Paper IV Environmental Toxicology UNITI Photochemical Smog Ozone




























- Slides: 43

Paper IV Environmental Toxicology UNIT-I Photochemical Smog, Ozone depletion & Acid rain

Contents Ø Ø Ø Air pollution around the world Transport of Air Pollutants Sources of Outdoor air pollution Photochemical Smog Temperature Inversion Stratospheric Ozone § § Ø Acid rain § Ø depletion Effects Recovery Montreal Protocol Effects Indoor Pollution

Air Pollution Around the World Air quality is deteriorating rapidly in developing countries Shenyang, China ◦ Residents only see sunlight a few weeks each year Developing countries have older cars ◦ Still use leaded gasoline 5 worst cities in world ◦ Beijing, China; Mexico City, Mexico; Shanghai, China; Tehran, Iran; and Calcutta, India

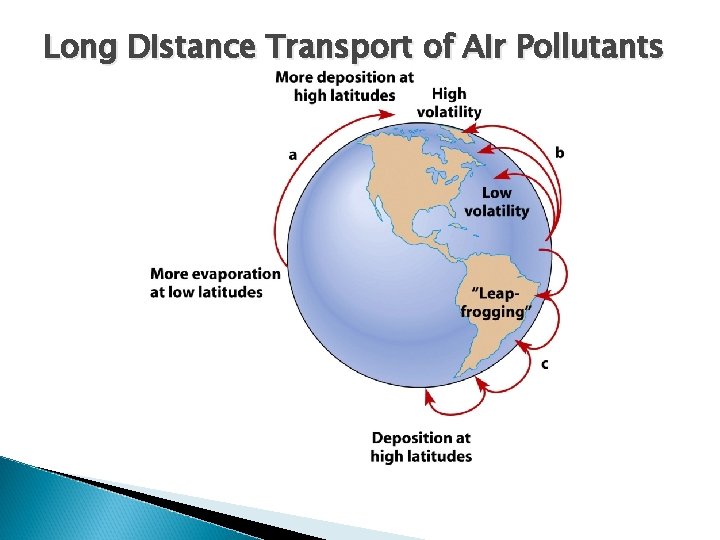
Long Distance Transport of Air Pollutants

Sources of Outdoor Air Pollution Two main sources ◦ Transportation ◦ Industry Intentional forest fires is also high

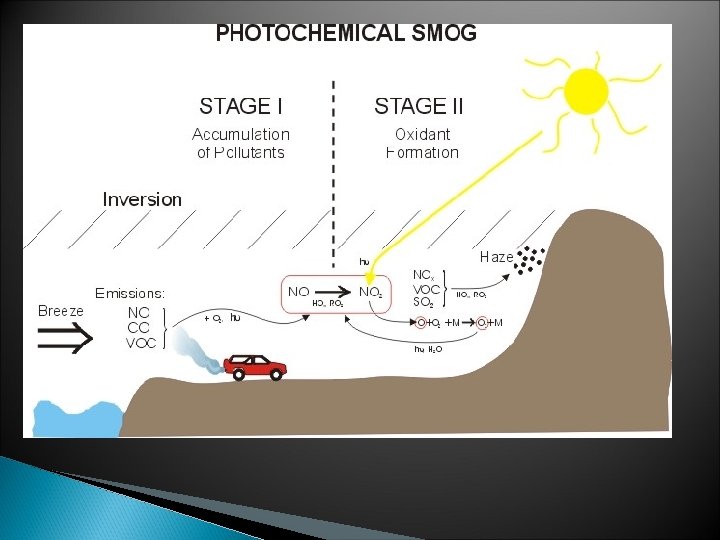
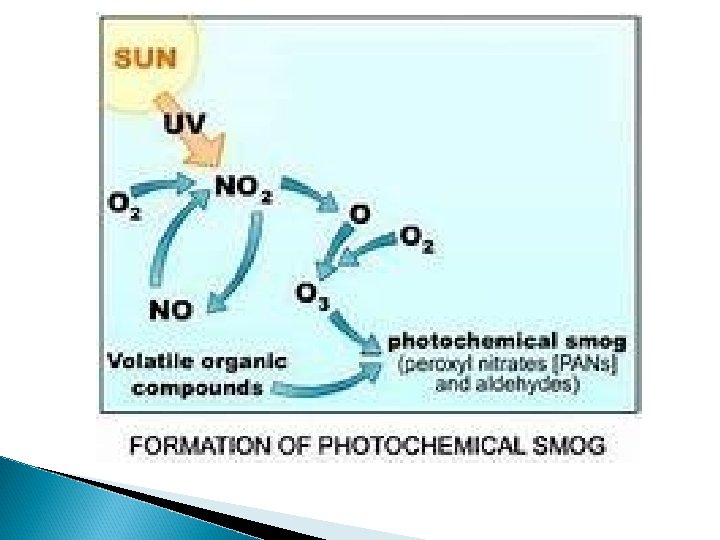
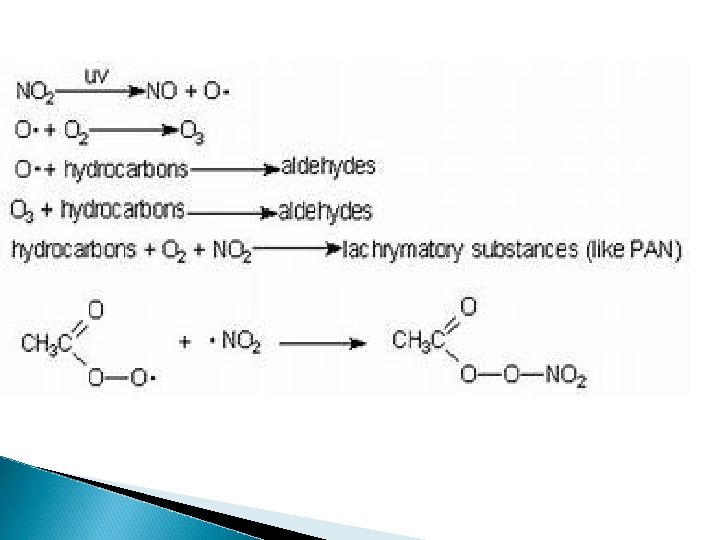
Photochemical Smog (ex: Los Angeles below) ◦ Brownish-orange haze formed by chemical reactions involving sunlight, nitrogen oxide, and hydrocarbons

