Paracetamol and salicylates poisoning By Elzahraa Ahmed Elrefaie

Paracetamol and salicylates poisoning By Elzahraa Ahmed Elrefaie Ali Assistant lecturer of Clinical Toxicology Section 7 & 8
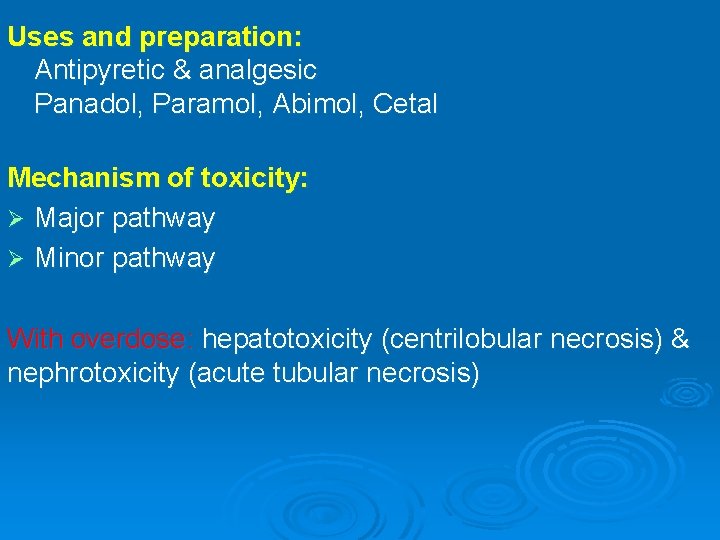
Uses and preparation: Antipyretic & analgesic Panadol, Paramol, Abimol, Cetal Mechanism of toxicity: Ø Major pathway Ø Minor pathway With overdose: hepatotoxicity (centrilobular necrosis) & nephrotoxicity (acute tubular necrosis)

Conditions of poisoning: 1. Accidental: in children, the most common. 2. Suicidal: by young adolescents (cheap, available & easy obtained). 3. Homicidal: rare due to large fatal dose Fatal dose: 140 mg /Kg. Fatal period: first 10 days (peak 7 days) from liver cell failure.

Clinical picture: Divided into 4 phases: 1. Phase I: 30 min-24 hrs. a. Anorexia, nausea and vomiting. b. Malaise, pallor, and diaphoresis. c. No blood chemistry changes

2. Phase II: 24 -72 hrs. a. Pain and tenderness in the right hypochondrium. b. Altered liver function tests: Elevation in liver enzymes (AST, ALT) and bilirubin, Prolongation of prothrombin time. 3. Phase III: 72 -96 hrs. a. Liver failure (jaundice, coagulation defects and encephalopathy and altered conscious level). b. ALT, AST > 1000 IU/ L. c. Death may occur due to multi-organs failure

4. Phase IV: 4 -10 days a. Complete resolution of hepatic dysfunction and liver heals without evidence of fibrosis. b. Lab: return to normal values

Investigations: A-routine studies: -Serum electrolytes -CBC -ECG -ABG -liver & kidney functions are mandatory 3 times in the first day, twice daily for the next 4 days then once daily till time of discharge.
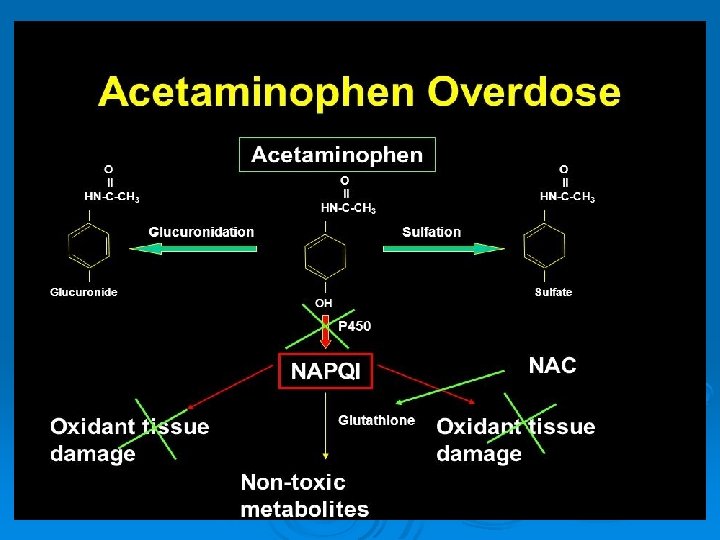
B-specific studies: Serum acetaminophen level “Rumack-Mathew nomogram for prediction of hepatotoxicity”

