Pandemic Influenza A H 1 N 1 Virus


































- Slides: 44

Pandemic Influenza A (H 1 N 1) Virus Illness Among Pregnant Women in the United States, 2009 Alicia M. Siston, Ph. D, MPH, MS Epidemic Intelligence Service Officer Centers for Disease Control and Prevention NCIRD, Influenza Division

Pandemic 2009 Influenza A (H 1 N 1) Virus (2009 H 1 N 1) • April 21 - 2009 H 1 N 1 pandemic reported • June 11 - WHO raised pandemic level to 6 • Early data on pregnant women with 2009 H 1 N 1 showed increased severity – Higher hospitalization rates than general population – Disproportionate number of deaths

Pregnant Women at Increased Risk for Severe Influenza Illness and Death • Increased risk of mortality during 1918 and 1957 pandemics • Increased risk of hospitalization related to seasonal influenza

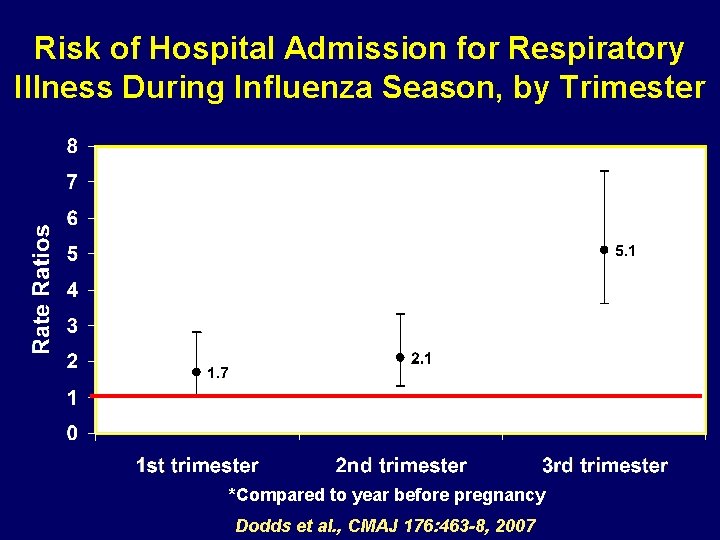
Risk of Hospital Admission for Respiratory Illness During Influenza Season, by Trimester *Compared to year before pregnancy Dodds et al. , CMAJ 176: 463 -8, 2007

Possible Reasons Pregnant Women at Increased Risk from Influenza • Physiologic changes during pregnancy to following systems: – Immune – Cardiac – Respiratory • Concerns regarding fetal effects of treatment might lead to delayed treatment – Mother’s refusal – Physician’s reluctance

Study Objectives • Describe 2009 H 1 N 1 illness severity among pregnant women in United States • Describe impact of antiviral treatment timing

Methods

Data Request to Health Departments • All state health departments and local health departments for Chicago, New York and Washington DC • Requested information on all pregnant confirmed and probable 2009 H 1 N 1 cases

Confirmed and Probable Case Definitions • Confirmed: person reported with acute respiratory illness AND laboratory-confirmed 2009 H 1 N 1 by real-time reverse-transcriptase PCR (r. RT-PCR) or viral culture • Probable: person reported with acute febrile respiratory illness AND a positive test for influenza A and negative influenza r. RT-PCR test for H 1 and H 3

Data Elements Collected • • Demographics Gestational age Illness Onset Severity (hospitalization, intensive care unit (ICU) admission, mechanical ventilation, and death) • Treatment (antiviral type and timing) • Underlying conditions • Delivery

Analysis • Relative risks and 95% confidence intervals (CI): – Risk of severe outcomes by treatment timing – Risk of ICU admission by treatment timing, stratified by pregnancy trimester • Mantel-Haenszel chi-square test and Fisher’s exact test (small sample size comparisons)