What’s in smog particulates (especially lead) nitrous oxides potassium Carbon monoxide Other toxic chemicals


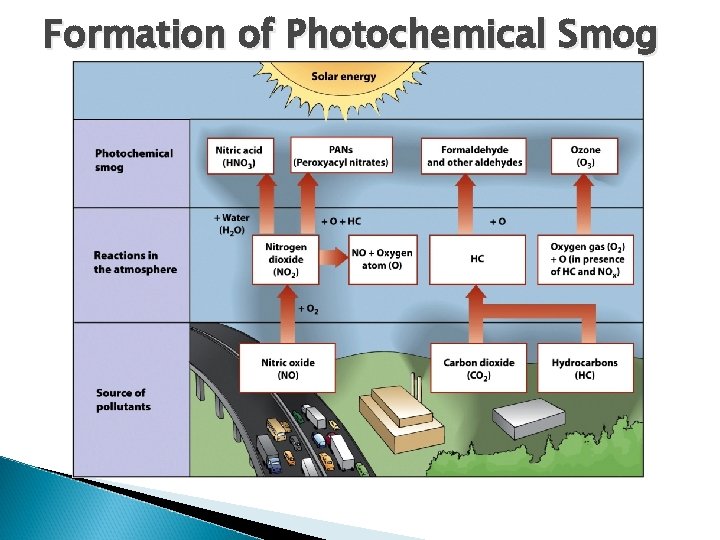
Formation of Photochemical Smog

Sources of Smog in Los Angeles

Case-In-Point Air Pollution in Beijing and Mexico City Beijing (left) Mexico City (above)