Treatment: Ø General measures: -emesis and gastric lavage have not a consideration in patients with isolated acetaminophen toxicity due to rapid absorption of acetaminophen -activated charcoal -saline cathartics

Ø Specific antidote: N- acetyl cysteine Indications: 1 - Empirically when the amount of ingestion is unknown or serum concentrations are not available. 2 - If the serum level falls above the upper (“probable toxicity”) line on the nomogram. 3 - If the serum level falls between the two nomogram lines, (probable toxicity) line and (possible toxicity) line, and the patient is at high risk for toxicity

Mechanism: NAC acts as a sulfhydryl group donor, substituting for glutathione to detoxify the toxic metabolite (NAPQI). Doses & protocols: oral route (72 hrs) • loading dose: 140 mg/kg. • maintenance dose: 70 mg/kg every 4 hours for 17 doses IV route (48 hrs) • loading dose: 140 mg/kg diluted in dextrose slowly over 1 h • maintenance dose: 70 mg/kg every 4 hours for 12 doses

IV route (20 hrs) • 150 mg/kg in 200 m. L of 5% dextrose over 15 minutes, • then 50 mg/kg in 5% dextrose over 4 h • and then 100 mg/kg in 5% dextrose over 16 hours Ø Other antidotes: Methionine Cimetidine N-acetylcysteine amide Cysteamine D-penicillamine

Ø Symptomatic treatment: -liver support: dextrose -liver transplantation in cases of fulminant hepatic failure -liver dialysis -hemodialysis for if renal failure present

Salicylates toxicity

Uses & available preparations: Salicylates are widely used asan analgesic, antipyretic and anti-inflammatory. 1. Aspirin and Rivo: One tablet contains 325– 650 mg acetylsalicylic acid. 2. Cold preparations 3. Methyl salicylate "oil of wintergreen" (topical keratolytic): One teaspoon of concentrated oil of wintergreen contains 5 g of methyl salicylate, equivalent to about 7. 5 g acetylsalicylic acid. 4. Bisthmus salicylate: antidiarrhea

Pharmacokinetic&toxikinetics: -Ingested salicylate is rapidly absorbed from upper GIT, while overdose may produce pylorospasm leading to delayed absorption, concretions and enteric coated also delay absorption. -low molecular weight & water soluble (favor hemodialysis) -Peak plasma concentration is reached within 30 m -low volume of distribution 0. 2 L/kg but can be increased by acidemia. Acidosis decrease sailcylate ionization and increase its tissue precipitation ( more tissue damage).

Pharmacokinetic&toxikinetics: -High plasma protein binding 80 -90% -metabolism in liver with conjugation with glycine and glucuronic acid -excreted by kidney (depend on urine PH). -The elimination half-life is 2– 5 hours but 20 hours after overdose (zero order kinetics)

Pharmacodynamics • -Anti-inflammatory: -Inhibition of PG synthesis by inhibition of cyclooxygenase pathway. -inhibition of migration of PMNs, leucocytes and other inflammatory cells to the site of infection. -inhibition of adhesion of granulocytes to the damaged blood vessel - inhibit kallikrin system

Pharmacodynamics -Analgesic: due to anti-inflammatory action -inhibition of pain perception at sub cortical level Anti pyretic: -inhibition of PG -vasodilatation of superficial blood vessels increase heat loss Anti-platelet: -reset hypothalamic thermal control Antipyretic -inhibit thromboxane A 2 which accelerate platelets aggregation Hypoprothrombinemia : -Decrease liver uptake of vitamin K