Results

Response Rate

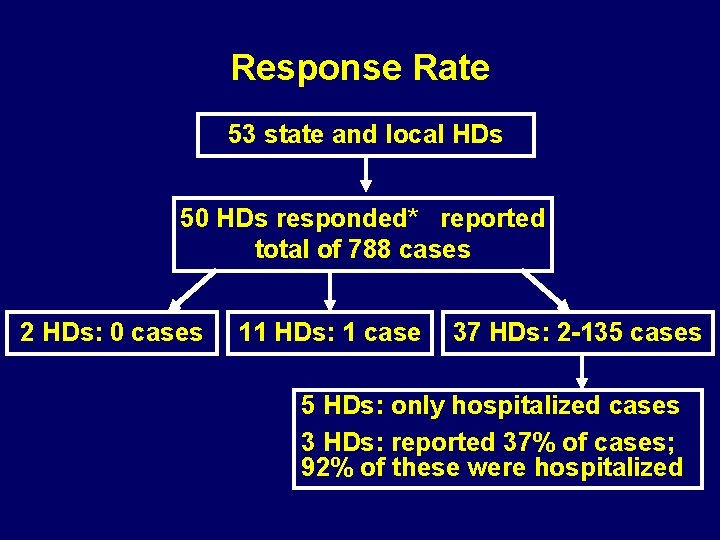
Response Rate 53 state and local HDs

Response Rate 53 state and local HDs 50 HDs responded* reported total of 788 cases

Response Rate 53 state and local HDs 50 HDs responded* reported total of 788 cases 2 HDs: 0 cases

Response Rate 53 state and local HDs 50 HDs responded* reported total of 788 cases 2 HDs: 0 cases 11 HDs: 1 case

Response Rate 53 state and local HDs 50 HDs responded* reported total of 788 cases 2 HDs: 0 cases 11 HDs: 1 case 37 HDs: 2 -135 cases

Response Rate 53 state and local HDs 50 HDs responded* reported total of 788 cases 2 HDs: 0 cases 11 HDs: 1 case 37 HDs: 2 -135 cases 5 HDs: only hospitalized cases 3 HDs: reported 37% of cases; 92% of these were hospitalized

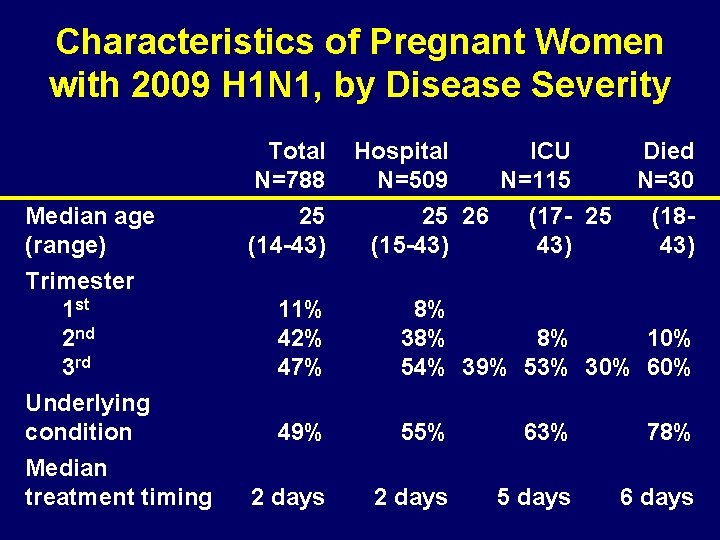
Characteristics of Pregnant Women with 2009 H 1 N 1, by Disease Severity Total N=788 Median age (range) Trimester 1 st 2 nd 3 rd Underlying condition Median treatment timing 25 (14 -43) Hospital N=509 25 26 (15 -43) ICU N=115 (17 - 25 43) Died N=30 (1843) 11% 42% 47% 8% 8% 10% 38% 54% 39% 53% 30% 60% 49% 55% 63% 78% 2 days 5 days 6 days