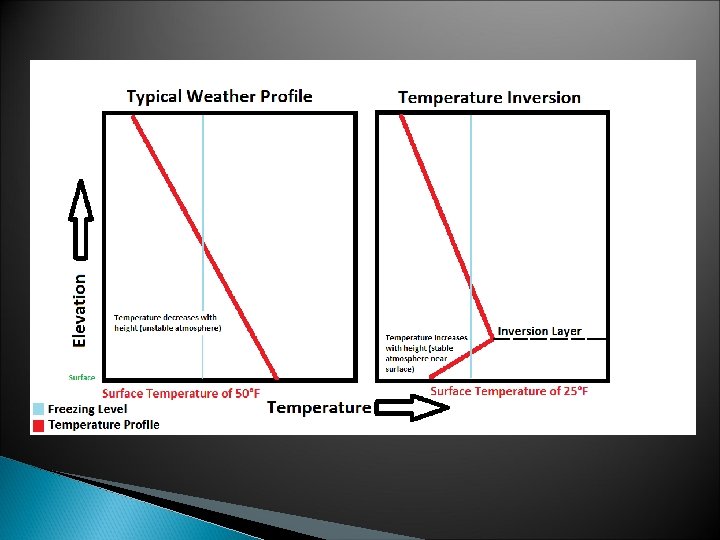
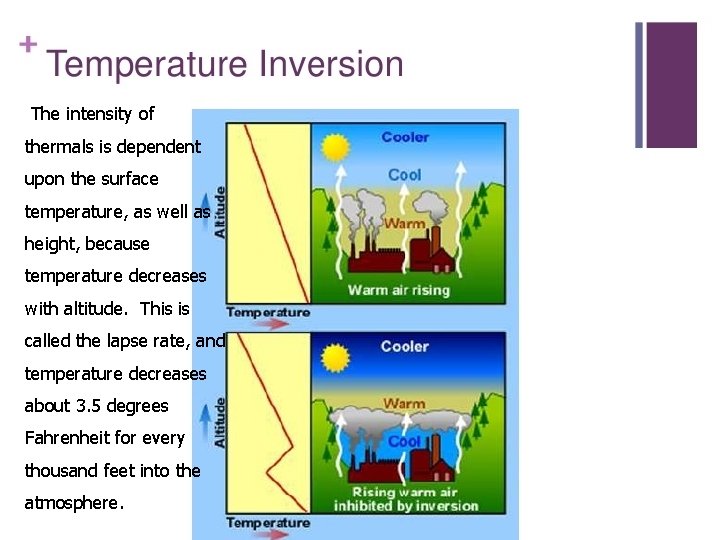
Temperature Inversion ØIn meteorology, an inversion, also known as a temperature inversion, is a deviation from the normal change of an atmospheric property with altitude. ØIt almost always refers to an inversion of thermal lapse rate. Normally, air temperature decreases with an increase in altitude. ØDuring an inversion, warmer air is held above cooler air; the normal temperature profile with altitude is inverted. [2] ØAn inversion traps air pollution, such as smog, close to the ground. An inversion can also suppress convection by acting as a "cap". ØIf this cap is broken for any of several reasons, convection of any moisture present can then erupt into violent thunderstorms. Temperature inversion can notoriously result in freezing rain in cold climates.


The intensity of thermals is dependent upon the surface temperature, as well as height, because temperature decreases with altitude. This is called the lapse rate, and temperature decreases about 3. 5 degrees Fahrenheit for every thousand feet into the atmosphere.



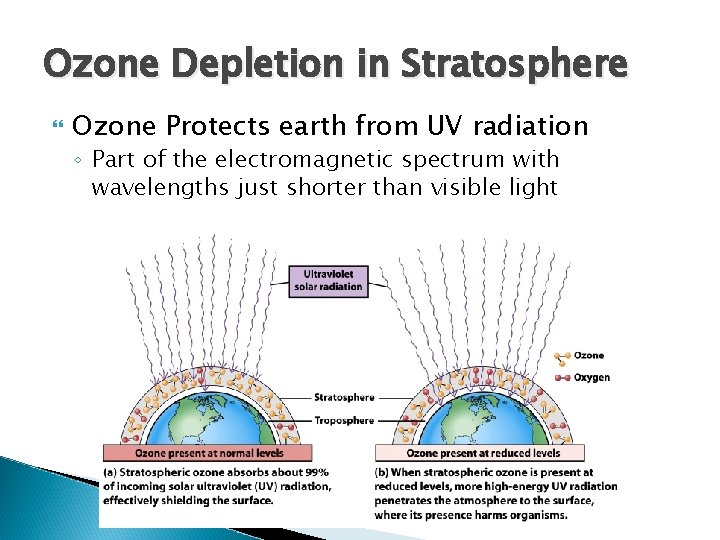
Ozone Depletion in Stratosphere Ozone Protects earth from UV radiation ◦ Part of the electromagnetic spectrum with wavelengths just shorter than visible light

Ozone Depletion in Stratosphere Ozone thinning/hole ◦ First identified in 1985 over Antarctica Caused by ◦ human-produced bromine and chlorine containing chemicals ◦ Ex: CFCs