Conditions of poisoning: Mode of poisoning: 1. Accidental: common in children 2. Suicidal: common in young adolescents. 3. Homicidal: rare due to large fatal dose Fatal dose: 390 mg/Kg (i. e. 60 - 70 tab. ) Cause of death : central asphxia in stage of depression Fatal period: 24 hour

Clinical presentation 1. GIT & hepatic manifestation: a. Burning pain in mouth and throat. b. Nausea and vomiting c. Gastric ulceration leading to GIT bleeding 'hematemesis'. d. hepatotoxicity similar to Rye's syndrome (Occur in children following viral infection manifested by nausea, vomiting, hypoglycemia, elevated liver enzymes, fatty infiltration of liver and coma)

Clinical presentation 2. CNS: Salicylates first stimulate and then depress the CNS: a. Restlessness, excitability, agitation and convulsions ( occur with hypoglyemia, causes brain edema) b. Coma in severe cases. 3. CVS: a. Hypotension, collapse and tachycardia. b. Noncardiogenic pulmonary edema (increase capillary permeability). 4. Hematology: bleeding tendencies due to: a. Inhibition of prothrombin synthesis (↑prothrombin time). b. Inhibition of platelet functions& decrease platelets adhesion (↑bleeding time). c. Decrease factors. II, VII, IX and X(vit K dependent factors) d. Increase capillary fragility.

Clinical presentation 5. Hypersensitivity reactions (Allergy): a. Urticaria and skin rash. b. Precipitation of bronchial asthma. c. Angioneurotic edema. 6. Hearing: Salicylism - Tinnitus and deafness duo to 8 th cranial nerve involvement and increase membrane permeability as inhibition of cyclo-oxgenase and decrease in PGs that interfer with Na K Atpase pump in striavascularis →vasoconstriction →decrease cochlear blood flow→ change membrane permeability →loss of outer hair cell of organ of corti →decrease autocaustic emissions

Clinical presentation 7. Kidney: Renal impairment due to: a. Direct effect of aspirin → renal tubular necrosis → uremia. b. Indirect effect due to decreased renal perfusion from dehydration. 8. Fluid and electrolyte disturbances: - Dehydration due to: a. Increased metabolic rate and hyperthermia. b. Vomiting, sweating and hyperventilation - Hypokalemia: due to vomiting, urinary loss. 9. Metabolic disturbances: -Short lived hyperglycemia then hypoglycemia due to increase metabolism and increase insulin secretion.
Clinical presentation 10. Hyperthermia (hyperpyrexia): due to: a. Uncoupling of oxidative phosphorylation. b. Impairment of body cooling mechanisms. c. Dehydration due to loss of fluids by sweating and vomiting
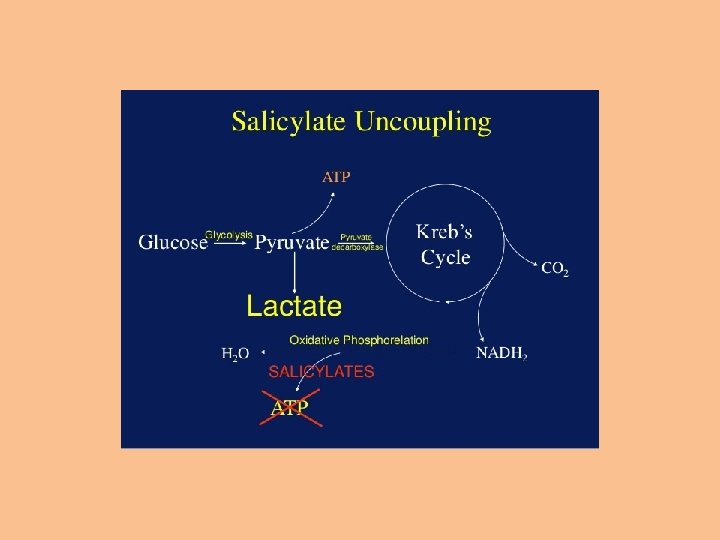