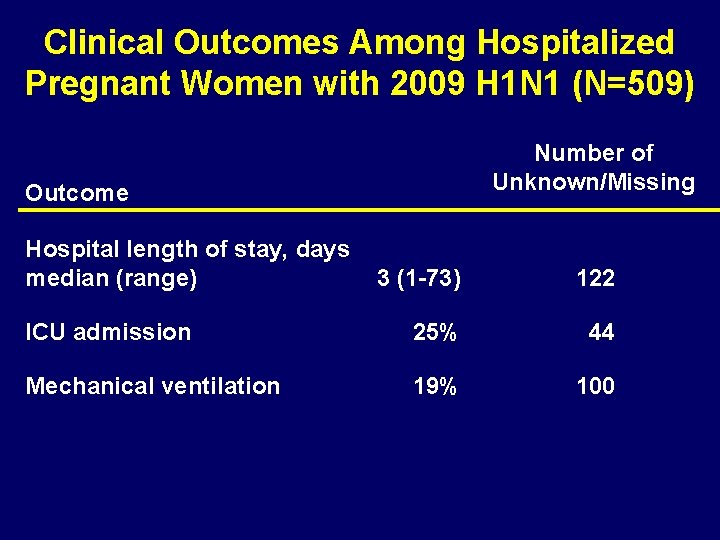
Clinical Outcomes Among Hospitalized Pregnant Women with 2009 H 1 N 1 (N=509) Number of Unknown/Missing Outcome Hospital length of stay, days median (range) 3 (1 -73) 122 ICU admission 25% 44 Mechanical ventilation 19% 100

Antiviral Treatment Timing Definitions Treatment timing (days)= (treatment start date)-(illness onset date) Early <2 days Intermediate 3 -4 days Late >4 days None No treatment given

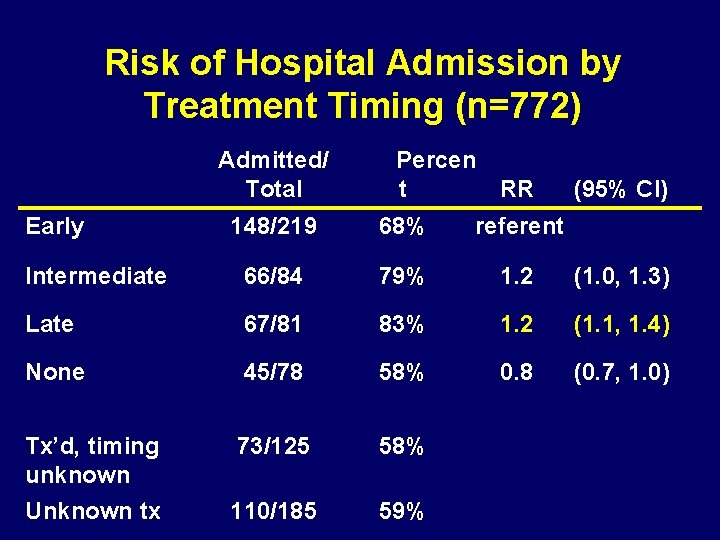
Risk of Hospital Admission by Treatment Timing (n=772) Admitted/ Total Early Percen t RR (95% CI) 148/219 68% referent Intermediate 66/84 79% 1. 2 (1. 0, 1. 3) Late 67/81 83% 1. 2 (1. 1, 1. 4) None 45/78 58% 0. 8 (0. 7, 1. 0) Tx’d, timing unknown 73/125 58% Unknown tx 110/185 59%

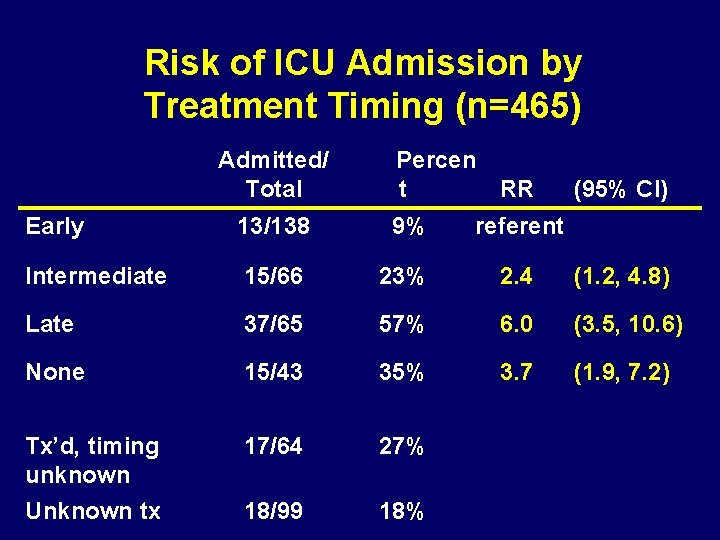
Risk of ICU Admission by Treatment Timing (n=465) Admitted/ Total Percen t RR (95% CI) Early 13/138 9% referent Intermediate 15/66 23% 2. 4 (1. 2, 4. 8) Late 37/65 57% 6. 0 (3. 5, 10. 6) None 15/43 35% 3. 7 (1. 9, 7. 2) Tx’d, timing unknown 17/64 27% Unknown tx 18/99 18%