Ozone Depletion in Stratosphere Hole over Antarctica requires two conditions: ◦ Sunlight just returning to polar region ◦ Circumpolar vortex- a mass of cold air that circulates around the southern polar region Isolates it from the warmer air in the rest of the planet Polar stratospheric clouds form ◦ Enables Cl and Br to destroy ozone

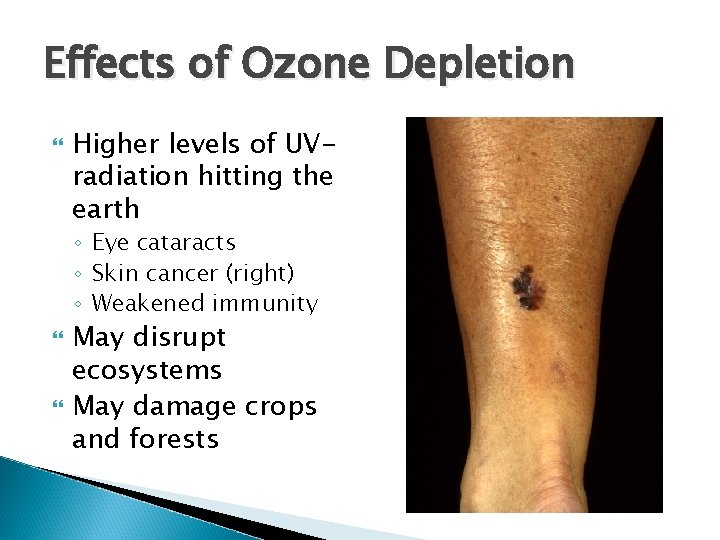
Effects of Ozone Depletion Higher levels of UVradiation hitting the earth ◦ Eye cataracts ◦ Skin cancer (right) ◦ Weakened immunity May disrupt ecosystems May damage crops and forests

Recovery of Ozone Layer Montreal Protocol (1987) ◦ Reduction of CFCs ◦ Started using HCFCs (greenhouse gas) Phase out of all ozone destroying chemicals is underway globally Satellite pictures in 2000 indicated that ozone layer was recovering Full recovery will not occur until 2050

Montreal Protocol Ø Ø Ø The Montreal Protocol is an international environmental agreement with universal ratification to protect the earth’s ozone layer by eliminating use of ozone depleting substances (ODS), which would otherwise allow increased UV radiation to reach the earth, resulting in higher incidence of skin cancers and eye cataracts, more-compromised immune systems and negative effects on watersheds, agricultural lands and forests. Since its adoption in 1987 and as of end-2014, it has successfully eliminated over 98 percent of controlled ODS, helping reverse the damage to the ozone layer. A very significant co-benefit is that it has - during period 1989 -2013 reduced cumulative CO 2 -eq. emissions by 135 billion tonnes. Important challenges remain. The transition from CFCs (high ozone depleting potential or ODP) to intermediate HCFCs (with lower ODP) has been completed, and the final transition is to alternatives that have zero ODP. The challenge is to develop/select alternatives (mainly in refrigeration, air-conditioning, and foam products) that are also climate-friendly.