Clinical presentation 11. Acid-base disturbances: Respiratory alkalosis: The predominant early acid-base disturbance - it is due to CNS and respiratory center stimulation → increased depth and rate of respiration→ CO 2 wash → less plasma CO 2 → less carbonic acid is formed→ increase p. H of the blood (respiratory alkalosis), This is compensated by vigorous excretion of HCO 3 in urine (compensatory metabolic acidosis). b. Metabolic acidosis: Rapidly follows alkalosis it is due to: 1. Salicylate is a weak acid. 2. Uncoupling oxidative phosphorylation → decrease production of ATP →anaerobic metabolism → accumulation of lactic and pyruvic acids.

Clinical presentation 3. Stimulation of lipid metabolism to compensate for energy demands→ (accumulation of ketone bodies). 4. Renal compensation for the initial respiratory alkalosis and excretion of blood HCO 3. 5. Impaired renal function resulting from salicylate toxicity leads to accumulation of sulfuric and phosphoric acids (strong acids)

Clinical presentation 6. Dehydration →lactic acidosis 7. increase Na & K with initial HCO 3 diuresis →decrease K →shift of H ions outside cells to maintain electricity 8. decrease liver lactate elimination 9. protein degradation →increase in aminoacids

Investigations Laboratory investigations General: ABG, electrolytes, serum glucose level, prothrombin time, prothrombin concentration, liver function and renal function. Specific: Plasma salicylate level: Not before 4 hours (peak serumconcentration) greater than 30 mg/d. L indicate toxicity and levels greaterthan 100 mg/d. L indicate severe toxicity. Radiological investigations: Chest x ray pulmonary oedema Abdomen x ray concretions

Treatment Emergency and supportive measures: Maintain open airway, cardiovascular and respiratory support. Symptomatic treatment: -correction of acidosis by IV Na. Hco 3 -correction of fluid loss -hyperpyrexia: cold fomentations -vit K, fresh blood and platelets for hypoprothrombinemia and bleeding tendencies -convulsions: diazepam -allergic manifestations: antihistaminic & corticosteroids -pulmonary edema: PEEP ventilation -correct hypoglycemia -correct hypokalemia

Treatment General measures: Decrease absorption: a. Ipecac-induced vomiting. b. Gastric lavage. c. Activated charcoal d. Whole bowel irrigation more effective in enteric coated tablets. e. Cathartics.

Treatment Enhanced elimination: a. Urinary alkalization: effective in enhancing urinary excretion of salicylate through increased ionization of aspirin with subsequent diminished reabsorption at renal tubules and subsequently increase excretion and also correct acidosis and dehydration. Indications: - Mild to moderate toxicity (serum salicylate level 30 -100 mg/dl) - Acidosis - Bolus 1 -2 m. Eq/ kg of hypertonic Na. Hco 3 followed by I. V infusion of 3 ampoules of Na. Hco 3 in 1 L 5% dextrose. Check urine p. H frequently to maintain urine p. H at 7– 8 - method: 150 ml ( 3 ampules) Na. Hco 3 + 850 ml 5% dextrose + 20 -40 m. Eq k / L by 250 ml / hour infusion.

Treatment b. Hemodialysis: very effective in rapidly removing salicylate andcorrecting acidbase and fluid abnormalities. Indications for hemodialysis: Serum levels higher than 100 mg/d. L. Any patient with severe manifestations as renal failure, severe acidosis, progressive deterioration in vital signs, CNS dysfunction and pulmonary edema. c. Repeated-dose activated charcoal: effectively remove salicylate There is no specific antidote for salicylate toxicity

DD of metabolic acidosis with high anion gap. alcohols, methyl alchols, Uremia , diabetic ketoacidosis, paraldhyde, iron, lactic acidosis ethelene glycols , sailcylate.

- Slides: 37