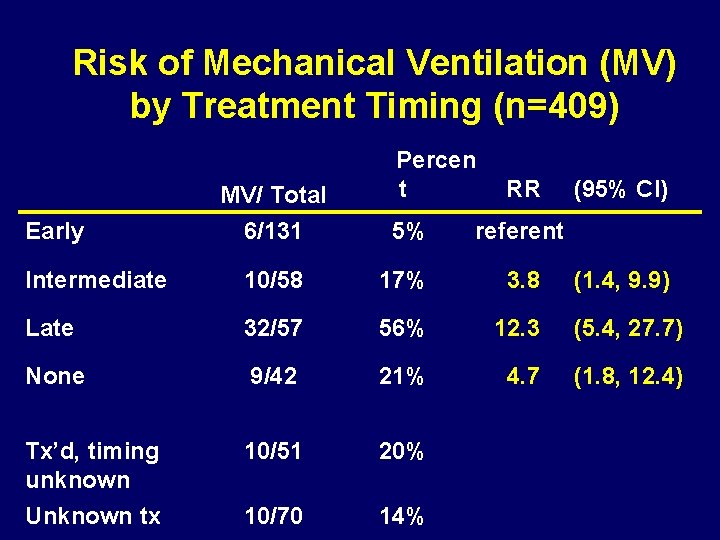
Risk of Mechanical Ventilation (MV) by Treatment Timing (n=409) Percen t RR (95% CI) MV/ Total 6/131 5% referent Intermediate 10/58 17% 3. 8 (1. 4, 9. 9) Late 32/57 56% 12. 3 (5. 4, 27. 7) None 9/42 21% 4. 7 (1. 8, 12. 4) Tx’d, timing unknown 10/51 20% Unknown tx 10/70 14% Early

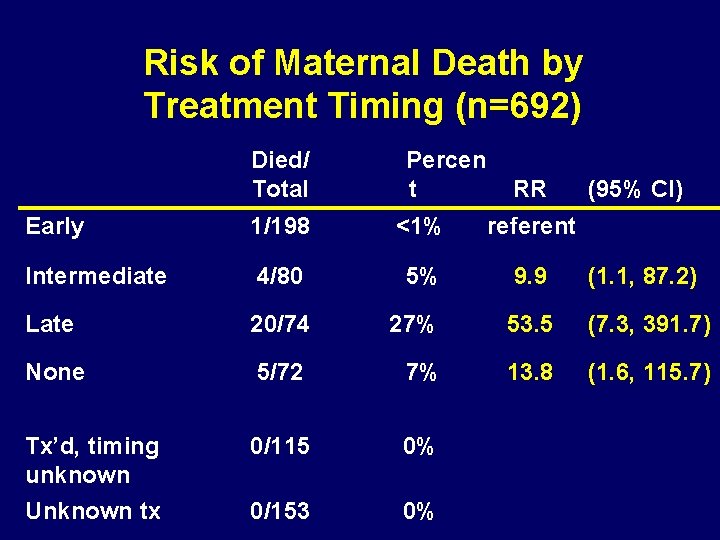
Risk of Maternal Death by Treatment Timing (n=692) Died/ Total Percen t RR (95% CI) Early 1/198 <1% referent Intermediate 4/80 5% 9. 9 (1. 1, 87. 2) Late 20/74 27% 53. 5 (7. 3, 391. 7) None 5/72 7% 13. 8 (1. 6, 115. 7) Tx’d, timing unknown 0/115 0% Unknown tx 0/153 0%

