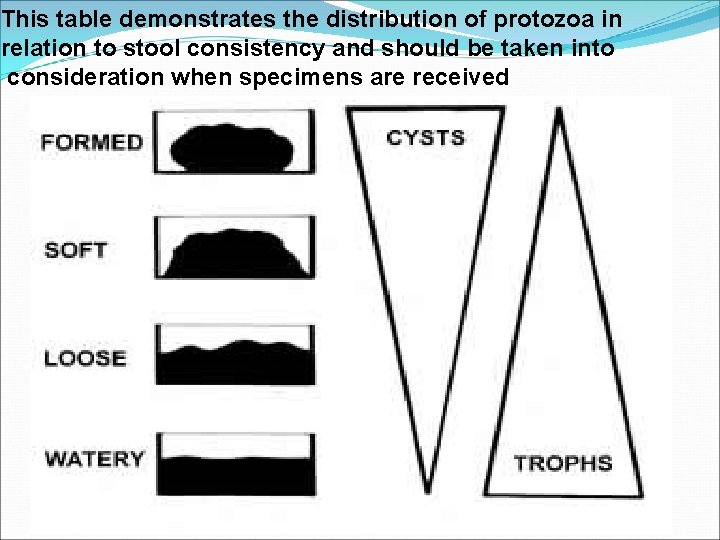
This table demonstrates the distribution of protozoa in relation to stool consistency and should be taken into consideration when specimens are received

Staining � 1 -Temporary : Saline, iodine, eosin � 2 - Semi permanent: Quensel solution � 3 - Permanent: Iron hematoxylin and Trichrome stain.

�Fecal Examination �- Macroscopic: � Color, consistency, blood, mucus, proglottids, adult - Microscopic: Wet mount : 1 - Saline 2 - Iodine

1 - Saline : -Saline preparation is employed to Microscopic examenation demonstrate worm eggs , larvae , protozoal trophozoites as well as RBCs and WBCs. on the left side of slide place a drop of saline and a drop of lugol's iodine on the right. Using a wire loop or a piece of stick , mix a small amount of feces and make a smooth suspension. place a cover slip on each of these suspensions.

Examine the preparations systemically with low power objective ; if suspicious objects are noticed switch to high power objective ( X 40 ) to observe the details.

2 - Iodine : place one drop of iodine on a slide. The rest of the procedure is as described for saline .

v. Iodine preparation is used mainly to stain glycogen and the nuclei of cysts ; iodine stain the non – bile staining eggs , hence this preparation is a less of importance for identification of helminthes eggs as bile staining and non bile staining eggs can not be differentiated.

2 - Concentration methods: If the number of cysts , eggs or larvae is low in feces , direct examination may not reveal them. , hence , the feces is concentrated.

The concentration techniques commonly used are : 1 - sedimentation techniques: - ( Formol ether concentration techniques. It is consider best over all techniques. 2 -Floatation techniques : - ( Zinc sulphate floatation techniques. )

1 - sedimentation techniques: - ( Formol ether concentration techniques. It is consider best over all techniques. It is used to concentrate protozoan cysts , helminthes eggs and larvae. Unlike the zinc sulfate flotation method , it is useful for concentrating operculated eggs.

1 - Mix enough feces with 10 -12 ml of saline to about 12 – 1 ml of fecal sedement after completion of 2 and 3 below. 2 -Strain through 2 layers of wet gauze into a paper cup or beaker. 3 -Transfer to a 15 ml conical tube and centrifuge for 1 min. at 1500 -2000 rpm. . 4 - Decant the supernatant fluid and resuspend the sediment in fresh saline ,

5 -Centrifuge and decant as before. 6 -Mix the fecal sediment with 10 ml of 10 % formalin and allow 10 min. for fixation. 7 -Add 3 ml of ether , stopper the tube and shake vigorously. 8 - Centrifuge at about 1500 rpm for 2 min; four layers should formed as follows:

�A. An ether layer on top. �B. a plug of fecal detritus. �C. A layer of formalin. �D. The bottom layer of sediment containing protozoa and helminthes eggs. 9 -Free the plug of fecal detritus from the sides of the tube by ringing with an applicator stick , pour off the supernatant fluid.

10 - Mix the remaining sediment with small amount of fluid that drain back from the sides of the tube. place drop of sediment on a slide and cover with cover slip , examine under LPM ( 10 X)

2 -Floatation techniques : - ( Zinc sulphate floatation techniques. ). This method concentrates protozoan cysts , most non operculated helminthes eggs and larvae. This method is suitable for fatty stools. The procedure is as follow : -

1 -Mix one part of feces with 10 parts of tap water. 2 -Strain about 10 ml of fecal suspension through wet gauze into a tube. 3 -Centrifuge for 1 min. at 2300 rpm pour off the supernatant fluid , additional water to fill the tube. 4 -Repeat step 3 once or twice until the supernatant become clear.

5 -Pour off the supernatant fluid; add 3 -4 ml of Zn. SO 4. to within 3 -4 ml of the tube rim. 6 -Centrifuge for 1 min. at 2300 rpm and allow the tube to come rest without interference. 7 -After 1 -2 mint. by using a loop transfer loop-full of material from the surface film to a clean slide. cover with cover slip and examine under LPM (10 X).

THANK YOU FOR YOUR ATTENTION
- Slides: 23