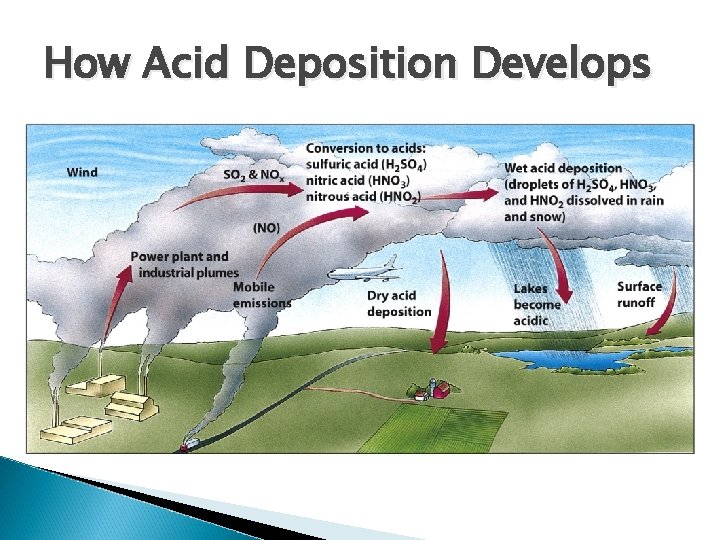
How Acid Deposition Develops

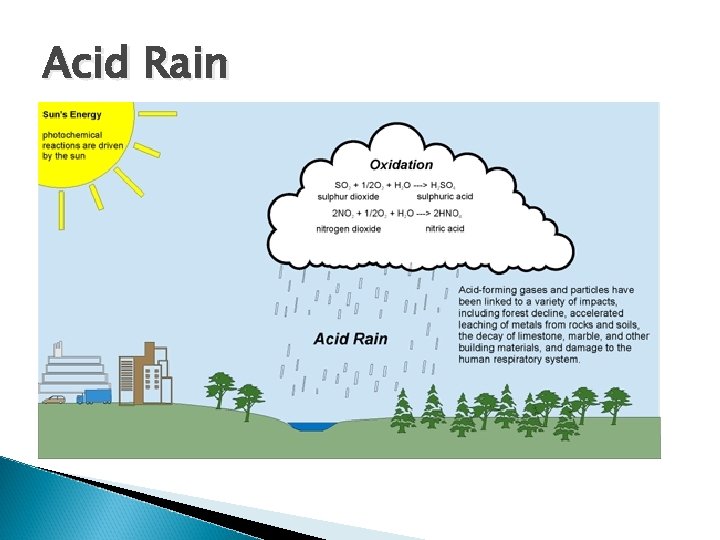
Acid Rain

Acid Deposition Sulfur dioxide and nitrogen dioxide emissions react with water vapor in the atmosphere and form acids that return to the surface as either dry or wet deposition p. H scale

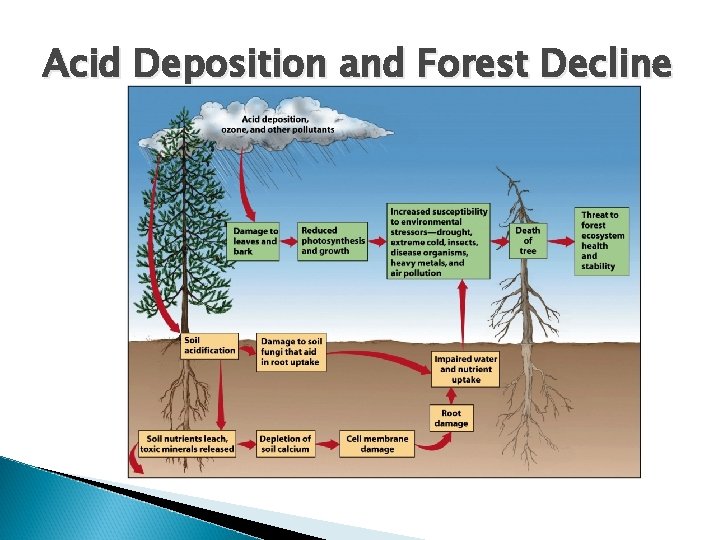
Effects of Acid Deposition Declining Aquatic Animal Populations Thin-shelled eggs prevent bird reproduction ◦ Because calcium is unavailable in acidic soil Forest decline ◦ Ex: Black forest in Germany (50% is destroyed)

Acid Deposition and Forest Decline

Effects on the environment Acid rain Ozone depletion Global warming In human populationrespiratory problems, allergies, strengthens lugs, and a risk for cancer

Acid rain contains high levels of sulfuric or nitric acids contaminate drinking water and vegetation damage aquatic life erode buildings Alters the chemical equilibrium of some soils

Strategies • • Air Quality Management Plan – Development of new technology- electric cars, cleaner fuels, low nitrogen oxide boilers and water healers, zero polluting paints, less polluting BBQ lighter fluids Use of natural gas Carpooling Follow the laws enacted

Urban Emissions • There are small emissions of NOx from industrial processes • The main emissions are from combustion. • There is negligible nitrogen in gasoline or diesel fuels so the nitrogen oxides arise from the N 2 and O 2 in the air. • Sulphur dioxides arise from the sulphur present in most fuels. • Particulate matter describes matter below 10μm aerodynamic diameter.

Role of Engines and Fuel Different engines and fuel combinations give out different emissions in different quantities. Some engines have catalysts which effectively remove part of the harmful gases.