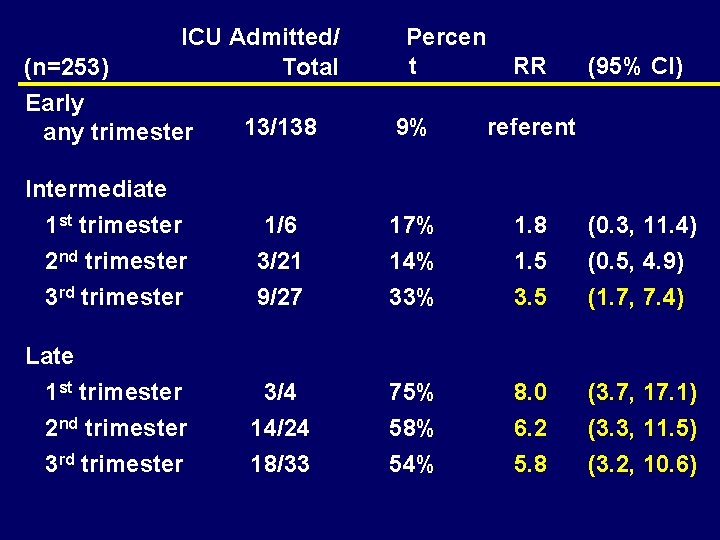
Impact of Trimester and Treatment Timing on ICU Admission

(n=253) ICU Admitted/ Total Percen t RR (95% CI) Early any trimester 13/138 9% referent Intermediate 1 st trimester 2 nd trimester 3 rd trimester 1/6 3/21 9/27 17% 14% 33% 1. 8 1. 5 3. 5 (0. 3, 11. 4) (0. 5, 4. 9) (1. 7, 7. 4) Late 1 st trimester 2 nd trimester 3 rd trimester 3/4 14/24 18/33 75% 58% 54% 8. 0 6. 2 5. 8 (3. 7, 17. 1) (3. 3, 11. 5) (3. 2, 10. 6)

Updated Data on Severe Outcomes (Deaths and ICU Admissions) August 21 – December 31, 2009

Pregnancy Flu Line • Launched mid-October, 2009 • Requested all state and local health departments to report: – Any pregnant women confirmed with influenza, and • • Positive rapid test r. RT-PCR Direct/indirect fluorescent antibody test Viral culture – Admitted to ICU or died • Retroactive reporting to August 21, 2009

Present Analysis and Updated Data on ICU Admissions in Pregnant Women Present N Flu Line N (%) 115 163 Total N (%) Total Trimester 1 st 2 nd 3 rd Unknown 278 8 (7) 8 (5) 38 (33) (39) 51 (44) (52) 18 (16) 63 16 (6) 101 85 (36) 136 7 (49) 25 (4) (9) Mech. Vent. Yes No Unknown 77 (67) 89 (55) 34 (30) (39) 4 (4) 63 166 (60) 97 11 (35) 14 (7) (5)

Present Analysis and Updated Data on Maternal Deaths in Pregnant Women Present Total Trimester 1 st 2 nd 3 rd Unknown Mech. Vent. Yes No Unknown N Flu Line N (%) 30 3 (10) 1 (4) 9 (30) (23) 18 (60) (69) 0 Total N (%) 26 56 6 4 (7) 18 (27) 1 (64) (4) 15 36 1 (2) 30 (100) 0 55 (98) 0 0 25 (96) 0 1 (4) 1 (2)

Discussion • Disproportionate number of deaths – Pregnant women represent 1% of US population – Pregnant women accounted for 5% of 2009 H 1 N 1 deaths • Data are consistent with other US and international studies that pregnant women are at increased risk for serious illness and death

Antiviral Treatment Timing • Delayed antiviral treatment associated with more severe illness and death – Increased illness duration – Increased symptom severity and mortality • Data suggest some benefit to those treated within 4 days of illness onset – consistent with seasonal influenza data in hospitalized patients

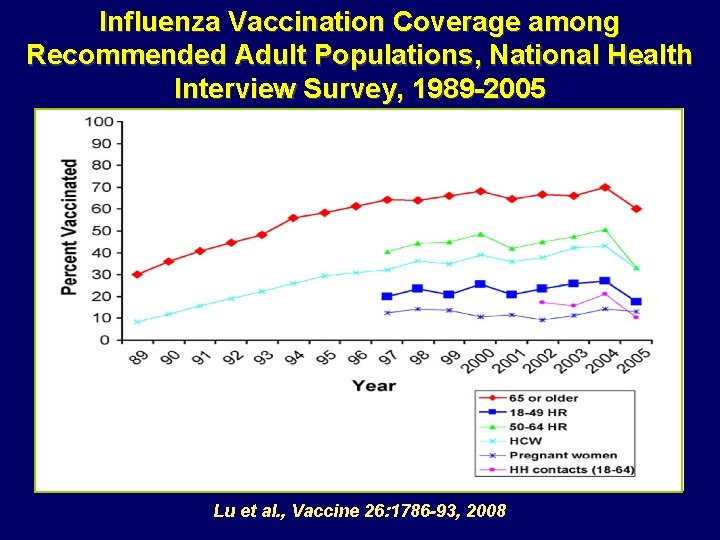
Data Support Public Health Recommendations for Pregnant Women • Early treatment with antiviral medications of pregnant women who present with possible influenza • Vaccination with 2009 H 1 N 1 monovalent vaccine • May present challenge as pregnant women have been least likely in the past to receive influenza vaccination

Influenza Vaccination Coverage among Recommended Adult Populations, National Health Interview Survey, 1989 -2005 Data from National Health Interview Survey Lu et al. , Vaccine 26: 1786 -93, 2008

Possible Reasons for Delayed Treatment • Concerns regarding fetal effects of treatment – Mother’s refusal to receive treatment – Physician’s reluctance to prescribe treatment • Delay in seeking medical care • Inappropriate reliance on rapid influenza test (sensitivity 10 -70% for 2009 H 1 N 1 virus)

Trimester Effect • Data similar to previous pandemics and nonpandemic influenza seasons – Higher proportion of cases among pregnant women in 2 nd and 3 rd trimesters • However, severe cases occurred during all trimesters, and women in their first trimester might not know they are pregnant • Due to small sample size, difficult to assess importance of treatment timing vs. pregnancy trimester in predicting outcomes

Limitations: Missing Data • Large amount of missing data – Data not missing at random – Missing data decreased as severity increased • Stratified analyses resulted in small sample sizes, and therefore unstable estimates (eg, risk estimates for maternal death) • Sensitivity analysis performed to estimate effect of missing data for treatment timing; resulted in slightly lower risk estimates

Limitations: Biased Sample • Study population represents most severe cases – Pregnant women had to have sought medical care and received diagnosis to be included in study – Testing criteria changed; testing limited to severe cases as outbreak progressed • States used different reporting criteria • Reporting criteria changed for some states during the data request period

Conclusion • Most complete national description of pregnant women with 2009 H 1 N 1 to date • Pregnant women disproportionately represented among deaths • Early antiviral treatment was associated with fewer ICU admissions and deaths • Data support Advisory Committee on Immunization Practices recommendations identifying pregnant women as target group for 2009 H 1 N 1 vaccine

Recommendation for Future Study Use multivariate analysis to explore impact of pregnancy trimester and antiviral treatment timing – Need unbiased sample – Need more complete data

Acknowledgments • State and Local Health Departments • Pandemic H 1 N 1 Influenza in Pregnancy Working • Group CDC – Margaret Honein, Ph. D – Sonja Rasmussen, MD – Denise Jamieson, MD – Lyn Finelli, Dr. PH – Katherine Seib – Kim Newsome, MPH The findings and conclusions in this presentation are those of the authors and do not necessarily represent the views of the Centers for Disease Control and Prevention.

Clinical Outcomes of All Reported Pregnant Women with 2009 H 1 N 1 (N=788) Unknown/ Missing (Yes/Total) % (509/773) 66% 16 Maternal death (30/692) 4% 96 Preterm delivery (51/169) 30% * Outcome Hospital admission *Unable to determine