Catalytic Converters and Particle Traps Catalytic converters can be fitted to cars to reduce NOx emissions. CO + HC + NOx Platinum Honeycomb H 2 O + N 2 + CO 2 Particle traps can be used to reduce PM 10 and NOx, but the effectiveness is severely reduced if the fuel the vehicle burns has a high sulphur content. The major target in the battle for cleaner cities is diesel.

Continue…


STRATEGIE The Clean Air for Europe (CAFE) approach: Ø Ø Based on scientific knowledge Using best available, quality-controlled real-world data With close involvement of stakeholders: Project future emissions and air quality resulting from full implementation of current EU legislation Explore scope and costs for further measures Analyze cost-effective policy scenarios Estimate benefits of policy scenarios

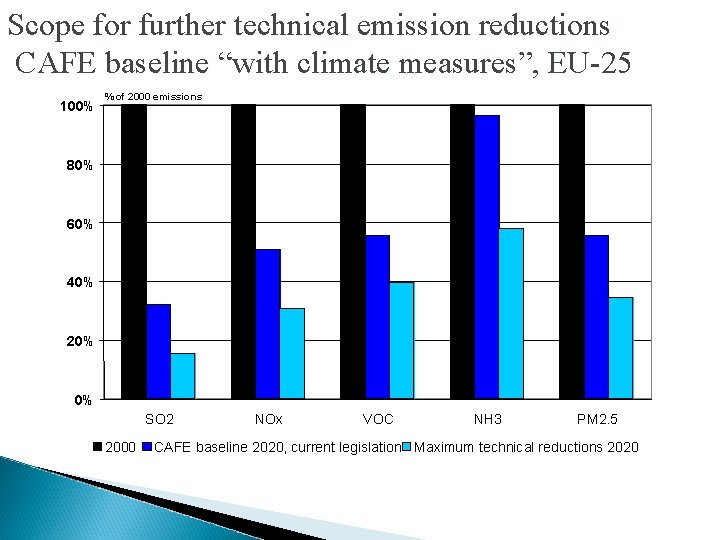
Scope for further technical emission reductions CAFE baseline “with climate measures”, EU-25 100% % of 2000 emissions 80% 60% 40% 20% 0% SO 2 2000 NOx VOC NH 3 PM 2. 5 CAFE baseline 2020, current legislation Maximum technical reductions 2020

Main pollutants used in the CAFE assessment

Particulate Matter (PM ) Pollution - Traffic emissions including diesel engines - Small combustion sources burnng coal and wood - Reductions of SO 2, N 0 x, NH 3 and VOC

Ground level ozone - VOC control to reduce ozone in cities - N 0 x reduction from traffic - Control of N 0 x emissions from ships - Methane reduction

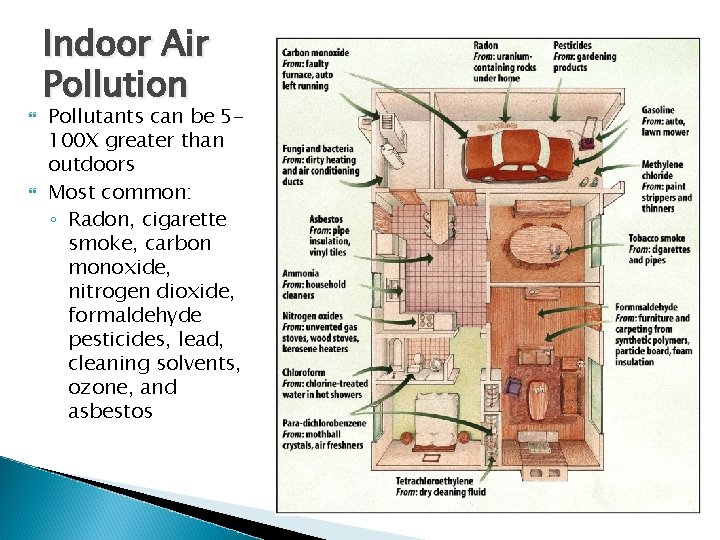
Indoor Air Pollution Pollutants can be 5100 X greater than outdoors Most common: ◦ Radon, cigarette smoke, carbon monoxide, nitrogen dioxide, formaldehyde pesticides, lead, cleaning solvents, ozone, and asbestos

Sources of Indoor pollution Efficient insulation Bacteria Molds and mildews Viruses animal dander and cat saliva plants house dust Mites Cockroaches pollen

THANKS!!